Diabetic macular oedema (DMO) is the primary cause of vision loss in diabetic retinopathy. The treatment of chronic DMO has become a significant clinical challenge, a consequence of the rising prevalence of DMO as well as the significant proportion of eyes that are refractory to treatment. The global prevalence of diabetic retinopathy among individuals with diabetes is around 35 %, with DMO present in 6.8 %.1 Given that the total number of people with diabetes worldwide is projected to rise from 366 million in 2012 to 552 million in 2030,2 so it is estimated that 37.5 million people worldwide will have DMO by 2030, representing a substantial global health burden. The treatment options for DMO include laser photocoagulation and the use of intravitreal drugs that inhibit vascular endothelial growth factor (VEGF), as well as steroids, including, more recently, the use of intravitreal implants.3 This article describes the proceedings of a symposium that evaluated current intravitreal implants, with a focus on the ILUVIEN® implant, which has been approved in Europe for the treatment of visual impairment due to chronic DMO considered insufficiently responsive to available therapies. Recently, the National Institute for Health and Care Excellence (NICE) recommended ILUVIEN as an option for treating chronic DMO that is insufficiently responsive to available therapies if the implant is to be used in an eye with an intraocular (pseudophakic) lens.4
Intravitreal Corticosteroids for the Treatment of DMO
Miss Louise Downey, Hull and East Yorkshire Eye Hospital, UK
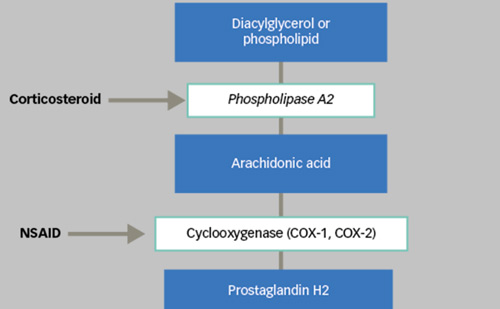
Miss Downey described the various steroid-based products that are available for intravitreal administration: Ozurdex® (750 μg dexamethasone [DEX]); 4 mg intravitreal triamcinolone acetonide (IVTA); ILUVIEN (190 μg fluocinolone acetonide [FAc] intravitreal implant) (see Table 1). The pharmacokinetics of steroids differ in terms of release kinetics: i.e. burst height and duration of release. Their functional response to these treatments is also variable. Some steroids (e.g. TA, DEX) are characterised by high initial intravitreal concentrations of drug (i.e. high burst) followed by rapid reductions, which leads to short duration of response and efficacy. This is in contrast to other drugs (e.g. FAc) where there is a relatively slower build up of concentration to therapeutic levels (low burst), but sustained duration of release and maintained efficacy. The ILUVIEN implant involves a minimal burst and a sustained drug release over 3 years and the pharmacokinetics have been verified in animal studies8–10 and subsequently confirmed in humans.
The intravitreal triamcinolone versus bevacizumab for treatment of refractory DMO (IBEME) study (n=26) compared the morphological and visual acuity (VA) outcomes associated with a single injection of intravitreal TA versus a single injection of bevacizumab. The results showed that intravitreal TA thinned the central macula more effectively over the 24-week study duration and also had a greater impact on VA outcomes.11 Thus leading the authors to suggest that one intravitreal injection of TA is associated with greater beneficial effects on vision and macular remodelling than a single intravitreal injection of bevacizumab for the short-term management of refractory diffuse DMO. The 12-month phase II Ozurdex plus laser versus laser (PLACID) trial (n=253) evaluated Ozurdex’s DEX intravitreal implant combined with laser photocoagulation versus laser alone for treatment of diffuse DMO. Patients in the DEX implant plus laser group received a mean of 1.67 DEX implant injections in this study. The mean improvement in best corrected VA (BCVA) was significantly greater with DEX plus laser than with sham plus laser (+7.9 versus +2.3 letters on the Early Treatment Diabetic Retinopathy Study [ETDRS] scale) at all time-points through to month 9 (p<0.013). In addition, the decrease in central retinal thickness was significantly larger in DEX plus laser group than in the sham plus laser group at four of the eight follow-up visits over the 12-month study.12 The efficacy and safety of ILUVIEN was evaluated in the Fluocinolone Acetonide for diabetic Macular Edema (FAME; n=956) clinical trial. A single injection of low-dose sustained release FAc (0.2 μg/day FAc; ILUVIEN) provided substantial visual benefit for up to 3 years in patients with DMO: a unique duration of action among intravitreal implants. Of the patients still in the trial at month 36, 28.7 % gained ≥15 ETDRS in the ILUVIEN group versus 18.9 % in the sham group.13
In conclusion, the release characteristics of corticosteroid intravitreal implants appear to mirror their efficacy profile, with ILUVIEN offering a favourable pharmacokinetic profile in terms of low dosage, low burst and sustained duration of release providing a 36-month duration of efficacy. Miss Downey stated: “This is really a paradigm shift in terms of intravitreal therapies. For the first time we have a treatment potentially active over three years.”
Efficacy and Safety of Fluocinolone Acetonide in Chronic DMO Patients
Professor Baruch D Kuppermann, Gavin Herbert Eye Institute, University of California, Irvine, US
The ILUVIEN intravitreal implant is a non-bioerodible microimplantable cylindrical polymer tube loaded with FAc.13 It is inserted into the vitreous cavity using a 25-gauge injector, creating a self-sealing wound. The ILUVIEN implant releases 0.2 μg/day of FAc for up to 36 months. ILUVIEN has received marketing authorisation in Austria, France, Germany, Portugal, Spain, Italy and the UK, and is under review in other countries.
Initial in vivo data were collected as part of the Pharmacokinetic and Efficiency Study of Fluocinolone Acetonide Implants in Patients with DMO (FAMOUS; n=37) study and show an initial decline in release rates followed by stabilisation to a consistent submicrogram dose.
The FAME trials were the pivotal licensing studies and consisted of FAME A and FAME B. These were performed under a single protocol as randomised, double-masked, sham injection-controlled, parallel group, multicentre studies conducted over a 36-month period.13 At 36 months, 34.0 % of chronic DMO patients in the ILUVIEN arm demonstrated a ≥15-letter improvement in BCVA over baseline versus 13.4 % in the control arm (p<0.001). The efficacy compared with sham/control groups was greatest in patients with chronic DMO. In this group at 36 months, the mean change in BCVA over baseline was +7.6 letters in the ILUVIEN arm versus +1.8 in the control arm (p=0.004).14 Professor Kuppermann commented as follows: ”That’s quite competitive and quite comparable to anything that’s come out with anti-VEGF. The RISE and RIDE studies which were the most aggressive that had the best result that overall three line gainers for three years was 40 %, but that was with monthly injections … 36 injections.”
In terms of safety, ILUVIEN accelerated the development of cataract, a known side effect associated with corticosteroid therapy; however, excellent visual outcomes were reported following cataract surgery: of pseudophakic patients, 31.6 % gained 15 letters versus 42.3 % that underwent cataract surgery (p=0.173; see Figure 1).15
In summary, the ILUVIEN intravitreal implant has been shown to be effective in clinical studies. The efficacy of ILUVIEN was most marked in patients with chronic DMO (i.e. 34.0 % versus 13.4 %; ILUVIEN versus sham, respectively).
Understanding Intraocular Pressure in DMO Patients Treated with Corticosteroids
Professor Rupert Bourne, Hinchingbrooke, Moorfields and Addenbrookes Hospitals, and Anglia Ruskin University, Cambridge, UK
One of the manageable side effects of corticosteroids is elevated IOP. FAc is a corticosteroid and Professor Bourne reviewed the management of increased IOP in the FAME study. The control and treatment groups had similar mean IOP at baseline (15 mmHg versus 15.2 mmHg, respectively). Over 36 months, in excess of 80 % of those receiving an ILUVIEN implant had an IOP ≤30 mmHg. For those who experienced an increased IOP, this was largely managed using eye drops: IOPlowering medications were required in 14.1 % of control and 38.4 % of patients in the ILUVIEN group, respectively. Laser trabeculoplasty was performed in 0 % of control versus 1.3 % ILUVIEN group and incisional IOP-lowering surgery in 0.5 % control versus 4.8 % ILUVIEN group.13
In 43.1 % of patients treated with ILUVIEN, the IOP never exceeded 21 mmHg. Of ILUVIEN-treated patients who required IOP-lowering medication, most (~70 %) required medical treatment in the first year. According to Professor Bourne, the IOP-lowering medications employed within the treatment group are consistent with current practice and included prostaglandins (17.3 %), beta blockers (17.6 %), topical alpha agonists (17.9 %), topical carbonic anhydrase inhibitors (12.3 %), topical carbonic anhydrase inhibitors + beta blocker (11.7 %) and topical alpha agonist + beta blocker (8.5 %).16
Despite the steroid-induced elevation in IOP that was observed in FAME, there was no significant difference between ILUVIEN and the sham groups in optic disc appearance, indicative of glaucomatous change. The threshold for clinically meaningful change is an increase in vertical cupto- disc ratio of ≥0.2, of which there were only four (of 345; 1.2 %) in the ILUVIEN group.17
One patient in the FAME studies had the implant removed by vitrectomy in order to reduce ocular hypertension. The IOP had returned to the pretreatment level within 25 days of implant removal (see Figure 2). Based on the findings of this patient and the overall trend for mean IOP, it can be assumed that in patients where an IOP occurs, the ocular hypertension caused by ILUVIEN is reversible, and that IOP will return to near-baseline levels when FAc levels are decreased.
In case of raised IOP, there was no negative impact on visual outcomes: neither the IOP elevation associated with the 0.2 μg/day implant nor the use of chronic IOP-lowering medication had any detrimental effect on BCVA.
and Non-chronic DMO Patients20 DMO = diabetic macular oedema; FAc = fluocinolone acetonide. In summary, ILUVIEN has good efficacy with manageable, class-specific side effects; hence, it has a favourable benefit-to-risk profile in patients that have vision impairment associated with chronic DMO. Only 18.4 % of patients had an IOP exceeding 30 mmHg and the time-to-first IOP treatment occurred in the first year in roughly 70 % of those in which an IOP elevation occurred. Elevated IOP associated with ILUVIEN use is manageable with medication and, to date, has not been associated with impaired visual outcomes.
Insufficient Response to Available Therapies in DMO
Mr Robin Hamilton, Moorfields Eye Hospital, London, UK
Mr Hamilton reported that in order to define those “insufficiently responsive to available therapies”, it is necessary to examine data from studies evaluating the efficacy of first-line treatment options in DMO, particularly since “in a lot of units, although we have access to ranibizumab, we do not have the capacity to give a lot of ranibizumab”. The Three-Year Outcomes of Individualized Ranibizumab Treatment in Patients with Diabetic Macular Edema (RESTORE) study was a phase IIIb, multicentre, 12-month, randomised core study (n=303) with a 24-month open-label extension study (208 patients completed this study), in which patients were eligible to receive individualised ranibizumab treatment at the investigators’ discretion. Data from the RESTORE extension study shows that at 12 months visual outcomes were significantly better for both the arms receiving ranibizumab (+7.9 letters, [prior ranibizumab] and +7.1 letters, [prior ranibizumab + laser] versus +2.3 letters [laser monotherapy]). However, by 36 months the difference between treatment groups had narrowed (+8.0 letters, [prior ranibizumab] and +6.7 letters, [prior ranibizumab + laser] versus +6.0 letters [laser monotherapy], although patients with ranibizumab alone performed better than those with ranibizumab plus laser. The data suggest that the effects of ranibizumab appeared to plateau after 12 months.18
In the Ranibizumab Injection in Subjects With Clinically Significant Macular Edema (RISE and RIDE) trials patients were randomised equally (one eye per patient) to monthly 0.5 mg or 0.3 mg ranibizumab or sham injection. In the third year, sham patients, while still masked, were eligible to cross over to monthly 0.5 mg ranibizumab. Results showed that adults with DMO of longer duration or where treatment with ranibizumab was delayed may not respond as well to anti-VEGF therapy as those with disease of shorter duration or receiving early treatment with ranibizumab.19 These observations arose when it was observed that despite improvements in retinal anatomy that occurred in the control population when commenced on rescue ranibizumab therapy, BCVA did not improve significantly. Mr Hamilton commented: “Early treatment of patients with DMO with anti-VEGF is great. But in those patients with chronic DMO as seen in RISE and RIDE it may not be so good and we may be considering using other therapies”. Similar trends in poorer response to treatment come from comparing the non-chronic control arm in FAME with the chronic control arm (see Figure 3). Thus duration of disease seems to have a negative impact on treatment with the chronic DMO control arm showing reduced response to the best standard of care (see Figure 3, top right panel) versus the non-chronic DMO group (see Figure 3, top left panel).20
By contrast, the relative efficacy of ILUVIEN compared with sham/ control groups was greatest in patients with chronic DMO in the FAME trials (see Figure 3).20 At trial entry the duration of DMO was 3.6 years in the ILUVIEN group.21 Reasons for the better efficacy of steroids in chronic DMO might be explained by the action on multiple pathways that are involved in the pathogenesis of DMO, unlike anti VEGFs where the therapeutic effects only affect one pathway. In this context it is worth noting that while VEGF is an important mediator in the pathogenesis of DMO, retinal inflammation is likely playing an important role as the disease progresses.22 The levels of cytokines in vitreous are related to the severity of DMO23 and supporting evidence is available from a prospective, interventional case series (22 eyes of 11 patients with bilateral DMO and six eyes of six patients undergoing cataract surgery) that investigated the changes in aqueous inflammatory and angiogenic cytokine levels after intravitreal injection of TA or bevacizumab in patients with bilateral DMO. In each patient, one eye received an intravitreal injection of 4 mg TA and the other eye received 1.25 mg bevacizumab. Following injection with bevacizumab (n=11), only VEGF was significantly reduced and all other parameters were unchanged (p>0.05). By contrast, following an injection with TA (n=11), interleukin (IL)-6, IL-8, interferon-inducible protein 10 (IP-10), monocyte chemoattractant protein-1 (MCP-1) and platelet-derived growth factor AA (PDGF-AA) were significantly decreased, but VEGF remained unchanged.24 These data demonstrate that the pathogenesis of DMO is not only related to VEGF, but involves other cytokines suppressed by corticosteroids.
Early Clinical Experience with ILUVIEN in the UK
Mr Tomas Cudrnak, Bradford Teaching Hospital, UK
This presentation focused on a number of case studies illustrating the effectiveness of the ILUVIEN implant. These were severe cases, representing different disease and prior treatment. In accordance with the NICE guidelines, all patients were pseudophakic.
Case 1 was a 72-year-old female with type 2 diabetes. She had received laser photocoagulation twice but BCVA in the left eye had steadily deteriorated from 75 to 60 letters. The central subfield (CSF) thickness of this eye was 343 μm. At this point, the patient received an ILUVIEN implant. Two weeks later, CSF thickness had reduced. At 9 weeks a substantial improvement in BCVA was recorded (76 EDTRS letters; +16 letters), accompanied by a reduction in CSF thickness (–81 μm from the pre-ILUVIEN value).
Case 2 was a 70-year-old female with type 2 diabetes. She had received one laser treatment in her right eye, followed by eight monthly injections of ranibizumab. Prior to ranibizumab, the ETDRS letter score was 63 and 8 months later it was 65 letters (+2 letters). CSF measured 210 μm at this point and ranibizumab treatment was stopped. Three months later the patient was re-examined and BCVA had increased slightly to 68 letters but CSF thickness had increased (to 487 μm). The patient then received an ILUVIEN implant and after 6 weeks, BCVA improved to 75 letters, and CSF had decreased (–120 μm).
Case 3 was a 68-year-old male with type 2 diabetes who had received three laser treatments in his right eye, followed by six ranibizumab injections. However, BCVA steadily dropped reaching 70 after 5 months, while CSF thickness was 387 μm. Another injection of ranibizumab was given, without alteration of BCVA, which was assessed 18 days after the treatment. CSF thickness had increased (to 401 μm). One month later, the patient had cataract surgery and received corticosteroid treatment (DEX) and non-steroidal anti-inflammatory drugs (NSAIDs) (ACULAR, ketorolac tromethamine 0.4 %). At this point BCVA was 73 letters and CSF was 387 μm. The patient received an ILUVIEN implant. Nine weeks later BCVA declined to 65 letters, and CSF had increased slightly (+27 μm).
Case 4 was a 79-year-old man with type 2 diabetes who had undergone cataract surgery and anterior vitrectomy in his right eye in 2008, followed by an iris clip for fixation of an IOL in 2009. The patient received one laser treatment followed by six monthly treatments of ranibizumab. At treatment initiation, his VA was 44 letters, which improved to 61 letters 6 months later. However, CSF had thickened slightly (from 443 to 446 μm). The patient received an ILUVIEN implant at the vitreous base. Two weeks later, his BCVA improved (+10 letters), as did his CSF thickness (–18 μm).
A summary of these cases in given in Table 2. On average BCVA was shown to increase (+6.3 letters) and CSF was shown to decrease (–48 μm) 6.5 weeks after ILUVIEN was implanted. To date, elevations in IOP have not been reported in any of these patients. These ‘real-life’ cases support the data obtained from clinical studies with ILUVIEN.
Summary and Concluding Remarks
The prevalence of DMO is increasing and it is estimated that 37.5 million people worldwide will have DMO by 2030. Given the progressive nature of DMO, effective management is vital. As the disease progresses, steroids have proved advantageous compared with anti-VEGF therapy, due to their ability to affect numerous pathways, in particular reducing the concentration of inflammatory cytokines, in addition to VEGF. The main messages of this symposium are summarised in Table 5.
The effectiveness and safety of the ILUVIEN implant has been demonstrated in the FAME studies. The relative benefit is most substantial in patients with chronic DMO, which is reflected in the licenced indication for ILUVIEN in Europe. These data have been supported by real-use clinical experience and suggest that patients with chronic DMO who are insufficiently responsive to available therapies may respond well to the administration of an ILUVIEN implant. NICE has recommended ILUVIEN as an option for treating chronic DMO that is insufficiently responsive to available therapies if the implant is to be used in an eye with an intraocular pseudophakic lens. The ILUVIEN implant represents an important treatment option that is available to patients with chronic DMO.