Glaucoma is the second leading cause of blindness in the world, affecting around 65 million people worldwide.1 It has been calculated that 76 million people by 2020 and 112 million people by 2040 will be affected and will require medical care for this progressive optic neuropathy.
Very well-defined guidelines have been issued by the European Glaucoma Society to suggest a correct treatment.2 Despite this, we wish to investigate if the medical strategy adopted for glaucoma is the same all over Europe. The data obtained by International Market Share (IMS), recording drug’s prescriptions in the five biggest European markets: Germany, France, Italy, Spain and UK, have been used to monitor the ophthalmologists choices for glaucoma medical treatment. IMS reports the real sale of any drug and it reflects faithfully the true market behaviour in any country.
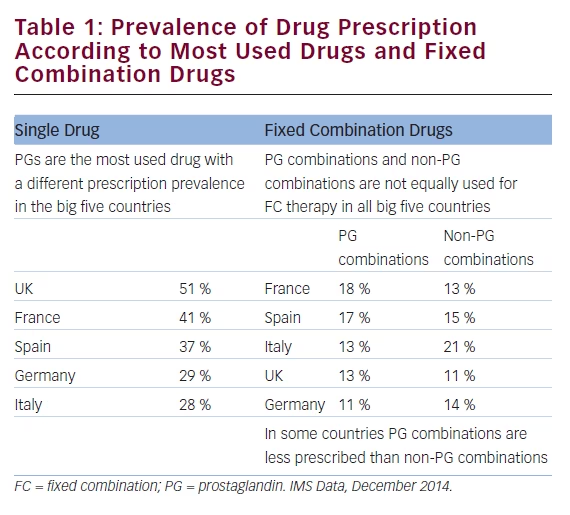
According to our data (see Table 1) prostaglandins (PG) were the most used glaucoma drug all over Europe, although a huge difference was observed among the five countries investigated: PG indeed showed a different prevalence with 51 % in the UK, 41 % in France, 37 % in Spain, 29 % in Germany and 28 % in Italy.
Fixed combination therapy was remarkably different in all the five investigated countries: prostaglandin combinations were more prescribed in France 18 %, Spain 17 %, UK and Italy 13 % and Germany 11 %, while non-prostaglandin combinations were more prescribed in Italy 21 %, Spain 15 %, Germany 14 %, France 13 % and UK 11 % (see Table 1).
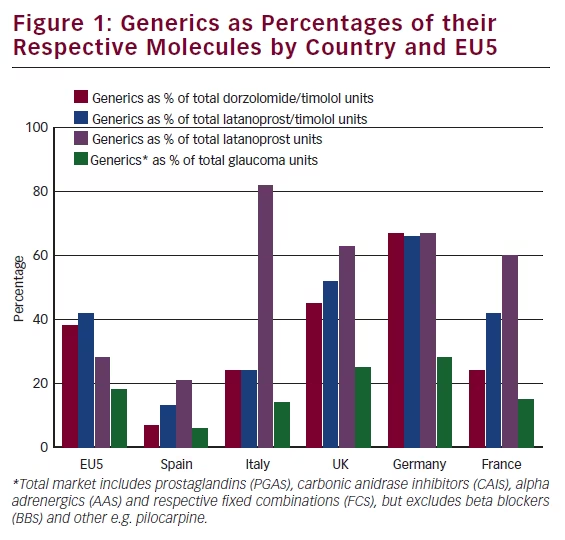
Since 2013, generic latanoprost eye drops have become available in the European market.3 The outcome of generic drugs has represented a real revolution for the ophthalmological market and for the ophthalmologist’s prescription habits. We therefore extended our investigation to the utilisation of generic drugs and prescriptions in the ophthalmic market within the big five countries considered (see Figure 1).
Generic drugs represent actually 18 % of the total glaucoma units in Europe, beta-blockers ex
cluded. This mean value presents however a great variation from country to country. Indeed, generics represent just 6 % of the glaucoma market in Spain compared with 28 % in Germany. If we consider drug sub classes, we have to stress that, in Europe, 58 % of the total latanoprost is generic. This is an enormous variation with Spain, where it represents only 21 %, versus Italy where the generic latanoprost represents 82 % of the total latanoprost.
It is quite hard to state if medical treatment of the same disease, glaucoma, has more similarities or discrepancies in Europe. What is clear is that prostaglandins are the most prescribed drugs everywhere. Generic drugs represent an important market share all over Europe. The different socioeconomic situation of each country, together with local restrictions to healthcare expenditures, may partially explain the differences in combination drug prescription and in generics prevalence.5–7 Also the tendency to start with surgery as a primary option, instead of medical therapy, may explain a different use and prevalence of drugs from country to country.8–10 We did not investigate the prevalence of surgery against medical treatment since we do not have data about surgery prevalence and above all the clinical and costeffectiveness of contemporary medication compared with primary surgery is not known.11