Retinal vein occlusion (RVO) is a common vascular disorder with a prevalence of 0.7–1.56%.1,2 It occurs due to thrombosis of a retinal vein and is defined by the location of the obstruction (central, hemi or branch vein occlusion).3–5 Branch RVO (BRVO) occurs at arteriovenous crossing sites that share a common adventitia.5 Central retinal vein occlusion (CRVO) is thought to be caused by external compression of the central retinal artery, which shares a common fibrous sleeve with the vein.6
Retinal vein occlusion (RVO) is a common vascular disorder with a prevalence of 0.7–1.56%.1,2 It occurs due to thrombosis of a retinal vein and is defined by the location of the obstruction (central, hemi or branch vein occlusion).3–5 Branch RVO (BRVO) occurs at arteriovenous crossing sites that share a common adventitia.5 Central retinal vein occlusion (CRVO) is thought to be caused by external compression of the central retinal artery, which shares a common fibrous sleeve with the vein.6
Visual handicap occurs due to macular oedema and neovascularisation, which are secondary to retinal ischaemia.7–11 A number of inflammatory chemokines are thought to be involved in these processes and are found in elevated concentrations in the aqueous humour. Interleukin 6 (IL-6), IL-8, monocyte chemoattractant protein-1 (MCP-1), IL1-α, vascular endothelial growth factor (VEGF) and platelet-derived growth factor AA (PDGF-AA) are found to be elevated in CRVO. There are mixed results in terms of their concentrations in BRVO.12,13
Pathogenesis and Risk Factors
The pathogenesis of RVO is believed to follow the principles of Virchow’s triad for thrombogenesis: vessel damage, stasis and hypercoagulability.14 Contributing factors include: atherosclerosis, inflammatory disease and hypercoagulable/thrombophilia states.15–17 A major risk factor for RVO is hypertension.18–22 Dyslipidaemia is also a prevalent finding.22 Associations have also been reported for diabetes mellitus,18–22 renal disease,23 cigarette smoking18,23 and thrombophilia.16–18,24–26 An additional ocular risk factor is glaucoma or elevated intraocular pressure (IOP), which may compromise retinal venous outflow.18 More than 90% of cases of RVO occur in the >50 age group.14 In patients >50 years of age a cardiovascular risk factor is usually present. In patients <50 years of age there is no obvious risk factor in up to 40% of cases.27,28
Natural History and Complications
The natural history for BRVO is variable but many patients have good prognosis.29 One acceptable management option is follow-up without treatment as half of all patients may reach at least 20/40 vision within six months.30 However, the BVOS study showed that only one-third of patients with macular oedema and vision of 20/40 or worse improved to better than 20/40 after three years with no intervention.7
CRVO tends to have a worse visual prognosis, especially for the non-perfused form.31 One-third of perfused CRVO may convert to non-perfused.28 Neovascularisation may occur in 20% of non-perfused CRVO and 60% of those will develop neovascular glaucoma.31 Initial visual acuity (VA) at presentation is a strong indicator of prognosis. The Central vein occlusion study (CVOS) showed that 65% of patients with initial vision of at least 20/40 maintained at least 20/40 vision. Only 1% of patients reached at least 20/40 vision if starting vision was 20/200 or worse.11
Despite a strong association with vascular disease, RVO does not appear to be an independent risk factor for death from cardiovascular causes.32–34 However, a post-hoc analysis revealed an association among persons <70 years of age.34
Management
There are two aims in the management of RVO: the identification of modifiable risk factors and their medical management and the recognition and management of sight-threatening complications. The current treatment options of RVO intend to minimise the damage, as there is no proven treatment to improve vision loss in the long term. The therapy aims to prevent further visual loss and its complications, such as macular oedema, ischaemia or neovascularisation.
Medical Treatment
The proved association of RVO with systemic vascular disease emphasises the need to screen new patients for vascular risk factors such as hypertension, dyslipidaemia and diabetes. Treatment of these underlying causes is of paramount importance in preventing considerable morbidity. Investigation of thrombophilia should be considered when no other obvious aetiology is present.35 Likewise, ocular risk factors such as glaucoma should be managed. Another medical treatment for RVO is isovolaemic haemodilution; however, the benefits are limited and it should be avoided in patients with cardiovascular (a common co-morbidity with RVO), renal or pulmonary diseases.35 Anticoagulants and antiplatelets treatments have not been proved to have any benefit.36
Laser Treatment
Various medical and surgical treatments have been attempted for RVO with different success rates. Laser therapy remains the gold standard for the treatment of macular oedema in RVO patients, in accordance with the results of the CRVO and BRVO study groups.7,37 Panretinal photocoagulation (PRP) should be considered in CRVO when anterior or posterior segment neovascularisation is clinically detected and confirmed by fluorescein angiography, to prevent progression to neovascular glaucoma.10 It is not recommended as a prophylactic treatment, as it does not prevent neovascularisation development and response to PRP treatment is more likely to occur in eyes that have had no previous treatment. Recommendations include careful observation with frequent follow-up examinations in the early months (including undilated slit lamp examination of the iris and gonioscopy) with prompt PRP of eyes in which anterior segment neovascularisation develops.10,11 Peripheral scatter laser photocoagulation of ischaemic retina is recommended in BRVO in the presence of retinal and disc neovascularisation to reduce the occurrence of vitreous haemorrhage.8
Macular grid photocoagulation is an effective treatment to improve vision in the presence of macular oedema for BRVO.8,11,14 However, this treatment has not been found to improve vision for CRVO (even if the treatment clearly reduces angiographic evidence of macular oedema).37 Until recently, guidelines recommend grid laser for BRVO when vision loss due to macular oedema persists for more than three months without spontaneous improvement and VA is within the range of 20/40 to 20/200.8 However, the results achieved are modest.
Common Surgical Approaches to Retinal Vein Occlusion
There are several treatment strategies that focus on the surgical treatment of the occluded retinal vein, and they are summarised as follows.
Radial Optic Neurotomy
Increased pressure within the confined scleral outlet was hypothesised to cause thrombus at this location as a result of decreased lumen size and increased turbulence. Radial optic neurotomy (RON) was thought to release the pressure, increase the CRV lumen size and venous blood outflow and thus allow thrombosis clearance.38 Moreover, RON was thought to induce the post-operative development of optociliary venous anastomosis or retinochoroidal shunts, leading to increased retinal venous outflow.38–42 The technique38 involved an incision on the nasal side of the optic nerve, radial to the nerve and parallel to the nerve fibre layer.
Although RON has been proposed as safe by most authors, the possibility of serious complications should not be overlooked. The procedure carries a potential risk of laceration of the central retinal artery or CRV, optic nerve fibre damage with visual field loss, globe perforation, retinal detachment, choroidal neovascularisation at the neurotomy site and anterior segment neovascularisation.38,43–45 Careful patient selection may provide better results after RON. Patients with pronounced peripapillary swelling at baseline and with an onset of CRVO of <90 days were reported to be more likely to benefit from RON.42 As a result, the benefits of RON appear to be controversial and its efficacy remains to be proved in prospective randomised clinical studies.
Chorioretinal Venous Anastomosis
Chorioretinal venous anastomosis (CRA), in which a shunt is performed between a retinal vein and the choroids, aims to bypass the occluded vein by an alternative route. It improves retinal outflow and relieves the venous obstruction.46–48 It can be induced by laser or by surgery. The success rate for laser-induced anastomosis has been reported in 33% eyes in the past.47 However, this treatment was associated with choroidal or vitreous haemorrhages, pre-retinal fibrosis, choroidal neovascularisation, segmental retinal ischaemia and retinal detachment.38,46,47 A recent report by McAllister et al.49 evaluated the effectiveness of a laser-induced CRA (L-CRA) as a treatment for non-ischaemic CRVO and found that CRA was created in 76.4% of eyes. Eyes that developed an anastomosis had a significant improvement (11.7 letters) in final VA after 18 months. Complications were progression to the ischaemic category, CNV, macular traction and haemorrhage. They were all managed successfully with careful follow-up and early intervention. Surgically induced CRA has been described by several authors, using several techniques.50–52 Visual improvement has been reported in 60–80% patients.50,52 However, the studies included small sample sizes. One study reported 30% of patients experiencing complications, including retinal detachment, vitreous haemorrhage and cataract.52
Vitrectomy with or without Internal Limiting Membrane Peeling
Pars plana vitrectomy (PPV) with internal limiting membrane (ILM) peeling has been suggested as beneficial for the rapid resolution of retinal damage and macular oedema in patients with RVO.53–55
Decreased retinal thickness and increased VA were observed post-operatively in >70% of RVO cases.53,54 These effects have been shown to persist for up to five years.55
Comparatively successful results have also been reported in vitrectomy without ILM peeling. Vitrectomy itself can provide resolution of macular oedema by removing VEGF and other cytokines within the vitreous cavity and enhance oxygen transport to the hypoxic retina.56,57 A statistically significant improvement in patients after vitrectomy with gas⁄air tamponade for macular oedema caused by BRVO was reported.58,59 However, several other authors do not confirm PPV benefits in eyes with CRVO.60
Direct injection of Tissue Plasminogen Activator via Retinal Vein Cannulation
Direct injection of tissue plasminogen activator (t-PA) includes PPV and an injection of a bolus of 200μg⁄ml t-PA towards the optic nerve head (ONH). Visual improvement was observed in 54% of 28 eyes with CRVO with this technique.61 In another study, a disappointing visual outcome and high complication rates with this surgical technique were observed in a study by Feltgen et al.62 It can lead to serious ocular complications, including vitreous haemorrhage, retinal tear formation, retinal detachment, neovascular glaucoma, endophthalmitis and phthisis bulbi. The Retinal vein occlusion and low-dose fibrinolytic therapy (ROLF) study compared intravitreal t-PA with haemodilution. There was improved VA for acute CRVO but not for BRVO with t-PA compared with haemodilution.63
Arteriovenous Sheathotomy
The surgical procedure includes PPV with posterior hyaloid detachment for the treatment of BVO.64 The end-point of surgery is elevation and separation of the arteriole from the underlying venule confirmed using a subretinal spatula.
Until now, most studies of arteriovenous sheathotomy (AVS) have been uncontrolled and have failed to show a convincing improvement in outcomes of BRVO that would justify the risks of the surgical procedure.65–67
Steroids
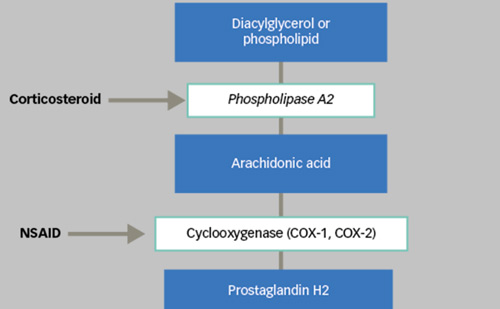
The rationale for the use of intravitreal steroids to treat macular oedema is that corticosteroids reduce retinal capillary permeability and angiogenesis, targeting cytokines such as IL-1b, inter-cellular adhesion molecule 1 (ICAM-1) and VEGF.68–72
Triamcinolone Acetonide
Intravitreal triamcinolone for RVO was first described in case reports in 2002.73 Evaluation of responses to treatment with a range of doses (4–25mg) show short-lived responses. Therefore, for effective treatment it appears that more than one injection is usually needed.74
The phase III multicentre Standard care vs. corticosteroid for retinal vein occlusion (SCORE) CRVO study evaluated a preservative-free form of intravitreal triamcinolone acetonide (IVTA) injection administered every four months (TRIVARIS, Allergan).75 The groups were divided into 1mg, 4mg IVTA or observation groups; the results showed that both doses of TRIVARIS produced both anatomical and functional improvement of macular oedema due to CRVO compared with observation. However, at month 12, the 1mg dose had a better safety profile compared with the 4mg dose in terms of a lower incidence of raised IOP >35mmHg (2%, 14 versus 1% for observation), incidence of cataract formation or progression (25 versus 35%, versus 13%) and need for cataract surgery (0%, 4 versus 3%). As such, laser is considered to have a more favourable risk:benefit profile to TRIVARIS in CRVO.
The SCORE-BRVO76,77 study evaluated the efficacy of different doses of TRIVARIS compared with laser photocoagulation for macular oedema secondary to BRVO. The results showed that both doses of TRIVARIS produced both anatomical and functional improvement, but this was similar in magnitude to laser. In addition, at month 12, both the 1 and 4mg doses had an inferior safety profile compared with laser in terms of a higher incidence of raised IOP (2 and 14%, versus 1%), incidence of cataract formation or progression (25 and 35%, versus 13%) and need for cataract surgery (0 and 4%, versus 3%) As such, laser is considered to have a more favourable benefit to risk profile to TRIVARIS in BRVO.
TRIVARIS is US Food and Drug Administration (FDA) approved, but is not currently widely available clinically.78 There is no evidence to suggest that the visual and anatomical responses seen with TRIVARIS in SCORE-BRVO would be replicated with off-label IVTA preparations such as KENALOG or TRIESENCE.79–81
Dexamethasone Biodegradeable Implant
The short half-life of intravitreal dexamethasone suggested that its use is limited in the management of chronic and/or recurrent macular oedema.82 This led to the development of biodegradable, sustained-release, intravitreal implants containing dexamethasone. They were shown to increase the duration of action of dexamethasone in rabbit eyes.83 Ozurdex (Allergan) is a sustained-release dexamethasone implant that is injected. The GENEVA phase III, multicentre, masked, randomised, sham-controlled, clinical trials have been performed to assess the safety and efficacy of the intravitreal dexamethasone implant (0.7 and 0.35mg) in macular oedema associated with BRVO and CRVO (with six-month open-label extension).84 After administration, the mean time to achieve at least 15-letter improvement in best corrected VA (BCVA) (main outcome measure) was significantly less in both dexamethasone implant groups compared with sham – the cumulative response rate curves for the DEX implant treatment groups separated from the curve for the sham group as early as day 30 (p<0.001). Mean BCVA improvement was greater in both dexamethasone implant groups compared with sham at days 30–180 (p≤0.006). The percentage of eyes with at least 15-letter BCVA improvement was significantly higher in both dexamethasone implant groups than sham at days 30–90 (p≤0.001), but not day 180. Improvements in BCVA with dexamethasone implant were seen in patients with both BRVO and CRVO. In terms of safety, for both CRVO and BRVO patients, raised IOP peaked at month two, but declined significantly by month three and was close to 0% by month six, with 19% of patients requiring an IOP-lowering agent at month six and 0.7% of patients requiring any IOP-lowering surgical procedures. Similarly, rates of cataract progression were low with 7% progression at month six compared with 4% in the sham group.84
Subanalyses from GENEVA also suggest that treating RVO patients with shorted duration of macular oedema results in better outcome for VA. Delaying treatment leads to a reduced possibility to gain significant vision.85
Based on the GENEVA study programme, OZURDEX has received FDA and EU approval for the 0.7mg preparation and is licensed in the UK for the treatment of adult patients with macular oedema secondary to either BRVO or CRVO (OZURDEX SmPC).86
Other Sustained-release Implants
The Verisome® drug-delivery system is an extended-release, biodegradable implant that is inserted into the eye with a 30G needle. IBI-20089 is based on this technology and is a novel triamcinolone slow-release insert that is designed to last up to one year with a single intravitreal injection. A phase I multicentre trial study evaluated the efficacy of this drug in patients with chronic cystoid oedema due to RVO. Two doses were investigated (6.9mg triamcinolone in 25μl and 13.8mg triamcinolone in 50μl). The study showed a good safety profile with decreased central retinal thickness. A 13.8mg dose was found to be more effective than 6.9mg.87 Retisert is a fluocinolone acetonide (FA) implant, which releases 0.59 or 2.1mg FA. It is currently in use for non-infectious posterior uveitis.88–90 It has also been tested in patients with macular oedema secondary to CRVO, achieving an improvement in VA and a reduction of the macular oedema at 12 months.91 However, implanted eyes had higher incidences of IOP elevation and cataracts.88,90,91
Iluvien is a smaller device, that releases 0.2 or 0.5mcg/day of FA and has been investigated for the treatment of diabetic macular oedema with promising results.92
Anti-vascular Endothelial Growth Factor Treatments
Concentrations of VEGF and IL-6 are significantly elevated in the aqueous humour of patients with retinal ischaemia.93–95 These factors have been shown to increase vascular permeability and angiogenesis96 and correlate with the severity of macular oedema and VA loss.94,95,97 This finding is the basis for anti-VEGF therapy in vein occlusions.
Bevacizumab (Avastin)
There are several retrospective and prospective studies showing improved VA and decreased macular oedema with intravitreal bevacizumab.98–105 However, the effect is usually transient (lasting six to eight weeks) and multiple injections are often needed.93,98,106 Most studies show no difference in overall outcome between 1 and 2.5mg doses.98,100,101,107–113 Ach et al. observed that CRVO patients who benefited from treatment were significantly younger, with lower central retinal thickness at baseline for CRVO. However, these predictive factors were not found for BRVO patients.114 Intravitreal bevacizumab has also been successfully used in RVO for reversing iris neovascularisation.115 Bevazicumab may be an important therapy for patients not responsive to other treatments. Rabena et al. reported 27 cases improving with 1.25mg/ml bevacizumab. More than 80% of these patients had been unresponsive to prior laser or intravitreal steroid treatment.101 No significant complications have been reported with this treatment modality although there is a lack of long-term follow-up data.98
Ranibizumab (Lucentis)
Treatment with intravitreal ranibizumab may require repeat treatments to sustain long-term benefit.116–119 Two phase III multicentre, double-masked controlled studies are currently investigating intravitreal ranibizumab efficacy and safety for vein occlusion (in terms of VA, central retinal thickness and visual functioning).120,121
BRAVO (A study of the efficacy and safety of ranibizumab injection compared with sham in patients with macular oedema due to BRVO) included patients with BRVO, VA 20/40–20/320 and macular oedema of >250μm. Patients were excluded if they had undergone laser photocoagulation within four months prior to the study. They were randomised into monthly: 0.3mg, 0.5mg or sham injections for six months. Rescue laser treatment could be used as needed. Six-month results for 397 subjects showed at least 15-letter improvement in: 55.2% (74/134) 0.3mg arm, 61.1% (80/131) 0.5mg arm versus 28.8% (38/132) receiving sham injections. Mean BCVA improvement began day seven (7.6 and 7.4 letter gain in 0.3 and 0.5mg arms, respectively, compared with 1.9mg for sham). More patients in the sham group required rescue grid laser. Serious side effects were rare, concluding it to be an effective, safe treatment for BRVO with macular oedema over six months.120
A Study of the efficacy and safety of ranibizumab injection in patients with macular edema secondary to central retinal vein occlusion (CRUISE) included patients with CRVO, VA of 20/40–20/320 and macular oedema of >250μm. Similar to the BRAVO study, patients were excluded if they had underwent laser photocoagulation within four months prior to the study. They were randomised into groups receiving monthly 0.3, 0.5mg or sham injections. Six month results for 392 subjects show at least 15-letter improvement in 46.2% (61/132) of the 0.3mg arm, 47.7% (62/130) of the 0.5mg arm versus 16.9% (22/130) of patients receiving sham injections. Mean VA improvement began day seven (8.8 and 9.3 letter gain in 0.3 and 0.5mg arms, respectively, compared with 1.1 for sham). Serious side effects were also rare in this study.121
Overall, both studies showed a significant improvement in VA in the ranibizumab group versus the control group with low rates of adverse side effects.96,97 These trials also highlighted that delaying treatment was still superior to no treatment.122
Pegaptanib Sodium (Macugen)
This drug is a pegylated anti-VEGF aptamer that specifically binds to VEGF165 There are limited small-scale studies investigating the effects of this drug for RVO, with promising results.123,124
Vascular Endothelial Growth Factor Trap
This is a soluble human competitive binding VEGF receptor. It is currently under investigation in two phase III studies for CRVO: the General assessment limiting infiltration of exudates in central retinal vein occlusion with VEGF trap-eye (GALILEO) and Controlled phase 3 evaluation of repeated intravitreal administration of VEGF trap-eye In central retinal vein occlusion: utility and safety (COPERNICUS). Patients are randomised to the treatment arm of 2mg VEGF trap every four weeks for 24 weeks versus a control group. The treatment group will receive treatment as needed for one year and be followed up until two years; the sham group will receive sham injections until week 52 and then VEGF trap treatment until week 76. The primary end-point will be the proportion of patients who gain at least 15 letters in best corrected VA at week 24.125,126
To date, anti-VEGF treatments have shown to increase likelihood of VA gain with minimal side effects. However, a high injection rate is often necessary and there is no definitive therapeutic scheme. Often, there is a rebound effect of increased macular oedema after an initial decrease, which may be due to upregulation of VEGF receptors.117 Approximately one-third of patients do not improve with treatment.127 These patients have been shown to have higher aqueous levels of VEGF and tend to be associated with ischaemic RVO.128
Combination Therapy
Available monotherapies show limited results in terms of VA improvement and duration of action. This has led to the rationale of the combination approach to target several mechanisms of oedema and angiogenesis simultaneously. Most current studies combine laser/intravitreal triamcinolone (IVTA) anti-VEGF/IVTA or surgical/ IVTA treatments. 129
Intravitreal Triamcinolone/Grid Laser Treatments
IVTA and anti-VEGF seem to have a rapid but transitory effect in contrast to laser, which displays a slower but longer-lasting result.130 This has led to the concept of combining these modalities. Initial results show beneficial results, although the sample size is small and there is a lack of a control group.79,131–133 A randomised pilot study of 24 eyes compared the effectiveness of subthreshold grid laser treatment with infrared micropulse diode laser alone or in combination with IVTA injection for the treatment for macular oedema secondary to BRVO. Over 12 months the combined subthreshold laser with IVTA showed significant improvement in VA compared with laser alone.134
Anti-vascular Endothelial Growth Factor Trap/Intravitreal Triamcinolone Treatment
Case reports and small case series report favourable outcomes with combining these modalities.135–137 The Treatment of bevacizumab and triamcinolone in treatment or macular oedema secondary to CRVO (MECROV) study aims to compare bevacizumab alone monthly versus bevacizumab monthly in combination with a 4mg intravitreal triamcinlone initial dose.138 The intravitreal avastin versus intravitreal avastin and triamcinolone in CRVO is also listed on the ClinicalTrials.org website.139 To date, no results from this study have been published.
Surgical/Medical Treatment
The benefit of combined vitrectomy (with or without internal limiting membrane peeling) with IVTA is unclear.140–142 Vitrectomy with sheathotomy has been combined with IVTA, showing favourable results of 1–3 Snellen lines for CRVO143–145 and BRVO.146,147 Vitrectomy with sheathotomy in combination with an injection of recombinant tissue plasminogen activator has also achieved VA recovery together with accelerated retinal haemorrhage reabsorption in the treatment of BRVO.148 RON combined with vitrectomy and IVTA has also been described with improved VA and decreased central macular thickness.149,150 In the combined treatment group, in terms of VA and macular thickness, haemodilution treatment for CRVO has been compared with haemodilution with IVTA with improvement.151 Lewis et al. compared maculopexy assisted by gas and/or triamcinolone for BRVO with macular oedema. Patients undergoing gas-assisted treatment showed greater VA improvement than the IVTA group.152 Promising results have been described for triple therapy with IVTA, bevacizumab and vitrectomy.153 However, there is a lack of large, multicentre, randomised controlled trials to support these results.
Conclusion
The currently recommended treatment of RVO consists of identifying and managing cardiovascular risk factors and retinal laser therapy. However, there are no updated clinical guidelines. There are promising results for intravitreal injection therapies as additional therapeutic options. Combination treatments may herald a new treatment era by targeting several disease mechanisms. There are still many unresolved matters for these therapies including number and timing of injections, long-term efficacy and indications for cessation of treatment. Despite new therapies, there are still many patients with visual loss. There is a need for large, well-designed, prospective, randomised controlled trials with a long-term follow-up of new drugs or combination therapies. ■