Greater corneal biomechanical stability has been demonstrated following photorefractive keratectomy (PRK) compared with traditional microkeratome laser-assisted in situ keratomileusis (LASIK).1,2 Thin LASIK flaps exhibit biomechanical properties similar to PRK but with the advantage of no haze formation or pain.2 These findings suggest that thin flaps created with consistent thickness across the cornea are the ideal choice for optimizing refractive and biomechanical outcomes. This article is an overview of our published studies using anterior segment optical coherence tomography (OCT) to analyze thin flaps created with the IntraLase® femtosecond (FS) laser (IntraLase, Inc. Irvine, California).3
Methods
In this prospective study, 25 eyes with myopia with or without astigmatism had excimer laser ablation following thin flap creation by the 60kHz IntraLase FS. Standardized flap parameters were programmed for each procedure with a superior hinged flap. Desired flap thickness was 110μm. Previous experience with our 60kHz Intralase FS laser demonstrated that we need to program the laser to a thickness of 100μm to obtain a thickness of 110μm. The other laser parameters were: 8.5mm diameter, hinge angle of 50º, side-cut angle of 75º, raster pattern energy of 1.30μJ, pulse separation of 8×8μm, and side-cut energy of 2.00μJ with the pocket enabled.
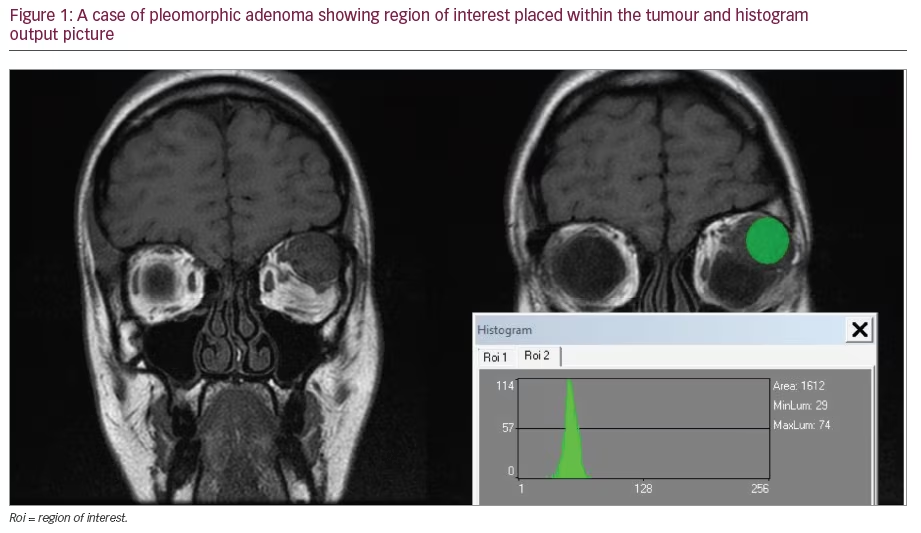
The eyes were evaluated during a one-month post-operative examination using the Visante™ anterior segment OCT (Carl Zeiss Meditec, Inc.) to image each flap. A skilled technician performed all the high-resolution corneal scans with 512 A-scans per line sampling and 0.25 seconds per line acquisition time (2,048 scans per second). The Visante can display a cross-section of an image at any specified meridian. We chose to display the 45°, 90°, 135°, and 180° meridians for each flap. Visante OCT requires manual (semi-automated) measurement. The Visante flap tool is a computer-controlled cursor that is placed on the corneal image at the desired location and that automatically measures corneal thickness. Within this total corneal thickness measurement, the cursor is manually placed on the visualized flap interface. Using the software’s flap tool, the flap interface was visualized and marked by the examiner to measure flap thickness and residual stromal bed thickness. The flap thickness was measured at four points for each of these cross-sections by one examiner who was masked to the attempted flap depth. Two points were ±3–4mm from center, and two were ±1–2mm from center. Thus, each flap’s thickness was measured at a total of 16 points.
Statistical Analysis
The 16 flap thickness measurements were entered into 16 separate columns in an Excel spreadsheet. The data were then analyzed with three different statistical methods: by pooling all data, by looking at each column separately, and by multivariate statistical analysis on the 16 columns simultaneously.
Various statistics were calculated for each method, such as averages, standard deviations (SD), 95% confidence intervals (CI), and p values. The p values were from the one-sample T test, where the data are compared with a hypothesized average value. For the multivariate statistical analysis, the corresponding test is Hotelling’s one-sample T-squared test. A p value ≤0.05 was considered statistically significant.studies suggest that the speed of the FS laser is important and does correlate with flap thickness predictability.
Results
All 16 measurements for each flap were analyzed. The flaps were uniform (planar) in appearance with a mean thickness of 112±5μm (range: 87 118μm). Multivariate statistical analysis compared each data point with the mean 112μm flap thickness; the p values were not significant (p>0.05). Therefore, the average flap is either uniform (planar) at 112μm or varies insignificantly from the average of 112μm. The 16 measurements per flap were analyzed for each patient individually and the average standard deviation within the individual flaps was 4μm (range: 1–8μm). This also demonstrates that patients had flaps that were uniform (planar).
Discussion
The high-speed non-contact Visante OCT has a high axial resolution to acquire cross-sectional imaging of the cornea and anterior segment.4–8 The ease of use, ability to image a wide area of the cornea, and capacity to image the flap interface and make multiple direct measurements of flap thickness in four meridians are all useful for analyzing flap dimensions. In addition, direct measurements of residual stromal thickness obtained with the OCT are helpful when considering LASIK enhancement procedures.
A US Food and Drug Administration (FDA) study demonstrated that the Visante has high sensitivity and specificity in detecting the LASIK flap from the first day post-LASIK to six months post-LASIK.9 The central corneal thickness measurements of the Visante were more precise than measurements made with ultrasound pachymetry and Orbscan®. The study also found that the Visante was capable of making repeatable and reproducible measurements in both pre- and post-LASIK populations. Li et al. reported that repeatability of OCT pachymetry measurements was 2μm.10 They also found that high-speed OCT is equivalent to ultrasound for central corneal thickness measurements before and after LASIK.
Automated computer algorithms have been developed for an OCT prototype similar to the Visante OCT for identification of the anterior and posterior corneal boundaries and flap interface thickness.11 Comparisons with ultrasound measurements have validated the accuracy of the algorithms for corneal and flap thickness. The Visante OCT requires the examiner to make manual (semi-automated) flap measurements, which may be more subjective than an automated measurement. Reported repeatability SD of the Visante OCT in measuring LASIK flap thickness at the +1.00mm location was 8.5μm and at the -1.00mm location was 8.7μm.12 The SD for combined flap and stroma thicknesses at these two locations were 6.3 and 6.5μm, respectively. Despite the demonstrated accuracy of the Visante OCT, there are limitations to all techniques that measure corneal thickness, including the Vistante OCT. This study demonstrates that the 60kHz IntraLase FS laser produces uniform (planar) flaps that are both highly predictable and reproducible (see Figure 1). The level of reproducibility in this study was a mean thickness of 112±5μm when a flap thickness of 110μm was attempted.
Increasing Femtosecond Speed Improves Accuracy
The faster 60kHz laser appears to have improved precision when comparing our results with a previous report concerning contact ultrasonic pachymetry of a 10kHz laser, which had a standard deviation of 12μm.13 Greater speed has allowed us to decrease our pulse energy. Less energy results in a smaller photodisruption bubble diameter. A smaller bubble diameter adds more precision in the dissection plane and therefore greater accuracy in the depth of the cut.
A comparative study of stromal bed quality with scanning electron microscopy by Sarayba et al.14 evaluated the Hansatome 15 and 30kHz IntraLase lasers. They reported no difference between Hansatome and a 15kHz laser; however, a significant improvement in stromal bed quality was observed with the faster 30kHz laser compared with the Hansatome and a 15kHz laser. Another comparative study with confocal microscopy evaluating flap creation by the 15 and 30kHz IntraLase lasers found the faster 30kHz laser had better flap thickness reproducibility with a smaller SD than the slower 15kHz laser.15 Binder16 reported SDs of 12.4, 14.3, and 15.2μm for flap thicknesses of 90, 100, and 110μm, respectively, with a 10kHz IntraLase laser. The SD was better with the faster 15kHzlaser: 10.8, 11.8, and 10.2μm, respectively, at the same flap thicknesses. These studies suggest that the speed of the FS laser is important and does correlate with flap thickness predictability. The importance of corneal biomechanics in refractive surgery outcomes has become apparent following recent research.17,18 The corneal stroma consists of lamellae and organized collagen fibers, which run from limbus to limbus. Traditional LASIK with mechanical microkeratomes creates a flap approximately 160μm thick that severs a significant number of collagen fibers compared with PRK. The loss of lamellar integrity following LASIK results in compromised corneal biomechanical integrity due to minimal biomechanical loading distributed throughout the flap. Hence, there is no contribution from the flap to the biomechanical stability of the cornea.1
Corneal Biomechanics
Cohesive tensile strength studies demonstrate that Bowman’s layer is the strongest structural component of the cornea followed by the anterior third of the corneal stroma.1,19 In fact, the peripheral anterior third of the corneal stroma is stronger than the paracentral and central anterior third of the corneal stroma.18 These findings are supported by morphological studies that demonstrate more collagen lamellar interweaving and collagen lamellae orientations that were transverse to the surface of the cornea in the anterior cornea.20–22 These studies suggest that a thin uniform flap would leave more of the strong anterior stroma untouched, which should provide greater corneal biomechanical strength than the thicker traditional LASIK flaps that sever more of these strong anterior fibers.
The biomechanical considerations described previously suggest less stability for traditional microkeratome LASIK compared with PRK. In vitro research with electronic speckle pattern interferometry and shearing interferometry by Marshall2 has demonstrated greater corneal biomechanical stability with thin flaps created in the compact anterior corneal stroma just beneath Bowman’s layer compared with thicker flaps. Based on these findings, we can hypothesise that a cornea with a thin uniform flap would have biomechanical stability more similar to that of PRK than traditional LASIK, but with the added benefit of no pain or corneal haze that occurs when utilising traditional LASIK. In addition, flaps made deep to the level of traditional LASIK in the weaker posterior cornea—where the lamellae lie more parallel and are less compact— create weaker corneal biomechanics.
The IntraLase FS laser also allows precise control over the diameter of the flap with SDs of 0.12–0.26mm.13 In this study, we programed a desired flap diameter of 8.5mm. This is smaller than flaps created with mechanical microkeratomes, which are typically greater than 9.0mm. These small-diameter flaps cut less of the strong peripheral corneal fibers, and may further add to biomechanical strength compared with larger flaps.
Mechanical microkeratomes can produce thin flaps approximately 100μm deep; however, most create meniscus-shaped flaps that are thicker in the periphery than in the center (see Figure 2), which results in an increase in the number of lamellae cut, thus further weakening the biomechanical properties of the cornea. Using very-high-frequency ultrasound, Alio and colleagues have recently demonstrated that the Carriazo-Pendular microkeratome (SCHWIND eye-tech-solutions GmbH & Co., Kleinostheim) creates a near-planar flap (JL Alio, written communication March 2, 2007). Analysis of flap dimensions in this study demonstrates IntraLase FS laser flaps have uniform thickness with a planar shape. The high predictability and reproducibility of the flaps created in this study make using the IntraLase FS laser an excellent technique for the creation of thin uniform flaps. The FS-laser technique is likely safer than using microkeratomes to create thin flaps due to the increased risk of flap button-holes when thin flaps are attempted with mechanical microkeratomes. However, if a thin planar flap can be created with a microkeratome, this should result in improved biomechanics too.IntraLase Laser Advantages
Advantages of the IntraLase laser compared with mechanical microkeratomes in flap creation have been demonstrated in published studies. Kezirian and Stonecipher reported fewer complications, better flap thickness predictability, and less surgically induced astigmatism in IntraLase eyes compared with microkeratome (Hansatome and Moria) eyes.23 A prospective, randomized, contralateral eye study comparing the IntraLase laser and the Hansatome by Durrie found significantly better uncorrected visual acuity outcomes in the early post-operative period and less astigmatism in IntraLase eyes.24 Tran et al. conducted a prospective, randomized, contralateral study up>to evaluate aberrations induced following LASIK flap creation only (no excimer ablation) with the IntraLase laser and the Hansatome.25 Microkeratome eyes had a significant increase in higher-order aberrations while the IntraLase eyes did not. Montés-Micó et al. compared contrast sensitivity following myopic LASIK with the IntraLase laser and microkeratome (Carriazo- Barraquer).26 IntraLase eyes demonstrated better contrast sensitivity at high spatial frequencies compared with microkeratome eyes under both photopic and mesopic conditions.
Conclusions
Thin uniform flaps appear to benefit from the strengths of both LASIK (no haze or pain) and PRK (biomechanical stability) procedures. Durrie and Slade have suggested that the name ‘sub-Bowman’s keratomileusis’ (SBK) be used for excimer laser procedures performed with a uniform thin flap that is custom-designed for the individual patient.27–29 This study demonstrates by anterior segment OCT that the IntraLase FS laser creates thin planar (uniform) flaps with high predictability and reproducibility.