In the past decade reports from numerous clinical trials have announced the arrival of a new era in pharmacotherapy for retinal diseases.1 This promises to be transformational in the fight against avoidable blindness where neovascular age-related macular degeneration (AMD) and diabetic retinopathy (DR) are recognized in a recent systematic review as leading causes, worldwide, of moderate and severe vision impairment.2 Intravitreal injection of anti-vascular endothelial growth factor (VEGF) therapies, such as ranibizumab,3,4 aflibercept,5 and bevacizumab,6,7 have transformed the prognosis of patients with neovascular AMD. For diabetic macular edema (DME) and retinal vein occlusions (RVOs), similar advances have occurred with anti-VEGF drugs8–11 and together with the developments in longer-acting intravitreal steroids,12,13 ophthalmologists now have better alternatives to offer their patients than ever before.
However, the initial optimism surrounding these treatments has been tempered by a slow realization that real-life outcomes are less favorable than clinical trials and that neovascular AMD may have a much longer active phase than previously realized.14 Health care providers are struggling to meet the increasing financial demands15 of providing costly anti-VEGF services over a prolonged period, but perhaps more important is the burden on patients, carers and health care staff to maintain an effective therapy service that requires long-term, regular monitoring and treatment visits.16 This has led some commentators to question whether health care economies can hope to meet the ever-increasing demand of delivering drugs by intravitreal injections.17
It is therefore not surprising that attention has turned recently to safer, less-invasive and more cost-effective ways of delivering drugs to the posterior segment. One of the routes that is being reconsidered is the use of topical eye drops for retinal disease. This has traditionally been regarded as a poor way of delivering drugs to the posterior segment, but perhaps new developments discussed in this review will help challenge that belief.
Pharmacology of Topical Drug Delivery to the Eye
Amongst other attributes, the ideal drug for retinal disorders should be easy to administer, safe, have a long duration of action, and be costeffective. Needless to say, to date, no such drug exists as far as we are aware, but how close does topical delivery come to meeting these aspirations? Compared with intravitreal drug delivery, topical delivery has some obvious advantages and disadvantages and these are summarized in Table 1.
Maurice wrote a seminal review of this subject in 2002, in which he was particularly interested in the passage of drugs delivered by eye drops to the posterior segment and their effect on the blood supply of the retina and optic nerve.18 He considered the experimental animal evidence of drug entry from eye drops to the posterior segment and concluded that only minute amounts of drug could theoretically get to the retina, but acknowledged that they might still have a therapeutic action. He described two routes by which eye drops could reach the retina:
- Corneal—into the anterior chamber and then through the lens, the pupil, or the iris or its route.
- Conjunctival—either directly across the sclera, choroid, choriocapillaris, and retinal pigment epithelium (RPE); or indirectly into the retro-bulbar space and thence the optic nerve head.
Eye drops are in widespread use and comprise about 90 % of all ocular medications.19 Despite this, eye drops have considerable disadvantages as a means of delivering drugs into the eye, not least because the maximum volume that the palpebral fissure can contain under normal conditions, without blinking and in an upright position, is 30 μl.20 Furthermore, the protective mechanisms of the eye, in the form of blinking and baseline and reflex tearing, rapidly remove most of the eye drop-delivered drug from the surface of the eye, and therefore limit the time for drug penetration through the cornea, which for hydrophilic drugs may only be from 2 to 5 minutes.21 To get around this, the contact time and thereby absorption of topical ocular medication can be prolonged by formulations such as gels, ointments, inserts, and contact lenses.22
The corneal epithelium is a further barrier to drug penetration into the eye, and is considered to be the rate-limiting factor for most drugs, particularly for hydrophilic compounds. It has been estimated that typically less than 5 % of topically applied drug permeates the cornea and reaches intraocular tissues.22–24 A large portion of drug applied by eye drops is absorbed systemically, either via the conjunctival and eyelid blood vessels, or via the nasopharyngeal mucosa.
The noncorneal or conjunctival/sclera route of absorption involves penetration through the conjunctiva and sclera and then into intraocular tissues. For some drugs that have poor corneal permeability, the conjunctival/ sclera route is thought to be significant and is probably effective because of the larger surface area of the conjunctiva/sclera (approximately 17 times greater than the cornea) and because the conjunctival epithelium is much more permeable than the corneal epithelium and allows for the passage of larger solutes.23,25 The sclera is a connective tissue consisting of 68 % water and probably acts more like a sponge than a true barrier to drug penetration.25 The RPE has been found to be 10–100 times less permeable than the sclera for large or hydrophilic drugs.26
The individual physico-chemical properties of the drug itself are probably the most important rate-limiting factors for posterior segment drug delivery. Such factors would include: the molecular radius; the molecular weight; the lipophilicity; the ionic charge; the shape of the drug; and the protein- and melanin-binding properties.25 The formulation of eye drops is also crucial. Ophthalmic suspensions are composed of drug particles usually suspended in an aqueous phase, for example prednisolone acetate, which require shaking before use to ensure that all the particles are evenly distributed and not settled at the bottom of the bottle. This can cause problems with achieving uniformity of dosing27 and suspensions are likely to suffer flocculation—the clustering in the suspension of particles into larger clumps of drug.
Nonsteroidal Anti-inflammatory Drugs
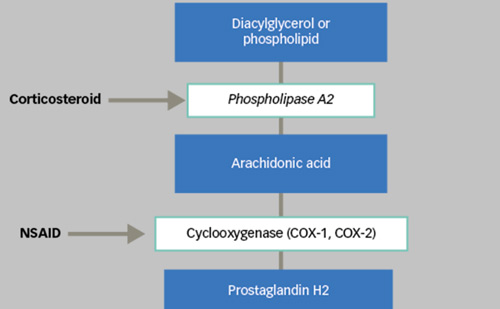
Topical ophthalmic nonsteroidal anti-inflammatory drugs (NSAIDs) have a long history of use as an adjunct to cataract surgery where they have been primarily used for the treatment of postoperative pain and inflammation.28 They have anti-inflammatory properties mediated by the inhibition of the cyclo-oxygenase (COX) pathway and prostaglandin synthesis. There are certainly three isoforms of COX—COX-1 and COX-2 being the most important—but in ocular disease their relative importance is not clear.29 Following early reports of the benefits of topical indomethacin by Miyake30 and Yannuzzi31 for treatment of postoperative cystoid macular edema following cataract surgery, a variety of NSAIDs have been reported as being effective. Subsequently, several topical NSAIDs have been reported as therapy for this condition including, ketorolac,32,33 diclofenac, and, more recently, nepafenac and bromfenac.
Several topical NSAIDs are available commercially, including ketorolac, diclofenac, nepafenac, bromfenac, and flurbiprofen. Ketorelac is reported to be the most important inhibitor of COX-1 whilst bromfenac and amfenac are the most potent inhibitors of COX-2.34
One concern about NSAIDs in prolonged use is corneal toxicity and, in severe cases, corneal melting, which was initially reported in 1999 in patients using topical diclofenac.35 Subsequently, it was suspected that a vitamin E solubilizer in the formulation of diclofenac was the cause of these severe reactions.36 However, as a result of these concerns, topical diclofenac was withdrawn from production for a period. However, this adverse effect is not so much a concern with newer preparations and may be ameliorated when the drug is put in a nanoparticle formulation.37
Bromfenac and nepafenac are NSAIDs that have excellent corneal and sclera penetration and have both been shown to reach the retina in therapeutic doses in animals,38,39 although bromfenac would appear to be superior.40 Recently, there has been interest in using bromfenac, ketorolac, and other NSAIDs as an adjunct to anti-VEGF drugs or intravitreal steroids41 for macular edema from DME, RVO,42 uveitis,41 and also choroidal neovascularization (CNV), as there is evidence that COX plays an important role in promoting angiogenesis in CNV.43
Some recent reports are encouraging, as they have shown that topical NSAIDs may be used as a combination therapy with anti-VEGFs for neovascular AMD44 and may have a synergistic effect. One result of this may be that topical NSAIDs used as a combination therapy will enable fewer anti-VEGF injections to be given,45 which would be a major potential benefit for hard-pressed injection services and for patients and carers. However, Zeifel et al., in a retrospective study of 21 patients, found no benefit of adding twice-daily topical bromfenac (0.9 %) to intravitreal bevacizumab or ranibizumab,46 although other more recent prospective studies have reported benefit from twice-daily bromfenac and further studies are required.47
Steroids
Traditional formulations of topical steroids, such as prednisolone acetate, penetrate very poorly to the retina and vitreous48 and even in aphakic eyes have little effect on postoperative macular edema.49 They also suffer the disadvantage of being formulated as ophthalmic suspensions, which have drawbacks for dosing and drug penetration as previously noted. However, more modern formulations have shown promise and difluprednate, a new steroid formulation, has an oil-in-water type lipid emulsion, which has both lipid and aqueous phases within the formulation. Emulsions allow drugs that are poorly water soluble to be dissolved in the lipid phase and together with the aqueous phase and a suitable surfactant this allows good absorption, more reliable dose delivery, and effective sterility.50
Nakano and others have demonstrated that steroid eye drops (difluprednate ophthalmic emulsion) can be effective in reducing retinal thickness in eyes with DME, in eyes that have been vitrectomized,51 and in eyes prior to vitrectomy.52 In their study of 22 eyes that had undergone previous vitrectomy, but had refractory DME, 11 eyes (in 10 patients) were treated with sub-Tenon injection of triamcinolone and 11 eyes (in seven patients) were treated with difluprednate eye drops four times daily for 1 month and thereafter twice daily, and followed up for 3 months. Comparable improvements of retinal thickness were observed in both groups, confirming clinically that these eye drops achieved therapeutic penetration to the macula. In a follow-up investigation by the same researchers, patients treated with difluprednate ophthalmic emulsion 0.05 %, had a significant improvements in vision and retinal thickness compared with a control group. Although this was a small study with only 3 months follow up, the results suggest that steroid eye drops may have a therapeutic role for DME.
Sigurdsson and co-workers have investigated the efficiency of delivering dexamethasone to the retina in rabbits via eye drops, and whether this can be enhanced by combining it with cyclodextrin.53 Cyclodextrins are circular sugar compounds that have the property of forming watersoluble inclusion complexes thus enhancing the aqueous solubility of lipophilic compounds.54 Sigurdsson et al. found that in rabbit eyes the cyclodextrin-based 0.5 % dexamethasone eye drop was able to deliver significant amounts of dexamethasone to the retina and vitreous by topical administration. This mode of topical drug delivery is promisingand is being investigated for a number of ocular conditions.54
Retinal Neuroprotection and Topical Medication
The strategy of treating a disease by preventing neuronal death is called neuroprotection and was first described in detail by Levin55 in relation to glaucoma, where the concept has gained widespread recognition. By contrast, DR has most typically been considered as a microvascular complication of diabetes, a view that is now beginning to change as it is recognized that neurodegeneration is an early event in the pathogenesis of DR.56 The main features of neurodegeneration (apoptosis and glial activation) have been found in the retinas of donors with diabetes without any microcirculatory abnormalities detected on ophthalmic examination before death.57 Research is beginning to focus on potential promising drugs that might be used as neuroprotective agents to prevent DR.
Brimonidine is an alpha-2 adrenergic receptor agonist, which was introduced in the 1990s as a topical ocular hypotensive agent.58 Brimonidine has been shown to protect retinal ganglion cells, bipolar cells, and photoreceptors in numerous models of experimental nerve injury, including retinal ischemia, ocular hypertension, retinal phototoxicity, and partial optic nerve crush.59 It is currently being evaluated in the EuroCondor clinical trial to test whether it is effective in preventing the development or progression of retinopathy in diabetic persons.57,60
Somatostatin (SST) is a natural neuroprotective and antiangiogenic factor synthesized by the retina, which is downregulated in the diabetic eye. Recent animal research has shown that SST eye drops have a potent effect in preventing retinal neurodegeneration occurring in the early stages of DR.61 Topical SST is currently also being evaluated in the EuroCondor Trial,60 which will be completed in 2016.
Topical Carbonic Anhydrase Inhibitors
There is considerable evidence for the benefit of using systemic carbonic anhydrase inhibitors (CAI) in patients with cystoid macular edema associated with retinal dystrophies.62 CAI are thought to work through their action on membrane-bound carbonic anhydrase receptors present in the RPE, and other cells within the neuroretina. The first report by Cox et al. in 1988, used oral acetazolamide in patients with chronic macular edema of various different causes, but included six patients with retinitis pigmentosa (RP) of whom four showed improvement in vision and two had fluorescein angiographic improvement.63 Subsequently, oral acetazolamide has been used in a variety of types of CME including uveitis and postcataract, but it has probably been most used by ophthalmologists in the treatment of CME associated with retinal dystrophies. However, systemic CAI treatment may be limited by frequent and unpleasant side effects such as limb paresthesia, fatigue, depression, and loss of appetite.
Grover et al. in 1997 reported using topical CAI in retinal disease. They used 2 % dorzolamide ophthalmic solution in five RP patients with CME and compared results in a double-masked, cross-over study with oral acetazolamide. Overall, oral acetazolamide was found to be more effective, although topical dorzolamide did show some improvement of CME by fluorescein angiography.64 Subsequently, several investigators have shown that topical dorzolamide is effective in reducing CME in RP and other retinal dystrophies, but that it may have a limited effect on vision improvement and a rebound phenomenon on stopping the drug has been recognized.65–67
Multi-targeted Kinase Inhibitors
The attraction of developing agents that could be delivered topically and be active against VEGF and other growth factors has led to a quest for smaller molecules than the current larger anti-VEGF drugs, which have to be delivered by intravitreal injection. An example is TG100801, a kinase inhibitor described by Doukas et al.,68 which was found to be active against a number of different growth factors—termed a multi-targeted kinase inhibitor—and that looked promising in animal studies. However, a pilot study (NCT00509548) was terminated and drug development, to our knowledge, has not continued.
Another powerful multi-targeted kinase inhibitor agent is pazopanib, which is approved for use by the US Food and Drug Administration (FDA) as an oral formulation for the treatment of renal cell carcinoma and soft tissue sarcoma. Pazopanib 0.5 % eye drops have been used in a trial investigating corneal neovascularization—preliminary results are promising with no serious adverse events being reported.69 It is lipophilic, has good permeability to the retina, and, although it has been shown to be effective both in reducing retinal vascular leakage and leukostasis in an experimental animal model of diabetes70 and in blocking VEGFinduced vascular leakage in the mouse retina, it was ineffective in the rabbit model.71 Subsequent pilot human clinical trials have unfortunately not been promising.
Regorafenib is a kinase inhibitor that targets multiple kinases, including VEGF receptors 1, 2, 3, and platelet-derived growth factor beta. It has been shown to be effective in an animal model of corneal neovascularization and it is currently being evaluated in a phase I trial of neovascular AMD.72 Topical bevacizumab is a potent treatment for corneal vascularization but in ordinary eye drop formulation (see below) it does not penetrate through the cornea into the anterior chamber and vitreous humor.73
Other Agents
Squalamine lactate (Ohr Pharmaceuticals Inc.) is a small molecule with antiangiogenic properties against a number of growth factors.74 It has shown promising results in phase I and II clinical trials and is currently undergoing further trials in a number of retinal disorders including neovascular AMD, proliferative DR, DME, and RVO. A phase II, randomized, double-masked, placebo-controlled study in neovascular AMD (IMPACT study) is ongoing, but has completed enrollment. Interim results released in June 2014 demonstrated benefit in visual function compared with placebo, and further results are awaited.
Nicotinic acetylcholine (nACh) receptors are found in vascular endothelium and are thought to play a role in controlling vascular permeability in the retina and the brain, as well as stimulating CNV.75 Mecamylamine is a nonspecific receptor antagonist nACh that was used as an oral antihypertensive agent in the 1950s. It reduces blood pressure by blocking neuronal nAChmediated transmission at sympathetic ganglia, permitting blood vessels to dilate. In a multicenter phase I/II clinical trial, 1 % mecamylamine eye drops were given to patients with chronic DME, twice daily for 12 weeks. The results of this pilot trial were mixed with some patients improving, some unchanged, and some deteriorating. Overall, however, the drug was well tolerated with no drug-related safety problems.76
OC-10X (Ocucure Therapeutics) is a selective tubulin inhibitor under development for the treatment of neovascular AMD as a topical agent and may be a promising new approach to treatment. It is a quinazolinone, is highly lipid soluble, and has a low molecular weight of 300 Daltons and has good penetrance into the retina, choroid, and vitreous. A phase I trial (NCT 01869933) assessing the ocular and systemic safety and tolerability of this drug was completed in 2013.
Serine-rich protein kinase-1 (SRPK1) has been identified as a regulator of pro-angiogenic VEGF splicing. Recent laboratory studies in rodent models have shown that SRPK1 inhibitors, namely SRPIN340 and a novel furan molecule, are potent and selective SRPK1 inhibitors and can reduce CNV when given by topical administration.77
OT-551 (Othera Pharmaceuticals) is a disubstituted hydroxylamine compound that is converted to an active component Tempol-H, which has anti-oxidant properties and has been reported to protect RPE cells in vitro and animal experiments.78 OT-551 eyedrops (0.45 %) were used in a phase II trial in 10 patients with bilateral GA, with one eye randomized to treatment and the other eye serving as a control.79 The drug was found to be well tolerated by participants from ocular and systemic aspects, but did not produce any significant benefit in terms of limiting geographic atrophy (GA) lesion area enlargement, although there appeared to be a slight benefit in best corrected visual acuity favoring the study eye. A fellow, larger trial, with 137 participants followed for up to a maximum of 18 months in a randomized, parallel group, placebo-controlled trial also showed no benefit.80
Tandospirone (AL-8309B) is a selective serotonin 1A agonist that has been shown to be neuroprotective in animal models.59 It was evaluated in the Geographic Atrophy Treatment Evaluation (GATE) study, which was a large multicenter, randomized, double-blind trial to evaluate the effect of topical tandospirone 1.0 % and 1.75 % ophthalmic solutions in participants with geographic atrophy. The study failed to show any benefit in terms of mean annualized lesion enlargement rate from baseline and was terminated (Clinical Trials.gov NCT00890097).
Means of Enhancing Drug Delivery to the Retina
The two major limiting factors for using topical medication for posterior segment conditions are the poor drug penetration to the retina and a short duration of action. The strategies that have been employed to increase drug bioavailability can be broadly divided into those that enhance drug penetration and those that provide sustained-release drug delivery, and are summarized in Table 2, modified from the recent paper by Souza et al.81 and are discussed below.
Iontophoresis is a noninvasive method of moving charged active compounds across membranes and it is reported that it was first used on the eye in 1908 by Wirtz, who studied the delivery of zinc salts for the treatment of corneal ulcers, although it was in the 1940s that it became more widely used for anterior segment disease.81,82 It works by the application of a small electric current across the eye and has recently been used to facilitate ocular penetration to the posterior segment as well.83 Currently, several technologies are available for iontophoresis including the Eyegate II System (EyeGate Pharma, Waltham, MA, US), Ocuphor (Iomed Inc., Salt Lake City, UT, US), and Visulex (Aciont Inc., Salt Lake City, UT, US). Recently iontophoresis has been combined with nanoparticle delivery systems to improve the ocular penetration of nanoparticulate drugs.84,85
Nanotechnology (nano from the Greek for dwarf) is a term used for technologies that use the properties of nanoscale substances to develop new functions, and it is being used for many applications in the medical field. In the field of drug-delivery systems, nanotechnology is being used in three ways: to enhance absorption and penetration of drugs across membranes; to allow controlled release of the drug; and to target drugs at distinct tissues.86,87 Liposomes are one sphere of interest in nanotechnology that may prove successful. They are small lipid vesicles composed of a phospholipid bilayer, which have both aqueous and lipid phases. Therefore, watersoluble drugs can be incorporated into their aqueous phase whereas lipid-soluble drugs can be enclosed into their lipid phase,88 which makes it possible to deliver drugs across a wide spectrum of biological membranes. Honda et al.,88 have also highlighted additional advantages that liposomes possess, and these are summarized in Table 3. They are currently being investigated as a means of delivering a wide variety of drugs to the retina, both topically and by intravitreal injection. These include, among others, topical avastin,89 diclofenac,90 nucleic acids and gene therapy,91 and numerous antibiotic and antiviral drugs.92
Conclusions
The ability to use topical medication to treat retinal and choroidal disorders could revolutionize the current care of many blinding disorders. However, the delivery of drugs to the posterior segment by noninvasive, topical means remains an extremely challenging goal, as shown by the small number of topical drugs that have so far been successfully developed. Nevertheless advances in novel drug-delivery systems for the posterior segment using topical medications may usher in a new, safer means of treating major blinding disorders, and bring our quest for the “perfect retinal drug” a step nearer.