Optical coherence tomography (OCT), first developed for ophthalmic imaging in the 1990s,1 has become a ubiquitous and invaluable tool in the retinal clinic. While it will never replace stereoscopic biomicroscopy, it has demonstrated clear advantages. Adjunctive OCT scanning objectively provides the examiner with 2D cross-sections of the retina, while newer machines are able to reconstruct 3D images. Adequate clinical examination of the vitreoretinal interface requires a fundus contact lens and suffers from inter-observer variation, subjective documentation, and missed pathology. From a patient’s perspective, contact lens examinations can be uncomfortable and distressing. The greater detail afforded by OCT scanning also enhances the clinician’s ability to clinch diagnoses and further vitreoretinal research.
The first-generation (time-domain [TD]) OCTs were capable of resolutions only between 10 and 20μm. Second-generation (spectral-domain [SD]) OCTs have a resolution of 5–6μm. We eagerly await the commercial availability of third-generation (swept-source [SS]) OCT, which yields resolutions as low as 1–3μm. These devices have already transformed the field of retinal research, allowing visualization and measurement of retinal vascular blood flow. Full-field OCTs are still not available for clinical use, but the promise of single-micron resolution is not far off.
Optical Coherence Tomography Imaging
Time-domain Optical Coherence Tomography
Conceptually, OCT operates on the same physical principles as an ultrasound scan, except it uses light as the carrier signal. As such, the spatial resolution of an OCT is much higher than a conventional 10–20MHz ultrasound as a result of the naturally shorter wavelength of light. The source of light in an OCT is produced by a super-luminescent diode, femtosecond laser or, of late, white light.2
Simplistically, the OCT works by splitting a beam of light into two arms: a reference arm and a sampling arm. First-generation OCTs are TD-OCT, so named because the length of the reference arm is varied with time in order to correlate with the back-reflected sample arm. This is achieved with the use of an adjustable mirror of known distance within the device. The sample arm is focused onto the retina with the use of an in-built 78D lens. The sample beam is reflected off the structures in the eye and is re-combined with the reference beam by using a Michaelson interferometer within the unit. A single cycle of this process yields one A-scan. This single scan comprises data on the distance the sample arm has travelled and the back-reflectance and back-scatter of the beam. Tissue layers at varying depths and optical characteristics produce differing reflective intensities. As in an ultrasound scan, in order to produce a B-scan image, multiple A-scans are obtained in rapid succession across the area of interest. Software combines this information to produce a 2D image either in greyscale or with arbitrary false-coloring. The result is a cross-sectional scan, a reconstructed 3D topographical image, quantitative thickness measurements or, more recently, z-plane or coronal scans.2
Frequency-domain Optical Coherence Tomography
Frequency-domain OCT (FD-OCT) operates on a slightly different principle. Instead of matching reference and sample arm lengths, the axial scan is calculated by Fourier-transformation of the acquired spectral interference fringes generated by the interaction of the reference and sample arms. Therefore, the reference arm in an FD-OCT does not have to move, allowing dramatically quicker scan acquisition speeds. SD-OCT, also known as Fourier-domain OCT, is based on the underlying principle of FD-OCT, but SD-OCT is able to extract more information in a single scan as it distributes several optical frequencies onto a detector stripe.3 Time-encoded FD-OCT, also known as SS-OCT, is again based on FD-OCT. However, unlike Fourier-domain OCT, which emits multiple optical frequencies all at once, the SS-OCT emits multiple frequencies in single successive steps. At present, SS-OCT is not commercially available, but it has shown promise in recent studies that have demonstrated significantly improved resolution and image penetration for imaging structures and pathology deep in the retina because it operates in the 1,050nm wavelength range.4,5 Each mode of frequency encoding has its advantages and limitations, but a detailed analysis is beyond the scope of this article.
Full-field Optical Coherence Tomography
The latest-generation OCT is the full-field OCT (FF-OCT), another version of the TD-OCT, and uses a broadband (white) light source instead of a laser or super-luminescent diode.2 Also known as T-scan (transverse) OCT or en face OCT, it acquires tomographic data by acquiring multiple coronal scans instead of the usual axial scans (A-scans) of the previously described TD-OCT.6 It should be noted that SS-OCT is also capable of this scanning scheme.7 The advantage of this method is that it is possible to generate not only B-scans but also C-scans (coronal) alongside a simultaneous, conventional fundus image (coronal plane). This presents a unique opportunity for developers to combine other imaging modalities with OCT, as the focusing requirements (in z-plane) are relaxed and dynamic focusing is possible. The downside is that image acquisition times are, at present, significantly longer, taking at least a few seconds compared with SD-OCT, which is able to acquire a full scan in under one second.8 Like a magnetic resonance imaging (MRI) scan, full data acquisition is required before a coronal scan can be produced with the FF-OCT due to the concavity of the fundus.6
Scanning Modes
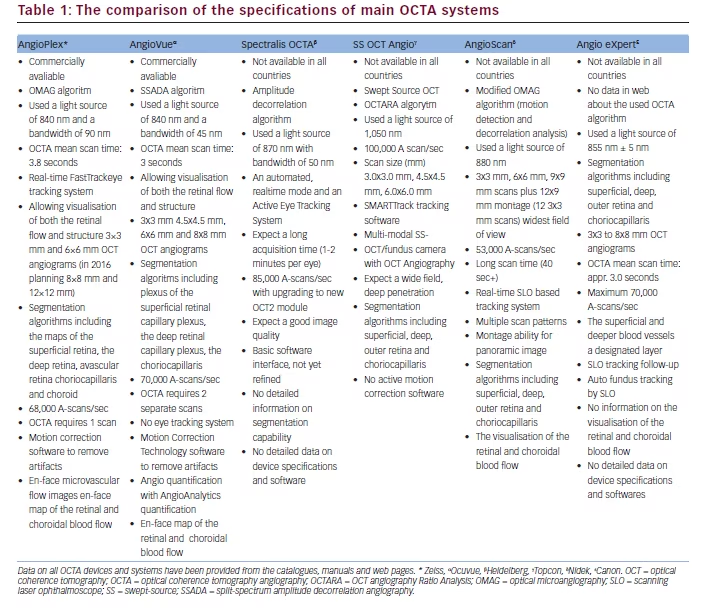
As briefly mentioned above, actually obtaining a scan requires sequential scanning in the form of multiple A-scans for TD-OCT and SD-OCT, and C-scans for FF-OCT. The volume of information obtained can be measured in volumetric pixels (voxels).7 Increasing the number of voxels captured can be achieved by either increasing the scanning frequency rate or using multidetector arrays, or both.3,7 Commercially available SD-OCT scanners became available in early 2006, and most have imaging speeds of 25,000 axial scans per second with an axial resolution of between 5 and 7μm (see Table 1). Technological optimization of these variables has produced OCT scanners that achieve up to 250,000 axial scans per second while maintaining axial resolution at 8–9μm, thus yielding more than 100 megavoxels.7 Clinically, this translates into a high-resolution macular scan in 1.3 seconds or even the potential to measure blood-flow velocities in vessels as narrow as 13.64μm.9,10 Higher scanning speeds are less likely to suffer from motion artefact.11 Signal strength is the measure of the amount of reflected light received by the scanner. It is graded to serve as a proxy measure of scan quality. A signal strength of at least seven should be aimed for in order to obtain consistently accurate results.12,13 High scan rates can reduce signal strengths to below this.7
Numerous software-controlled scanning protocols translate voxel datasets into clinically representative images. Post-scan software processing also determines surface segmentations such as the internal limiting membrane (ILM) and retinal pigment epithelium (RPE), among others, and assigns a false color to each layer depending on the signal reflectance (see Figure 1). However, greyscale and proportion-corrected OCT images reveal a finer gradation of signal reflectance and can be used to demonstrate additional information not present in false-color images.14 Software outlining of the ILM is necessary for the calculation of retinal thickness, however, it has been shown to be inaccurate in up to 19% of scans.15 New algorithms are being constantly developed to overcome these shortfalls.15–17 In a direct comparison of TD-OCT and FD-OCT, the measurements obtained were comparable. Results depended more on the algorithm used than the hardware, emphasizing the need for robust software.18 It is also pertinent that the clinician always double-checks these analyses as artefacts exist (43.2% in one study, with 30% requiring manual remeasurement due to spurious central point thickness [CPT]).19
Clinical Applications
General Ophthalmic Applications
Following its introduction for retinal and coronary imaging in the 1990s, OCT has diversified to other medical fields as varied as hair-growth analysis for discovering steroid-doping athletes.20 In ophthalmology, OCT is used to image the anterior segment,21,22 retina and, with recent technological developments, the choroid in greater detail than before.23 OCT is well-established in glaucoma practice, allowing the quantification of nerve fiber loss and disc morphology.24
Macular Topography
OCT provides an objective quantification of retinal structures, unlike fundus fluorescein angiography, autofluorescence or retinal photography. Additionally, the rapid, non-contact, non-invasive nature of OCT scanning lends itself to the busy vitreoretinal clinic. Obtaining an OCT image requires mydriasis to ensure an artefact-free scan. Non-mydriatic scanning is possible but may result in vignetting of the macular scan as the edges of the sample beam are clipped by the pupillary margin. If it is not possible to dilate the patient, scanning in a dark environment would reduce this phenomenon as the vast majority of OCT scanners use near-infrared light, which does not induce pupillary constriction. The macula is visualized on the monitor and the area of interest is aligned with the aid of fixation targets. It is useful to ensure the patient blinks several times prior to acquiring the scan to ensure an even tear film. Even the presence of contact lenses can affect retinal nerve fiber layer thickness measurements.25 Dense media opacities will degrade image quality, although OCT is able to quite effectively penetrate most cataracts, asteroid hyalosis and vitritis. Mathematical models have been developed to improve the quality of these degraded images,26 however, the experienced examiner is usually still able to discern sufficient detail in most cases.
Central retinal thickness was compared between six commercially available OCT scanners in a study involving healthy eyes. A variation of between 0.45 and 3.33% was found. The discrepancies were explained by the slightly different segmentation algorithms employed by each device.27 In effect, this means that the line the software uses to determine the outer retinal boundary differs, so different OCT systems should not be used interchangeably.27,28
On the fast macular thickness map protocol with TD-OCT scanning,6 radial sampling scans of the macula are acquired and a macular topographical color-coded map of the macula is produced. The software interpolates adjacent thickness values in the interspersing macular areas that lie between the six radial scans. Clinicians should therefore be aware of small lesions suspected to lie within these areas, as they may not be picked up with this protocol. This is not an issue with the newer SD-OCT, which acquires a faster series of high-resolution images.11
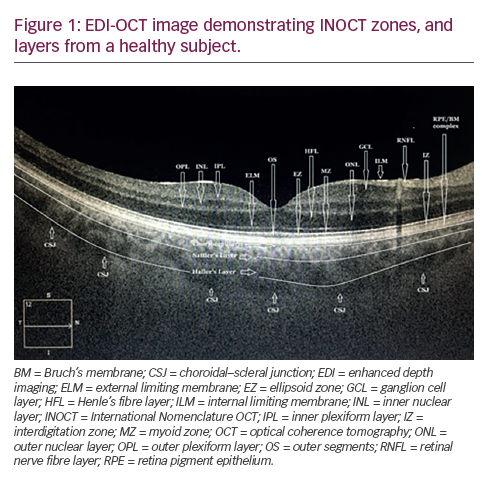
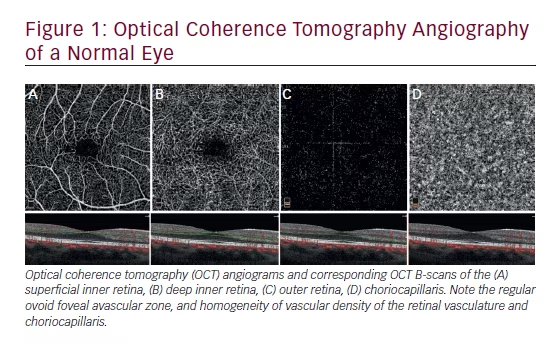
With higher-resolution scanning, a cross-sectional B-scan can reveal most layers within the neurosensory retina (see Figure 2).29 A few histologic studies have correlated the appearance of the OCT with histologic sections of human and animal retinas.30,31 Further studies have correlated the segmentation (bands) on an OCT with retinal layers by surgical sequential ablation of these layers.32,33 The outer red line (ORL) is frequently assumed to represent the RPE alone, but in actuality corresponds to the highly reflective chorio-retinal complex as a whole (see Figure 1). The predominant contribution to the ORL is by the Bruch’s membrane and inner choroid, with a smaller contribution by the RPE.33 Of particular interest is the band correlating to the junction of the inner and outer segment (IS/OS) of the photoreceptors (see Figure 2). This is better visualized as a ‘red line’ just inside the ORL on the higher-resolution SD-OCT. The IS/OS band is a high-reflectance signal at this junction resulting from the abrupt change in the refractive index stemming from the highly organized stacks of membranous disks in the photoreceptor outer segments.29 OCT changes in this area have been studied in a number of conditions, and visual acuity (VA) has been significantly correlated with OCT detection of the IS/OS junction in retinitis pigmentosa,34 macula-off retinal detachments,35 full-thickness macular holes,36–38 central serous chorioretinopathy,39 age-related macular degeneration (AMD)40 and macular edema associated with branch retinal vein occlusions.41,42 As IS/OS disruption (see Figure 3) reflects VA, the external limiting membrane (ELM) appears to show prognostic promise.35 In a series of consecutive retinal detachments (RDs), IS/OS disruption was observed in macula-off eyes. As predicted, post-operative VA was significantly correlated with IS/OS integrity. None of the eyes with pre-operative disruption of ELM and IS/OS regained post-operative IS/OS integrity. By contrast, seven of the 11 eyes that had intact ELM on OCT pre-operatively regained the IS/OS junction during follow-up.35
Vitreomacular Traction Syndrome
Vitreomacular traction syndrome (VMTS) is a condition in which the vitreous is separated from the peripheral fundus but remains adherent in the macular area. OCT is instrumental in diagnosing and monitoring progression, or in some cases confirming resolution. Prior to the availability of OCT, most cases were diagnosed with careful contact lens fundoscopy. More recently, some surgeons have proposed utilizing OCT as a pre-operative surgical planning tool to guide posterior hyaloid membrane peeling in VMTS by locating the area of greatest separation of the posterior hyaloid from the macula.43
A novel method of vitreoretinal interface imaging combines FF-OCT with simultaneous scanning laser ophthalmoscopy (SLO) to produce a coronal image of the retina, which provides a broader overview of vitreoretinal attachment.44 Second, as the SLO image is co-registered with the OCT, it is easier to orientate and localize pathology. A more anatomical perspective of the posterior hyaloid obtained in this manner will also allow the ophthalmologist to better assess the severity and concurrence of other vitreoretinal disorders, such as epiretinal membranes (ERMs), which frequently co-exist with VMTS.45
Age-related Macular Degeneration
Vitreomacular traction has been associated with neovascular AMD, however, Lee and colleagues46 reported that by comparing eyes with wet AMD versus contralateral normal eyes of the same patient, no statistically significant differences in vitreomacular adhesion were found with respect to choroidal neovascular membrane (CNVM) location or type.46 Nevertheless, OCT is invaluable in the initial assessment of AMD either when FFA is not available or as an adjunct to FFA. As a screening test, it is able to detect the presence and location of fluid or atrophy, thus reducing the need for unnecessary FFAs. With the increase in anti-vascular endothelial growth factor (anti-VEGF) treatments, OCT has become an indispensible tool in the screening and monitoring of CNV presence and progression or response to treatment, especially since it can demonstrate subtle disease activity prior to symptomatic or biomicroscopic changes.47 In the past, this was the sole remit of the resource-consuming and more invasive FFA, which is now mainly used for diagnostic confirmation and typing in most units in the UK. Furthermore, OCT provides objective documentation of progress compared with FFA, which has been reported to have comparatively poorer inter-observer agreement, especially following treatment.48–50 A study assessing the sensitivity of TD-OCT against SD-OCT in the detection of subretinal and intraretinal fluid as a proxy for CNV activity following intravitreal ranibizumab injection found that 3D SD-OCT had the highest detection rates, followed by SD-OCT linear scans and, lastly, TD-OCT.28 A good indicator of visual outcomes after anti-VEGF therapy is the preservation of the IS/OS junctions of the photoreceptors (see Figures 2 and 3).40 Sayanagi and colleagues used four different SD-OCT machines to evaluate the status of the IS/OS in 23 eyes of 22 AMD patients who had received a mean of 5.4 (SD=3.2) anti-VEGF injections over an average of 10.4 months (SD=8) prior to imaging. There was a significant difference in detection of the IS/OS between the AMD patients and the eight healthy eyes. Statistically, eyes with detected IS/OS had better best corrected VA (BCVA) on the day of imaging and significantly improved BCVA since the start of anti-VEGF treatment.40
Reduced reflectance from the IS/OS has also been demonstrated in areas of drusen. This phenomenon can be explained by either photoreceptor damage or elevatory disruption of the vertically orientated photoreceptors by the underlying drusen.51,52 Further studies with larger populations are required to determine the clinical significance of this finding, which may in the future provide clinicians with valuable prognostic information in the management of wet and dry AMD alike.
New SS-OCT technology, with its improved axial beam penetration, allows for improved structural delineation and qualitative evaluation of lesions located beneath the RPE.4 It has allowed clear demonstration of polypoidal choroidal vasculopathy (PCV) lesions in vivo, pinpointing its location between displaced RPE and the outer part of Bruch’s membrane,53 which is in agreement with histopathological studies.54 Identification and differentiation of such lesions from wet AMD is important, as treatment regimes differ, with PCV responding to photodynamic therapy.55,56
Epiretinal Membranes
ERMs are caused by glial proliferation on the surface of the macula and can cause varying degrees of vascular distortion, or even macular edema. OCT displays this membrane as a thin, smooth, hyper-reflective line along the surface of the retina (see Figure 4). Careful evaluation of the OCT can reveal the extent of ERM adherence to the underlying retina, the character of which correlates with the pathogenesis.57 Focal attachments of the highly reflective ERM to the less reflective inner retinal surface can sometimes be easier to discern than global ERM adherence, which can make the ERM indistinct from the ILM. Focal adhesions are more commonly seen in secondary ERMs, while global adhesion is more prevalent in idiopathic ERMs.57 ERMs are usually smooth, but can produce retinal folds, inner retinal irregularity and diffuse retinal thickening, which can be fully appreciated on 3D OCT rendering.58 3D scanning could also aid surgical planning as it reveals the manner and direction of ERM traction on the retina.58
Some studies have found a correlation between VA and retinal thickness in eyes with idiopathic ERMs;57 however, this has not been found in other studies.59,60 Higher-resolution OCTs have improved pre-operative ERM evaluation60 (see Figure 4) and have demonstrated that central foveal thickness (CFT) correlates with both pre-operative VA and IS/OS abnormalities.61,62 Suh et al. also found that patients with intact IS/OS had better BCVA post-operatively. By grading the status of the IS/OS (see Figures 2 and 3) as present, absent or abnormal, Mitamura and colleagues found that logMAR BCVA correlated with both pre-operative CFT and IS/OS grades. However, at three and six months post-operatively, BCVA correlated only with the status of the IS/OS and not CFT. The percentage of eyes with a normal IS/OS gradually increased post-operatively.
Macular Holes
Full-thickness macular holes (FTMHs) are easily confused clinically with lamellar and pseudoholes. Accurate diagnosis is essential as management differs greatly. In one study, almost two-thirds of cases (63%) found to have a lamellar macular hole (LMH) on OCT were initially incorrectly diagnosed as either a FTMH, ERM or macular pseudohole on biomicroscopy.63 OCT grading of FTMH status can guide the urgency of treatment.
LMHs are now primarily an OCT-based diagnosis as biomicroscopy provides insufficient resolution to detect residual outer retina, foveal photoreceptors, or intraretinal split,63 the presence of which explains the relatively preserved vision in this group of patients. The almost constant finding of an ERM in the presence of LMH63,64 could shed light on its evolution. Gass,65 along with a few other authors,66–68 proposed that LMHs were a result of an abortive process of a FTMH.
Gass also described the formation of LMH following chronic cystoid macular edema (CME).65 An observational case series reporting four patients followed up with OCT confirmed that the occurrence of an LMH following refractory (diabetic) CMO is most likely due to the rupture of the inner wall of the thinned, cystic spaces. None of the four patients experienced any change in VA despite remarkable anatomical reduction in foveal thickness (from a mean of 509 to 166μm). In all cases, high-resolution OCT revealed that the outer retinal layer remained, with at least intermittently recognisable IS/OS.69
FTMHs have been extensively investigated with the use of OCT; the clinical features associated with improved visual prognosis are well-known and include better VA on presentation, duration of symptoms and hole diameter.70–72 Post-operatively, a few studies have revealed that the presence of an intact IS/OS is an important indicator of visual recovery, with the number of patients with detectable IS/OS increasing over time following FTMH repair.37 The post-operative status of the IS/OS line on OCT correlates well with VA,36–38 with a significant correlation between the post-operative presence of the IS/OS and the pre-operative duration of symptoms.
Post-operative IS/OS line ‘healing’ appears to occur at differing rates,36,37 and it is still unknown how some patients continually show gradual visual recovery years after FTMH repair. There are probably numerous other prognostic determinants yet to be discovered as, histologically, photoreceptors are reported to remain intact in the fluid cuff of a FTMH73 and, if viable, could possibly regenerate inner and outer segments.74 These findings infer photoreceptor recovery and provide signs of anatomical success on an almost histological scale.
Summary and Future Directions
OCT has proved to be a powerful and versatile medical tool. It provides a non-contact method of objectively quantifying many conditions affecting the retina and has revolutionized the manner in which certain vitreomacular conditions are diagnosed and managed. It has shown promise as a tool for surgical planning and provides fine anatomical detail of the retina, which can aid post-operative prognostication. Newer-generation OCTs have already been widely used for research purposes and we eagerly await their commercial availability.