Physiological medicine is a field that deals with the treatment and prevention of disease by the optimisation of physiology to youthful levels. This field is more properly called multimodal physiological medicine1 because it is of paramount importance to identify and correct as many physiological errors as possible. An analogy, to bring this concept home to the field of ophthalmology, would be to understand that proper refraction requires a sphere, a cylinder and an axis; without all three components, the creation of 20/20 vision is not possible. The refraction of the eye is an example of multimodal physiological medicine. Unfortunately, the medical literature is inundated with single-modal analyses. It is ingrained in the minds of ophthalmologists to study individual substances to deduce their effects and side effects.
This thought paradigm derives from pharmaceuticals that are new, patented creations that can only be studied in this manner. However, single-modal studies are potentially misleading as they will fail to detect circumstances where multiple physiological errors account for a symptom, physical exam finding or laboratory abnormality.
Over the last 15 years many specialists have taken an interest in antiageing medicine2 given the newly understood safety of bioidentical hormones such as oestrogens, testosterone and progesterone.3–5 The phrase ‘antiageing medicine’ is imprecise because the word ‘ageing’ implies time, and to go against time is impossible. Rather, a better understanding of this field would be to look at it as correcting the acquisition of errors of physiology that occur with age and are identifiable in blood testing.
The acquisition of errors in physiology can be directly correlated with a number of diseases, such as high cholesterol, migraine, menopause, erectile dysfunction, depression, anxiety, osteoporosis, attentiondeficit disorder, fibromyalgia and alcoholism.6 The enormous number of conditions that are related to physiological error has led to the concept of convergence. Convergence implies that many diseases that may be thought of as different are actually similar as they derive from root causes in physiology.1 Every speciality in medicine therefore needs to be aware of these causes and to be able to optimise physiology.
Cholesterol and Low Hormone Levels
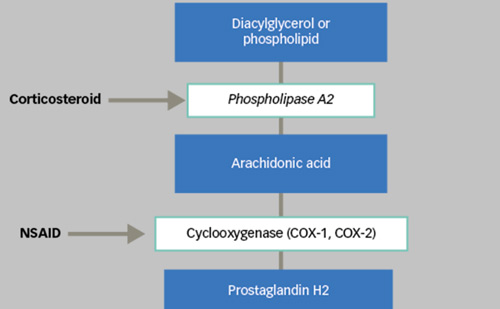
One of the most impressive examples of the concept of multimodal physiological medicine is the elevation of cholesterol. Cholesterol is the precursor molecule from which our steroidal hormones are made (see Figure 1). Cholesterol converts to pregnenolone, which has been called the mother of all hormones. Pregnenolone is converted into dehydroepiandrosterone (DHEA), progesterone, testosterone, cortisol, oestrogens and many other hormones. Dzugan7 showed that elevation of cholesterol is the result of suboptimal levels of steroidal hormones and that restoration of optimal levels of steroidal hormones will lower cholesterol. He explains that the elevation of cholesterol is a physiological compensation attempting to fix the underlying steroidopenia.
Stem Cells and Physiological Optimisation
Stem cells represent one of the most exciting new therapies on the horizon to treat a great number of conditions. Acquired errors in physiology – especially the steroidal hormones – have a negative impact on stem cell function.
DHEA significantly increases the growth rate of human neural stem cells.8 DHEA regulates neurogenesis in the hippocampus and modulates the inhibitory effect of increased corticoids on both the formation of new neurons and their survival.9 DHEA, interleukin-10 (IL-10), IL-4 and melatonin all possess potential regenerative and stem-cell-activating properties.10 DHEA increases the proliferation of progenitor cells.11
Progesterone enhances oligodendrogenesis and production of myelin protein – fundamental steps for repairing traumatic injury inflicted to the spinal cord.12 It has neuroprotective and pro-myelinating effects in lesions of the peripheral and central nervous systems.13
Allopregnanolone (AP-alpha) increases neuroprogenitor cells in the rat hippocampus and human neural stem cells derived from the cerebral cortex. It may serve as a neurogenic/regenerative therapy for restoration of neurons in patients suffering from Alzheimer’s disease.14 Low testosterone is associated with low levels of circulating progenitor cells and endothelial progenitor cells (EPCs). Testosterone treatment increases both of these cell types through a possible direct effect on the bone marrow.15 EPCs decline in the menopause and hormone restoration increases their numbers by 25%.16
Nutraceuticals are also known to optimise adult stem cells. They include vitamins D3, B1, B3 and K, omega-3 fatty acids, folic acid, betacarotene and others.17–23
An Overview of Macular Degeneration and a New Hypothesis
Dry age-related macular degeneration (AMD) is characterised by drusen at the level of the retinal pigment epithelium, which damages photoreceptors, causing degradation of vision. It is the most common cause of blindness in the western world, but there is no known cure and little understanding as to its cause. According to the National Eye Institute, risk factors include smoking, poor diet, lack of exercise, high blood pressure and being overweight.
A new hypothesis has been proposed that multiple acquired errors of physiology, including hormonal deficiencies and imbalances, are in whole or part the root cause of AMD. It is known that such errors include the need for antioxidants, beta-carotene, vitamin C, vitamin E and zinc. All of these were found to be helpful by the longterm Age-Related Eye Disease Study.24 Zinc in particular is beneficial as its deficiency may represent one of the major factors for involution of the thymus and consequent cellular immunological dysfunction. Zinc induces several cytokines, predominantly IL-1, IL-6 and tumour necrosis factor-alpha (TNF-α). It therefore has an immense immunoregulative capacity.25
Macular Degeneration and Hormones
The premise that multimodal hormonal physiological optimisation would be of value to the neurosensory retina began with observations in the brain and the realisation that the retina is part of the brain. Patients with depression, sleep disturbance, fatigue, memory loss, hot flushes and migraine routinely notice improvements in their symptoms when given bioidentical hormones and natural supplements.6
It is known that pregnenolone, progesterone, DHEA and oestrogens are neurosteroids. In animal models, pregnenolone and DHEA have been shown to profoundly stimulate the healing of neurological injury.26–29 Women whose progesterone levels drop develop personality changes.30 Women who enter into menopause at a young age develop macular degeneration, presumably because of the absence of oestrogens and progesterone.31 Blocking oestrogens with tamoxifen is harmful to the retina.32
Steroids such as DHEA, progesterone, testosterone and oestradiol are neuroprotective against ischaemia and traumatic damage.33–37 There is impressive evidence in the literature that testosterone slows down the progression of Alzheimer’s disease.38 DHEA protects the eye against oxidative damage
Patients with Age-related Macular Degeneration Have Low DHEA When it was published that DHEA, a crucial hormone and precursor of other hormones, was found to be exceptionally low in macular degeneration patients43 relative to a control group, an intense investigation of hormones in macular degeneration began. An internal review of 53 consecutive macular degeneration cases (26 male and 27 female; see Table 1) showed that profound deficiencies existed in:
• DHEA levels in men and women;
• progesterone in women;
• oestrogens in women;
• pregnenolone in both men and women;
• progesterone in men (milder deficiency); and
• testosterone in men and women (milder deficiency).
Anecdotally, at the time of writing, patients are volunteering the observation of improvement in dark adaptation, which the authors suspect is a direct stimulatory effect on the retinal pigment epithelium.
Cardiovascular Disease, DHEA and Age-related Macular Degeneration
DHEA deficiency explains the association of macular degeneration with vascular disease, as it is known that macular degeneration is an independent risk factor for stroke and coronary artery disease, as are low levels of DHEA.44,45 In one study, AMD predicted a five-fold higher cardiovascular mortality rate and a 10-fold higher stroke mortality rate.46 After controlling for traditional cardiovascular risk factors, AMD predicted a doubling of cardiovascular mortality. Hormone deficiency is a prime suspect that links the vascular system to the eye.
Neurosteroids in the Macula
A further review of the literature revealed a major article on neurosteroids in the retina by Guarineri et al., who reviewed the importance of hormones.47 In this review article it is shown that the retina is an authentic steroidogenic tissue capable of transforming cholesterol into pregnenolone, DHEA, corticosteroids and sex hormones. This implies that steroid synthesis has a role in visual function (see Table 2).
Most cholesterol is synthesised in the neural retina and transported to the rod outer segment membranes of photoreceptors, where it is required for continuous renewal during life.48 The renewal process also involves the retinal pigment epithelium, where cholesterol is additionally provided from an extracellular source through low-density lipoprotein (LDL) cholesterol receptors and the apolipoprotein E (ApoE) synthesised locally.49 High cholesterol in the retinal pigment epithelium, changes in ApoE expression by the Müller glia of the human retina and apolipoprotein polymorphism have been suggested to increase the risk of macular degeneration.50
Oestrogens in particular are thought to have a role in age- and gender-associated ocular diseases and to prevent retinal ischaemia perfusion injury.51–54
All of these steroids possess antioxidant properties. It is thought that steroids work by lessening inflammation triggered by the immune system, providing neurotrophic factors and enhancing synaptico-genesis and dendritic arborisation through both genomic and non-genomic actions.37,38
Drusen, Cholesterol and Hormones
Recently, Malek and others55–57 found that cholesterol was present in drusen. This led some to think that cholesterol-lowering agents, such as statins, might help in macular degeneration. However, there is concern that statins actually increase the risk of dry macular degeneration advancing to the neovascular form of the disease.58 The presence of cholesterol in drusen suggests that the retina is trying to make hormones but it cannot. As a result, drusen accumulate. In other words, the macula cannot absorb hormones from the blood as the levels are low and the diseased macula cannot convert cholesterol into hormones. This is similar to the problem Dzugan described with regard to hypercholesterolaemia applied locally to the macula.7
Retinal Pigment Epithelium, Stem Cells and Physiological Optimisation
There is growing excitement that stem cells may play a role in macular degeneration. Animal models indicate that it may be possible to re-populate the retinal pigment epithelium with transplantation of stem cells derived from embryos59 or by way of cells that can be ‘induced’ to become similar to embryonal stem cells.60,61 As it appears to be possible to create cells that behave like the retinal pigment epithelium from stem cells, this therapeutic option shows promise. However, surgical intervention would be necessary.
Interestingly, it has also been shown that the bone marrow can produce stem cells that will differentiate into retinal pigment epithelium cells.62–64 There is evidence that retinal stem cells also exist throughout life within the retina.65
Multimodal physiological medicine can potentiate stem cells. For this reason it remains to be seen whether optimisation of physiology alone can regenerate the retinal pigment epithelium. It may be that a combination of stem cell transplantation plus physiological optimisation will be the future for the management of macular degeneration with stem cells. Time will tell.
Discussion
Macular degeneration can be conceptualised, in whole or in part, as an example of a disease caused by the acquisition of multiple physiological errors. The list of physiological errors is extensive and includes those vitamins found useful in the Age-Related Eye Disease Study and melatonin.
A new class of deficiencies that may lead to AMD includes those related to the neurosteroids pregnenolone, DHEA, oestrogens, progesterone and testosterone. It is known that in AMD, DHEA levels are lower than in age-matched controls without AMD. It is also known that the macula needs hormones to function properly. Low DHEA is also associated with cardiovascular mortality, as is AMD. The normal macula has a back-up strategy when blood levels of hormones fall, whereby it can synthesise the hormones it needs. It is speculated that patients whose steroid-producing faculty is not functioning properly will develop macular degeneration if blood hormone levels are low. These patients cannot convert cholesterol to pregnenolone, which results in the accumulation of cholesterol that is found in drusen.
The restoration of hormonal levels to youthful levels should directly stimulate the macula, given that the retinal pigment epithelium has receptors for hormones. Early results indicate that dry macular degeneration patients dark-adapt more easily soon after starting a multimodal physiological medicine programme.
In addition to the fact that the maculas needs hormones, it should be remembered that hormones are anti-inflammatory and antioxidative. The final major reason to provide an optimised physiology with hormones and natural supplements is to maximise the potential for stem cells – whether latent in the macula or derived from bone marrow – to re-populate and regenerate the retinal pigment epithelium. It should be the mission of the ophthalmologist to identify and reverse all physiological errors in patients with macular degeneration in the pursuit of preventing and treating this major cause of blindness in the western world. Restoration of physiology to youthful levels may cause the macula to regenerate or slow the progression of this blinding disease. Further study is warranted to determine whether reversal of physiological error can prevent or treat macular degeneration. However, the current evidence mandates that patients with macular degeneration be physiologically optimised as the therapy itself is safe, will improve the patient’s quality of life and may reduce cardiovascular mortality.