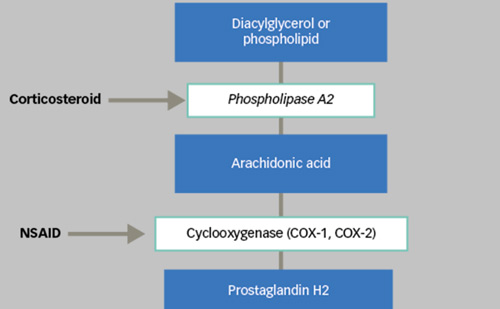
Diabetic macular oedema (DME) is a severe ocular complication of diabetic retinopathy (DR).1 Despite the fact that significant advances are being made in early diagnosis and treatment of patients, numbers of patients likely to develop vision loss or blindness due to DR and DME are expected to rise in parallel to the incidence of diabetes. Estimates have projected that, by 2050, there will be in excess of 50 million diabetics in the US, half of whom are projected to develop DR.2–4
The treatment of DME has evolved dramatically over the past decade. While laser therapy presented the standard of care for many years,5 recent times have seen the introduction of several new therapeutic approaches. All of these therapies have in common that they are applied intravitreally. For many of these treatments, initial data from pivotal clinical trial programmes are now being supplemented by clinical practice data. This article aims to identify and review the available data to determine how newer therapies perform in real-world practice compared with the initial clinical trial setting.
A search of the US National Library of Medicine, National Institutes of Health (http://www.ncbi.nih.gov/pubmed) was performed for the time period between June 2009 and March 2016, based on the following search terms: “diabetic macular edema” and “outcomes” and the respective active compound (“aflibercept”, ”bevacizumab”, “dexamethasone”, “fluocinolone” or “ranibizumab”). Publications of individual study reports as well as review articles were included to generate a balanced review of the current state of play.
The two fundamental therapeutic groups now available for the indication of DME are vascular endothelial growth factor (VEGF) inhibitors and corticosteroids.
Intravitreal anti-vascular endothelial growth factor therapies
Licensed VEGF inhibitors include ranibizumab and aflibercept. Additionally, bevacizumab is used as an off-label option in clinical practice.
Ranibizumab and bevacizumab
Key information on study set up as well as study results for ranibizumab and bevacizumab are listed in Table 1.
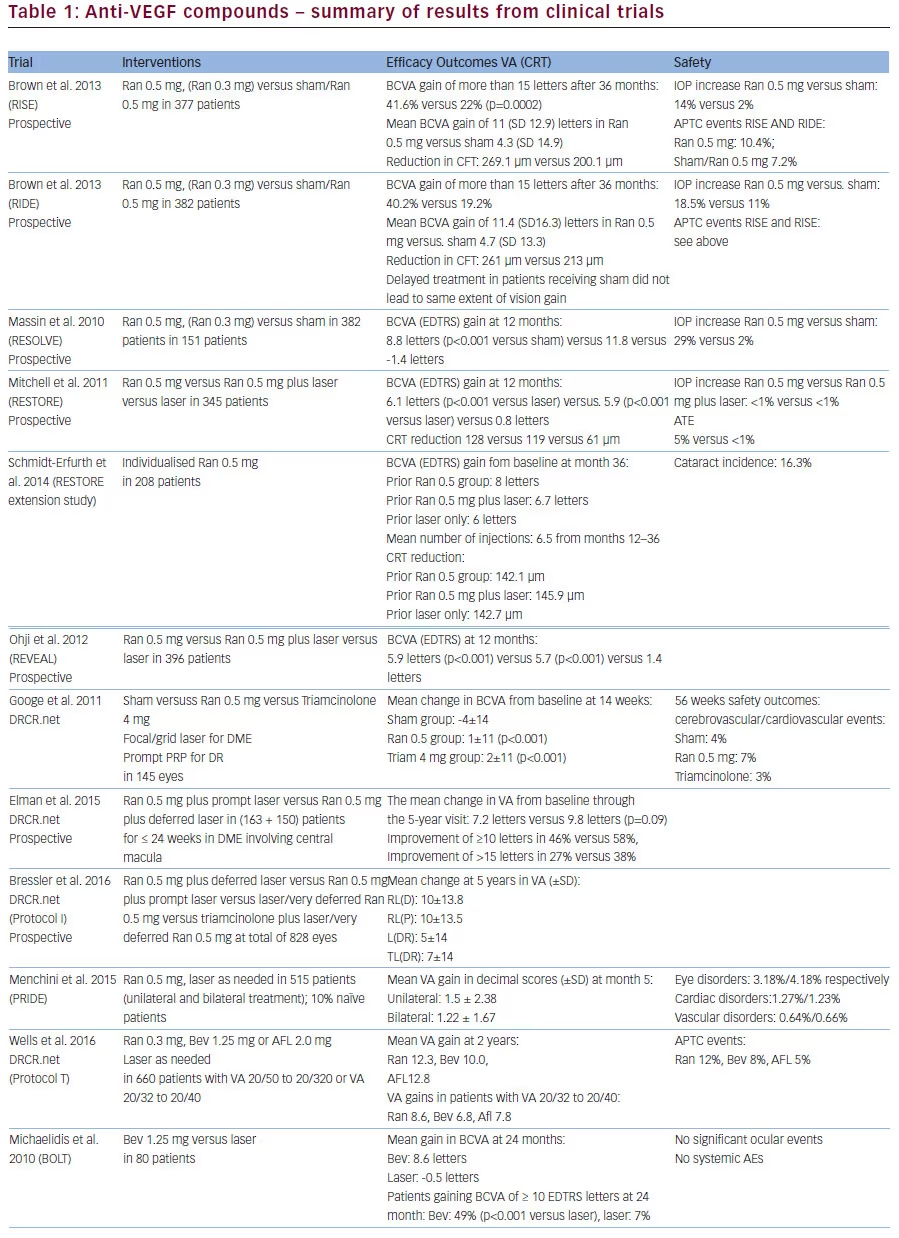
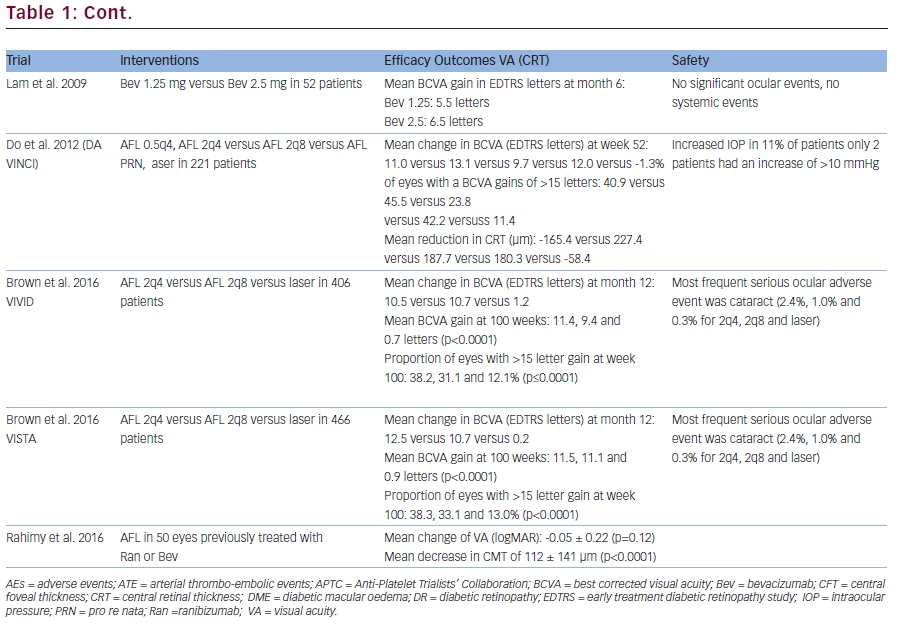
Ranibizumab was initially investigated in DME patients in a phase I/ II open-label evaluation (READ-2 study) as well as a phase II trial (RESOLVE).6 Several phase III studies in DME followed. The RISE and RIDE studies were two parallel studies which ran for a time period of 36 months. Gains in visual acuity (VA) and improvement in retinal anatomy was sustained over three years. The proportions of patients who gained at least 15 letters from baseline in the sham/0.5 mg, 0.3 mg and 0.5 mg ranibizumab groups were 19.2%, 36.8% and 40.2%, respectively, in RIDE and 22.0%, 51.2% and 41.6%, respectively, in RISE.7
REVEAL and RESTORE compared ranibizumab alone to laser alone or to combination treatment. Notably, the addition of laser did not confer additional benefit.8 The RESTORE three-year outcomes study demonstrated that ranibizumab was effective in improving and maintaining best corrected VA (BCVA), central retinal thickness (CRT) outcomes with a progressively declining number of injections per year (mean of <3 injections).9
Real-world clinical practice data are also available from open-label extensions of the original clinical trials, as well as from post-licensing studies, giving physicians the opportunity to inject the product on an as-needed basis (pro re nata [PRN]). Data from the extension of RESTORE demonstrated that improvements in BCVA and CRT were also maintained at year three, with patients requiring fewer injections over this time.7
The Diabetic Retinopathy Clinical Research Network (DRCR.net) study demonstrated that the addition of one intravitreal triamcinolone or two ranibizumab injections in eyes receiving focal/grid laser for DME and panretinal photocoagulation (PRP) is associated with better VA and decreased macular oedema by 14 weeks.10 Another DRCR.net study, performed in patients with decreased VA and DME involving the fovea over five years, reported that patients treated with ranibizumab plus prompt laser or ranibizumab plus deferred laser maintained visual gains from year 1 to 5, requiring less treatment after year 3. Notably, patients from the latter group required more ranibizumab injections.11 A similar study included two additional patient groups receiving laser-only or triamcinolone plus deferred ranibizumab. Authors concluded that eyes receiving initial ranibizumab treatment are likely to have better long-term vision improvements.12 An Italian study provided additional evidence. Use of ranibizumab was well tolerated and improved or maintained VA in 40.2% to 68.8% of patients, with no difference between unilateral or bilateral cases.13
A recent DRCR.net study compared the effectiveness of aflibercept, ranibizumab or bevacizumab for DME over two years (PROTOCOL T). The median number of injections was 15, 16 and 15, respectively over two years, with lesser injections in year 2. Overall, all three treatments improved VA from baseline. While VA outcomes were similar for eyes with better baseline VA, aflibercept had better outcomes compared with bevacizumab over two years, as well as to ranibizumab over the first year in eyes with worse baseline VA. Higher Anti-Platelet Trialists’ Collaboration (APTC) event rates with ranibizumab warrant a continued evaluation in future trials.14
Bevacizumab is not licensed for the indication of DME, however, it is used in current medical practice. Several studies have demonstrated the efficacy and safety of bevacizumab in DME. The BOLT study compared use of bevacizumab with laser treatment (see Table 1).15 Faghihi et al. reported a similar injection rate and efficacy for bevacizumab 1.25 mg and bevacizumab 1.25 mg plus a single laser treatment over a period of six months.16 In a six-month study in patients with diffuse DME, Lam et al. demonstrated a similar efficacy of two different doses of bevacizumab (1.25 mg and 2.5 mg).17 Additional trials have investigated a combination of bevacizumab 1.25 mg and triamcinolone 2 mg. Ahmadieh et al. found that the BCVA for the combination was not significantly better than bevacizumab alone.18 Soheilian et al. compared bevacizumab 1.25 mg plus triamcinolone 2 mg versus bevacizumab alone and laser alone. Results at 24 months did not favour any of the groups. Lim et al. demonstrated that treatment with a combination of bevacizumab plus triamcinolone, bevacizumab alone or triamcinolone alone did not yield statistically differences between groups in terms of BCVA and CMT. The ATEMD study also found comparable efficacy in the bevacizumab 1.25 mg plus triamcinolone 4 mg group and each monotherapy alone. The adverse event (AE) profile is comparable to ranibizumab. Studies are not powered to detect small differences in rates of infrequent events. Overall, bevacizumab was well tolerated.8
Aflibercept
Key information on study set up as well as study results for aflibercept are listed in Table 1.
A multicentre phase II study evaluated the efficacy and safety of different aflibercept concentrations compared with laser in DME patients. Significant gains in BCVA from baseline achieved at week 24 were maintained or improved at week 52 in all aflibercept groups.19
The efficacy and safety of aflibercept 2 mg applied monthly (2q4) or every two months (2q8) was compared with laser treatment in two phase III studies. The authors concluded that, in both studies (VISTA and VIVID), the 52-week anatomical and visual superiority of aflibercept injections over laser treatment were significant and similar at week 52 and through week 100. At week 100, the mean BCVA gain was 11.4, 9.4 and 0.7 letters in VIVID and 11.5, 11.1 and 0.9 letters in VISTA for aflibercept 2q4, aflibercept 2q8 and laser, respectively.20 Real-world outcomes in 50 eyes with persistent DME previously treated with ranibizumab or bevacizumab and then switched to aflibercept were recently reported by Rahimy and colleagues. Conversion to aflibercept resulted in significant anatomic improvements, but only trends towards improved VA and reduction in IOP were observed.21
Intravitreal corticosteroid delivery systems
Fluocinolone
Key information on study set up as well as study results for fluocinolone are listed in Table 2.
The long-term efficacy and safety of the fluocinolone acetonide (FAc) non-degradable implant was assessed in patients with persistent DME in spite of one or more laser treatments. Subjects were eligible for laser rescue treatment six weeks after injection. Almost all phakic patients in the FAc implant groups developed cataract. However, their actual visual benefit, as determined after removal of the cataract, was similar to pseudophakic patients in the study. Intraocular pressure (IOP) elevations considered an AE were reported in approximately 35% of patients receiving FAc 0.2 μg/day.22 In the active group, 18.7% of patients received two and 6.3% three or more implants. Laser treatment was administered in 40% of patients in the active group. Also 13.4% of patients from this group received additional off protocol treatments such as intravitreal triamcinolone or anti-VEGF compounds. It was concluded that the FAc 0.2 μg/d implant provided substantial benefit for up to three years and thus would provide an option for patients who do not respond to other therapies.23 A recent study produced data in six pseudophakic patients with bilateral, chronic DME previously treated with laser and anti-VEGF therapy. The FAc implant in both eyes was a feasible, effective second- or third-line choice for patients with severe bilateral DME, without notable increases in IOP.24
Dexamethasone
Key information on study set up as well as study results for dexamethasone (DEX) are listed in Table 2. The DEX implant contains 700 μg DEX in an inactive biodegradable polymer matrix. The latter ultimately degrades to water and carbon dioxide and DEX is released over six months. It is indicated patients who have not respond sufficiently to non-corticosteroid treatment, pseudophakic patients and patients deemed to be unsuitable for non-corticosteroid treatment. Efficacy and safety of DEX biodegradable implant is reported in several phase II and III trials. PLACID was a 52-week, masked, multicentre, randomised, sham-controlled phase II trial.25 CHAMPLAIN was a 26- week, open-label trial to assess the safety and efficacy of DEX implant in patients with DME who had had pars plana vitrectomy. Patients with DME and a history of vitrectomy showed a benefit of treatment up to six months after implantation in terms of reduction of macular thickness and increase in BCVA.26
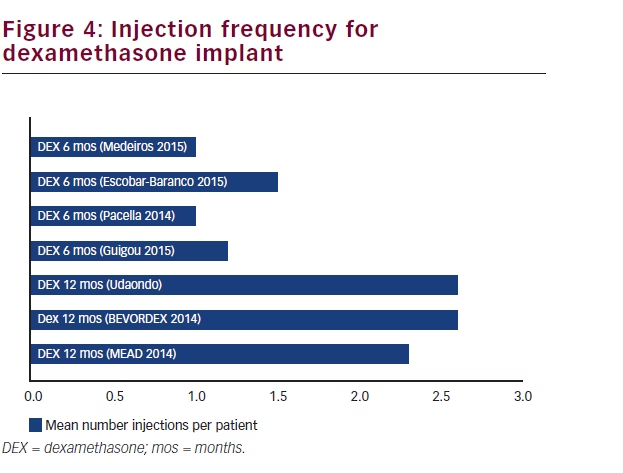
The MEAD study, a phase three trial, included a total of 1,048 patients. After the first DEX implant treatment, an additional implant could be administered every six months if the thickness of the oedema increased or persistent oedema was present. Patients could receive up to seven treatments during the trial. A very rapid initial response was demonstrated in BCVA values, maintaining statistical significance up to approximately 15 months. This was followed by a drop in BCVA, coinciding with the development of cataract. A subgroup analysis of the patients that were pseudophakic at baseline (approx. 25%) showed consistency in BCVA improvement throughout the three-year study period compared to sham. VA decreased between the time of reporting cataract AE and cataract surgery but improved to pre-cataract levels after surgery.27 A subgroup analysis of patients previously treated for DME (with laser, anti-VEGF therapies, intravitreal triamcinolone acetonide (IVTA) or a combination of these therapies) showed that the DEX implant significantly improved visual and anatomic outcomes.28 The BEVORDEX trial compared efficacy and safety of the DEX implant with bevacizumab. At month 12 the percentage of patients reported with ≥10 logMAR letters were 40 in the DEX implant and 41 in the bevacizumab group, while CMT was reduced by 187 μm and 122 μm, respectively.29 The VA improvement in both groups was maintained at 24 months, while the CMT reduction in the bevacizumab group caught up to be comparable with the DEX implant group. The injection frequency difference for both groups was less pronounced in year 2, still favouring the DEX implant.30 A 12-month single-masked, randomised controlled study comparing the efficacy of bevacizumab 1.25 mg with a combination of bevacizumab 1.25 mg and the DEX implant in eyes with persistent ME came to the conclusion that the combination significantly improves VA as well as the morphology of refractory DME, although VA gains were not superior to continued bevacizumab therapy.31
Recently, data from use of the DEX implant in clinical practice has also become available. A prospective study compared the efficacy of a single administration of DEX implant with monthly intravitreal injections
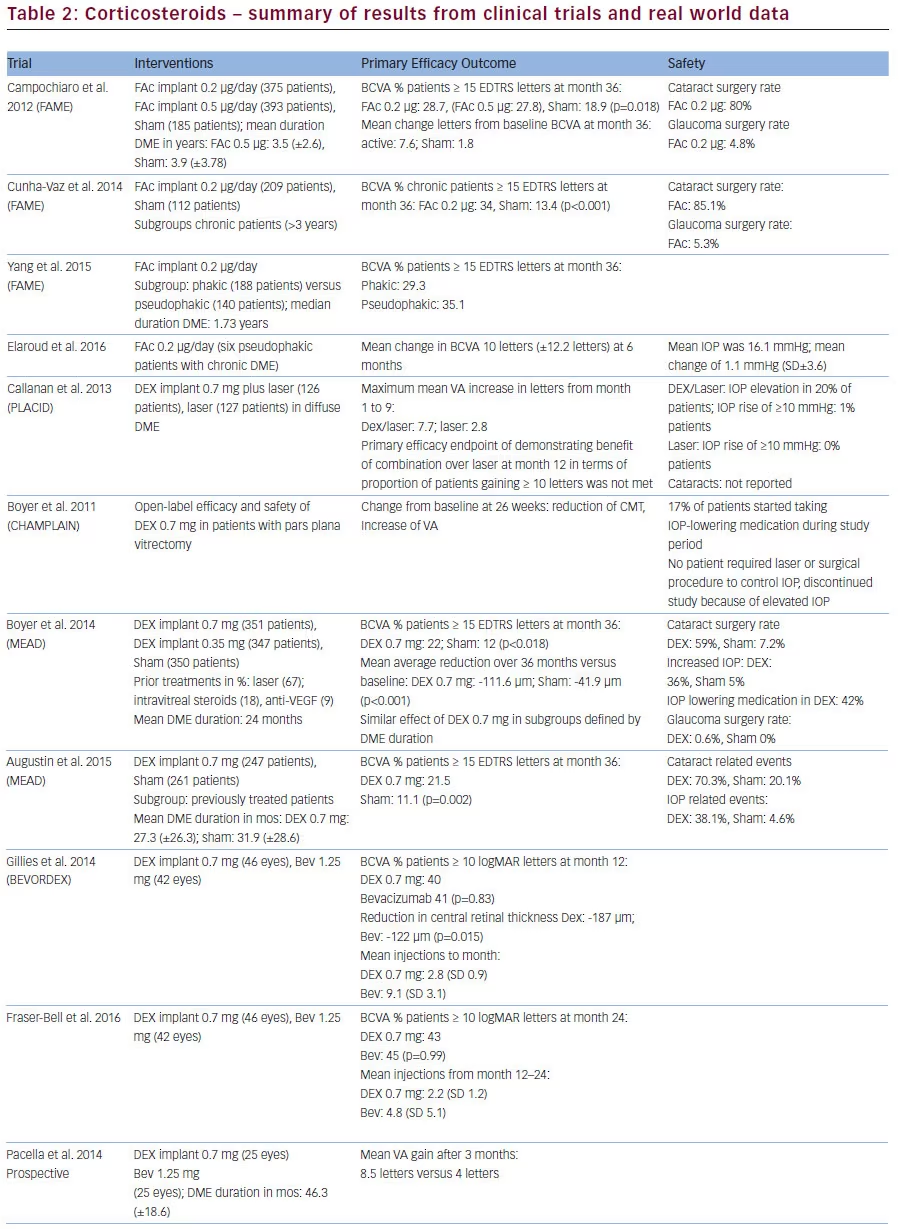
of ranibizumab. After three months, DEX implant showed a better efficacy (mean VA gain of 8.5 versus 4 letters). This significance was lost after six months (overall VA gain of 2.5 versus 2 letters).32 A prospective study comparing bevacizumab administered as monotherapy with the DEX implant in the treatment of persistent DME reported that both groups had similar VA gains, while patients in the DEX implant group achieved a significantly greater reduction of central subfield thickness (CST) compared with the bevacizumab group.33
The authors of a retrospective study reported DEX implant to be safe and effective over a mean treatment period of 9.8 months. The mean improvement in VA at six months was 7.6 letters. The mean re-injection interval was 5.4 months with on average.34 A prospective study on refractory and naïve DME patients treated with DEX implant and laser as needed reported a significant improvement in VA from baseline (p<0.001) in both groups, especially in the naïve group.35
A retrospective study analysed data of refractory DME patients receiving a single DEX implant. The authors concluded that the DEX implant is effective in difficult-to-treat patients with long-standing oedema.36A prospective study in patients unsuccessfully pre-treated with bevacizumab demonstrated that DEX implant significantly increased mean BCVA (logMAR) at one and three months. IOP increases were well controlled with topical anti-glaucoma monotherapy.37 Similar efficacy was reported by Lazic et al. from a prospective dataset in the same patient type.38
A retrospective comparison over six months in vitrectomised versus non-vitrectomised patients with refractory DME reported similar BCVA improvements and reduction of central foveal thickness (CFT) in both groups.39
Discussion
Based on the existing data, VEGF inhibitors as well as corticosteroids have demonstrated efficacy and safety in DME. Furthermore, the use of intravitreal drug delivery in combination with mechanical approaches such as laser therapy show promise for improving visual outcomes and decreasing the need for re-treatment.40
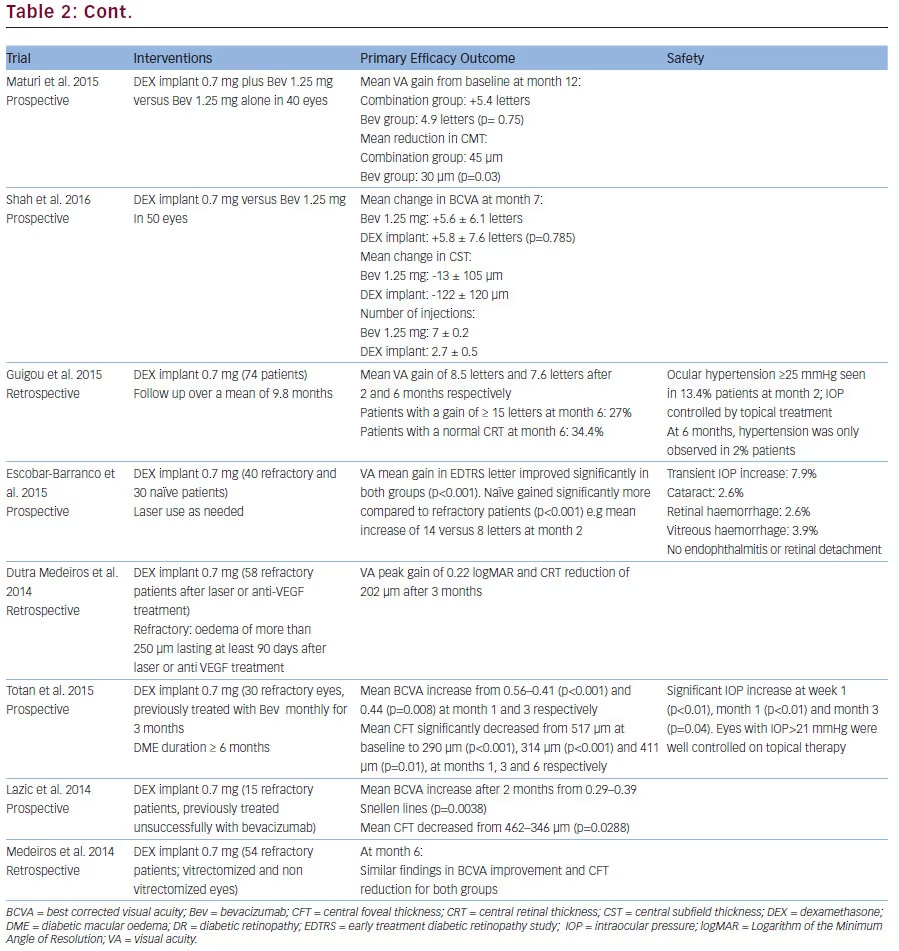
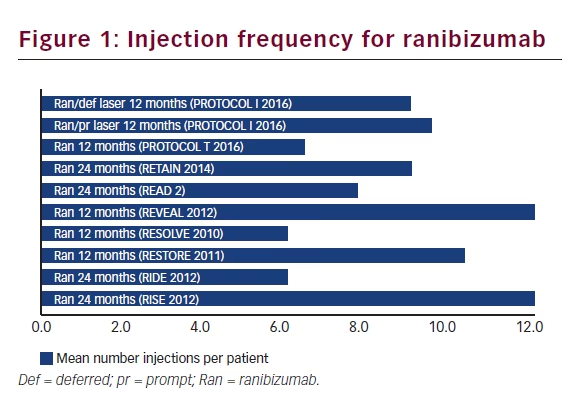
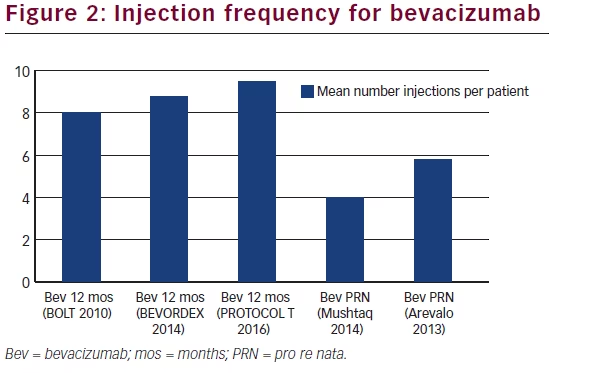
VEGF inhibitors are easy to apply and used as treatment of first choice. Based on the recent PROTOCOL T findings, aflibercept seems to achieve better outcomes in eyes with worse baseline VA. An obvious drawback is the high frequency of injection. However, some longer-term data suggest that this frequency is reduced overtime, while still maintaining VA gains previously achieved. Overall, real-world outcomes data do not suggest a lower treatment frequency, if similar efficacy results want to be obtained (Figures 1, 2 and 3).
However, frequent injections increase the number of treatment-related AEs. Furthermore, they put a great burden on the patient suffering from this chronic disease and decrease patient compliance. Delivery systems can lead to a reduction of these disadvantages.41
Available delivery systems are so far loaded only with corticosteroids. The non-biodegradable delivery system releasing FAc is only available as a last resort option in very restricted patient populations (chronic patients who are resistant to other treatments), with little real-world data currently available. Reasons for this include the long-term nature of the therapy, a potential need to remove the empty device and concerns about lesser control of emerging side effects.
In contrast, DEX implant is indicated in all types of DME for pseudophakic patients or in patients, where alternative treatments are not effective or are inappropriate. The biodegradable implant is injected and does not need to be removed. Compared with anti-VEGF injections, it needs to be administered less often. A fair amount of shorter-term clinical practice data has started to emerge, reporting that treatment intervals remain the same or are slightly shorter or longer. Results suggest a similar to better efficacy compared with the phase III data clinical trial data, and similar efficacy to anti-VEGF. Additional findings suggest that improvement of macular morphology with the DEX implant could be more pronounced. Furthermore,
data on combining the DEX implant with anti-VEGF compounds is also emerging.
Major limitations to comparing pivotal trials with clinical practice data are the differing, mostly shorter study periods, different reporting of efficacy endpoints such as VA, smaller patient numbers and less clearly defined patient populations, especially in respect to duration of DME. Based on the pivotal trial findings, the current assumptions should be that delayed treatment of patients results in lower overall vision gains.
Apart from injection-related ocular AEs, side-effect profiles between VEGF inhibitors and corticosteroids differ. Main concerns for the latter group revolve around the significant progression of cataract and an increase of IOP. Even though rise of IOP has also been reported in anti- VEGF compounds, this is to a much lesser extent.
A growing concern with intravitreal ranibizumab use in other retinal diseases (such as age-related macular degeneration) is the possible increased risk of occurrence and increased growth of geographic atrophy. Intravitreally applied corticosteroids at low doses have not been observed to exert relevant retinal toxicity.36
Systemic AEs in general are not associated with corticosteroids, but rather with VEGF inhibitors. There are no consistent reports so far. Nonocular serious AEs (myocardial infarction, stroke and death) have been reported for ranibizumab in a slightly higher incidence in the treatment than the sham group in studies for DME and other retinal diseases (e.g., 2.5% versus 0.8% in the MARINA study). Cerebrovascular accidents occurred in 4% of ranibizumab 0.5 mg patients in RISE and 2.4% of ranibizumab 0.5 mg patients in RIDE. Reported rates of AEs differed amongst studies and groups. However, studies were not powered to detect small differences in rates of infrequent events.7 The authors of a recent study comparing outcomes of all VEGF inhibitors in DME propose that due to higher APTC event rates found in ranibizumab, that such events should also be monitored in future.12 A recent systematic review and meta-analysis of prolonged monthly anti-VEGF use in DME, the assessment of the highest-level exposure group (patients receiving monthly treatment over two years) revealed a possibly increased risk for death and potentially for cerebrovascular accidents. Therefore, the authors concluded that total exposure to anti-VEGF drugs in patients at high risk for vascular diseases should be taken into consideration.42
Conclusions
Management of DME is complex and often multiple treatment approaches are needed. Emerging data on the newly licensed therapies show VEGF inhibitors and corticosteroids to be equally effective. Corticosteroids appear to be a less treatment-intensive alternative compared with anti-VEGF treatment in DME. While corticosteroids have a widely known safety profile and are already used in well-defined patient populations, recent safety findings on anti-VEGF treatments suggest that there is a need to select patients more carefully.
Currently available datasets scarcely allow direct comparison due to different reporting formats for efficacy and safety parameters, as well as diverging reporting periods. Future real-world data will need to be generated over longer periods of time to inform about longterm treatment efficacy, ideally using uniform reporting parameters to facilitate better comparison of data sets for the same treatment, as well as between treatments.
Future-generation devices should allow even longer release durations and increased target specificity to prolong their action, minimise side effects and achieve patient compliance.43