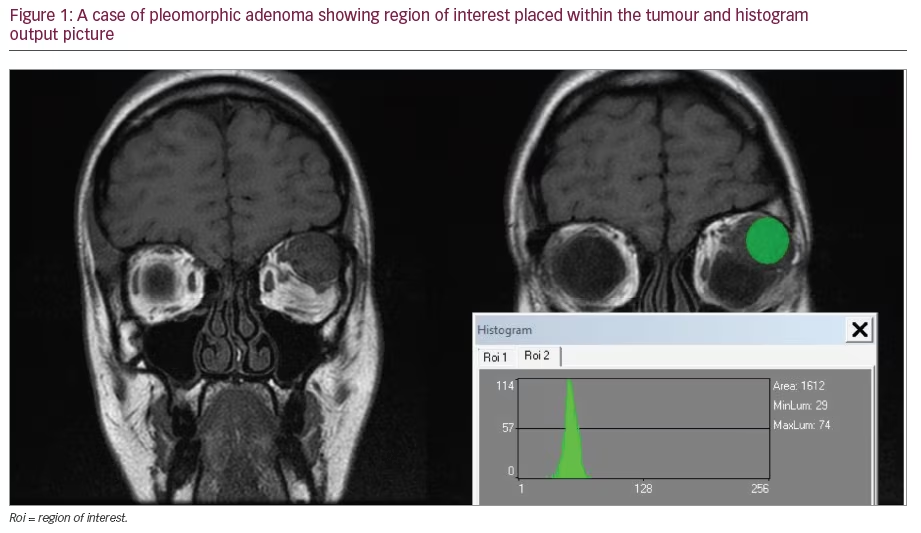
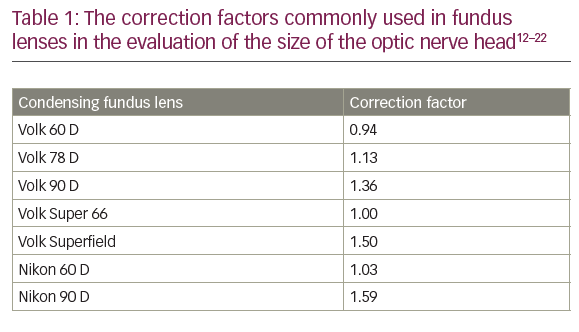
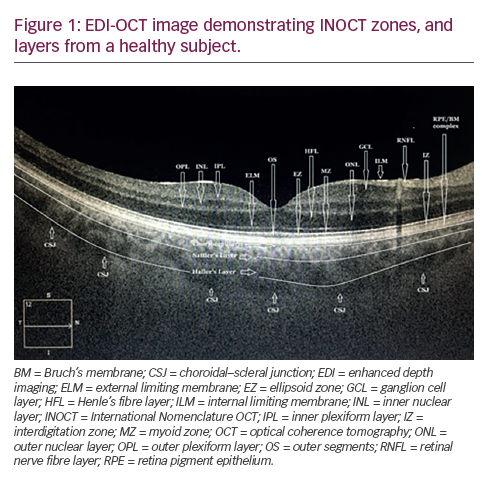
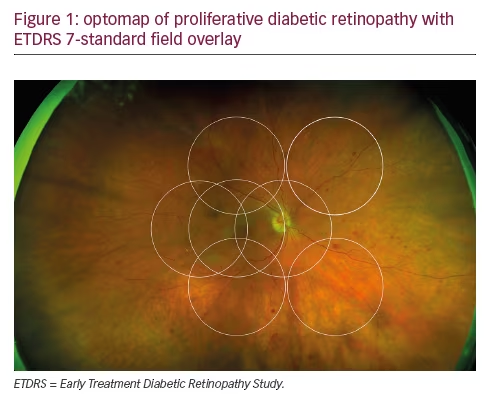
It is 16 years since David Huang and colleagues in the James Fujimoto laboratory at the Massachusetts Institute of Technology developed optical coherence tomography (OCT).1 Following the introduction of Zeiss Stratus Optical Coherence Tomography (third-generation OCT or OCT 3) by Carl Zeiss Meditec Inc., it became a ‘gold standard’ for ocular examination of macular diseases. The technology used during examination enables the presentation of the retinal structure analogous to histological sections. This allows visualisation of the retinal and optic nerve pathology. Nowadays, no retina specialist can imagine his or her work without OCT. Various clinical disorders have been presented in OCT. As new developments in ophthalmic therapy emerged, it became clear that perfect visualisation of the retina and measurements of retina thickness allow monitoring of therapy with triamcinolone or anti-vascular endothelial growth factor (VEGF) drugs.
OCT is based on the measurement of back-reflected infrared light. This is performed indirectly because of the high speed at which light spreads. The technique is called low-coherence interferometry. In this technique, a back-reflected beam of light is interfered with by a reference beam with a known echo time delay. The optical interference measures the echo time delay of light from the ocular structures and gives us axial information similar to A-scans in ultrasonography. The sum of many A-scans provides the opportunity to produce a cross-sectional image similar to a B-scan. Even though this technology was so revolutionary to ophthalmology it has still two important disadvantages, the first with resolution and the second with imaging speed. Although the resolution of Stratus OCT is the best-known commercially available diagnostic instrument in medicine, the resolution is still too low to detect early structural changes in ocular tissue. This can be improved by using a more advanced light source such as femtosecond titanium:sapphire lasers.2–4 This technology allows axial image resolution more than three times higher than that of Stratus OCT to be achieved. However, the application of this technology is limited because this light source is large and expensive. Low-coherence interferometry does not allow increased imaging speed, nor would a longer examination be more precise because it increases the amount of artefacts created by eye movements. Current commercially available systems allow 512–1,024 axial scans per image up to 1.9 seconds.5 With these aspects in mind, the search for improvement needed new technology.
It would appear that the recently developed spectral OCT (SOCT) – relating to the Fourier Domain OCT – might solve the resolution and speed problems. Wojtkowski et al.6 were the first to report retinal imaging using SOCT. Wojtkowski et al./7 presented a combination of Fourier Domain OCT and ultra-high-resolution OCT. SOCT can measure all echoes of light from different delays simultaneously. This increases the speed of imaging by 50–100 times, thus providing much more information. The sensitivity of the examination is increased. In recent publications we observe growing interest in Fourier Domain SOCT and rapid development in this field. The use of femtosecond titanium:sapphire technology will probably not be so important in the future since rapid improvements in super-luminescent diode technology have been demonstrated. They also allow the achievement of ultra-high axial image resolutions of 3.1μm.8
Another consideration may be time-encoded frequency domain OCT, such as swept source OCT or optical frequency domain imaging. This technology was presented recently by Bajraszewski et al. and enables the achievement of high-resolution imaging with an 800nm light source.9 These briefly mentioned facts represent enormous development in the field of OCT over recent years. However, these fantastic improvements are poorly reflected in the increased knowledge of ophthalmologists in this field. Why is this so? The answer is easy. A lot of time is needed to introduce the commercially available instruments. The majority of ophthalmologists have no access to the investigational devices. Additionally, information shown by the centres equipped with experimental high-resolution OCT cannot present numerous patient data because the single examination is complicated and lasts a long time, and the device is too large for a standard examination room. Fourier Domain OCT examination shows new possibilities of technique, as presented by Wojtkowski et al.10 They have shown that not only is better resolution of single scans possible, but also OCT fundus images, 3D images and multiple scans in the same place.
Multiple scans present the pulse of the vessels, quantitative measurement and mapping of intra-retinal layer thickness and topographic features of the optic disc. A large field for exploration will be presented by ophthalmologists in the near future. In February 2006, our group examined the first commercially available Fourier Domain OCT (Copernicus, Optopol, Zawiercie, Poland). Since the examination did not take long and the picture quality was similar to that presented in literature for high-resolution OCT, we examined the majority of our patients to learn more from these images and to find out what the clinical implications of the examination were. We will present this technique, using the examples of macular degeneration and vitreomacular interface diseases.
Clinical Information on Spectral Optical Coherence Tomography
Age-related Macular Degeneration
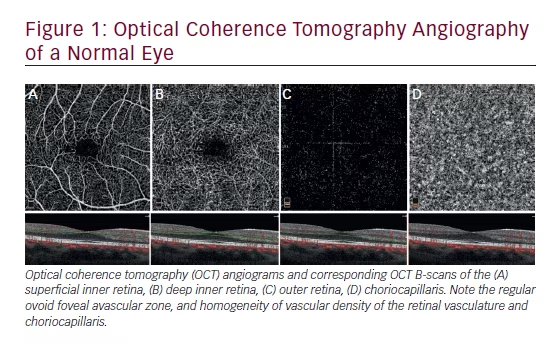
Early detection of active neovascular membrane seems especially important in the era of anti-VEGF therapy. Van de Moere et al. noted that the most important OCT parameters correlated with activity of choroidal neovascularisation (CNV) after photodynamic therapy (PDT) were cystoid or sponge-like macular oedema and the presence of subretinal fluid.11 In highresolution SOCT it is possible to identify even small areas of subretinal fluid with the presence of a thickening of the retina. The short scanning time enables an acquisition sequence of consecutive B-scans. This enables 3D scanning of the macular area. By digital processing of 3D scans, topographic maps of retinal thickness without extrapolation and retinal pigment epithelium deformation maps can be achieved (see Figure 1). These maps play an important role in monitoring patients with early age-related macular degeneration (AMD) changes, or in follow-up patients after VEGF therapy.
Vitreomacular Interface Diseases
Witkin et al.,12 in an ultra-high-resolution OCT study, presented that epiretinal membranes were evident in almost all cases of lamellar macularholes. Our study,13 performed with ‘Copernicus’ SOCT, showed that lamellar macular holes should be classified as a subtype of epiretinal membraneformation. The higher number of epiretinal membranes found in cases with lamellar macular hole was possible probably due to the higher resolution of SOCT. This also enabled us to detect photoreceptor defects that may play an important role in the explanation of functional results. Hangai et al.14 presented 3D images of macular holes. It was shown that detachment of posterior vitreous with attachment of the vitreous to the flap is present. Better resolution of the technique allowed the presentation of early changes in the photoreceptor layer in the form of elevation of the photoreceptor layer and disruption of external limiting membrane (ELM). Ko et al.,15 on the basis of ultra-high-resolution OCT, presented that after macular hole surgery the following retinal abnormalities appear:
• outer foveal hyporeflective defects;
• persistent foveal detachments;
• foveal lesions of moderate reflectivity;
• epiretinal membranes; and
• nerve fibre defects.
We also observed retinal pigment epithelium defects, photoreceptor layer irregularities and lesions of all retinal layers in fovea (unpublished data). Our observations presented at the combined American Society of Retina Specialists/European Vitreoretinal Society (ASRS/EVRS) meeting in Cannes have shown some dynamics of these findings, which present a healing process during the first months after surgery.16 We have also proved that photoreceptor defects measured on a 3D plane correspond statistically significantly with post-operative visual acuity (unpublished data). Examination of the eyes after retinal detachment and subsequent repair surgery17 showed the presence of distortion of the photoreceptor inner/outer segments junction, pockets of subretinal fluid, cystoid macular oedema and epiretinal membranes. We observed photoreceptor defects in all patients after retinal detachment repair, but the size of photoreceptor defect was greater in patients after retinotomy than in patients after pars plana vitrectomy or cerclage combined with pars plana vitrectomy. Following retinal detachment surgery, the exact extent of the photoreceptor damage positively correlates with final visual outcomes (unpublished data).
Conclusion
In recent literature, a lot of information can be found about physics and new developments in OCT technology, but data concerning ocular pathology are still limited. Different companies work on commercially available SOCT and some of them try to combine SOCT with angiography or scanning laser ophthalmoscopy. If ophthalmologists bring these instruments into daily clinical practice there will be a great expansion of knowledge concerning different eye diseases. It seems that in the near future – in addition to slitlamp illumination of the eye – ophthalmologists will have to learn about microscopic changes in different diseases in order to observe and treat them according to the completely new knowledge achieved by SOCT examination in both the anterior and posterior segments of the eye.