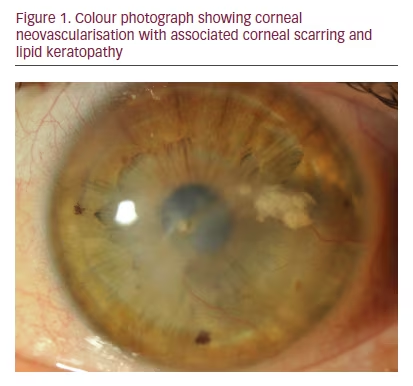
Corneal neovascularisation (CoNV) is a sight-threatening condition caused by new vessel formation from the limbal vascular plexus and marginal corneal arcades and invasion into the cornea in response to inflammation, infection, trauma and hypoxia.1,2 CoNV may lead to profound visual decline by compromising corneal clarity. Pathologic vessel formation may compromise corneal transparency by blocking and diffracting light, causing lipid and protein exudation and serving as a conduit for inflammatory cells that damage the structural integrity of the cornea leading to scarring (see Figure 1).3 As one of the main causes of corneal blindness in developed countries and a major risk factor for immune allograft rejection after corneal transplantation, CoNV represents a major health burden to the public.4 Although the global impact of CoNV is not known, the incidence rate has been estimated to be 1.4 million per year in the United States.5 Current treatment options of CoNV are limited and prevention of visual loss remains the main challenge for clinicians when facing patients with CoNV. Recent investigations, however, have improved our understanding of the complex mechanisms involved in corneal haem- and lymphangiogenesis and new insights into molecular pathways have opened new doors for potential future treatment strategies.6”
Corneal angiogenic privilege
The cornea is a complex sensory organ and its transparency, which presupposes the absence of blood and lymph vessels, is critical for optimal vision. Corneal avascularity is maintained by a highly regulated and delicate balance of naturally occurring pro- and anti-angiogenic factors (angiogenic privilege). Various signaling cascades and molecular mechanisms maintain corneal avascularity under homeostatic conditions.7 Corneal and limbal epithelial cells have an angiostatic effect on the cornea and limbal epithelial cells further function as a barrier against haem-and lymphangiogenesis.8,9 It has been demonstrated that different cytokine traps for angiogenic and inflammatory factors are constitutively expressed by the intact corneal epithelium: soluble vascular endothelial growth factor (VEGF)-A receptor-1 (sVEGFR1) acts as a decoy receptor for secreted VEGF and inactivates membrane-bound VEGF-A receptors 1 and 2 by heterodimerisation;10 VEGFR3 binds and inhibits activation of VEGF-C and VEGF-D which promote lymphangiogenesis;11 and sVEGFR2 controls the ingrowth of lymphatic vessels.12 Other inhibitors of angiogenesis found in the corneal epithelium include angiostatin, which plays a role in the maintenance of corneal avascularity after wounding,13 and pigment epithelium derived factor (PEDF), a serine protease inhibitor responsible for excluding vessels from invading the cornea.14 Furthermore, the cornea actively counteracts hypoxia-driven upregulation of VEGF by low, if any expression of hypoxia inducible factor (HIF)-1a and by expression of IPAS, an inhibitor of hypoxia-driven HIF-1a-signaling.14 The corneal epithelial basement membrane (EBM) also plays an important role in regulating angiogenic privilege: Potent anti-angiogenic factors such as endostatin, thrombospondins (TSP-1 and -2), and tissue inhibitor of metalloproteinase-3 are derived from the extracellular matrix component of the EBM.15–17 Endostatin inhibits the endothelial cell cycle in G1 phase and mitogenic activities of VEGF in vascular endothelial cells by inhibiting binding of VEGF to surface receptors (KDR/Flk-1) and blocking downstream signaling events.18 TSP-1 inhibits angiogenesis
by inducing vascular endothelial cell apoptosis through binding to CD36 receptors19 and binding to CD36 on the surface of macrophages suppresses the TGF-b induced expression of VEGF-C and VEGF-D, which are potent promoters of lymphangiogenesis.20 TSP-2 inhibits cell-cycle progression in endothelial cells in the absence of apoptosis.21 Moreover, heparan sulfate proteoglycans in the EBM have been shown to bind and inhibit VEGF and fibroblast growth factor (FGF-2) and sequester their proinflammatory and angiogenic effects.17 The lower temperature of the cornea, the extensive innervation, and aqueous humor factors further contribute to the avascular state of the healthy cornea.22
Pathogenesis of corneal neovascularisation
Disruption of the balance between pro- and anti-angiogenic factors and overweighing of proangiogenic factors results in pathological vessel formation.1 Although many regulatory factors have been identified, not all mechanisms involved in the development of CoNV are completely understood. Inflammation and macrophage recruitment play a key role for corneal angiogenesis: activated macrophages are known to secrete inflammatory cytokines such as tumour necrosis factor alpha (TNF-α) and VEGF-A, -C, and -D, resulting in the induction of both haem- and lymphangiogenesis and further macrophage infiltration.23–25 VEGF-A is considered to be one of the most important members of the VEGF family and a main driver for pathologic haemangiogenesis.26 Apart from macrophages, corneal fibroblasts and epithelial cells are the most important sources of VEGF-A.27 The action of VEGF on conjunctival blood and lymphatic vessels is thought to be mainly via VEGFR-2 and VEGFR-328 with resultant budding from pre- existing blood vessels at the limbal vascular plexus or from vascular endothelial progenitor cells that express VEGFR-2 (Flk-1), CD34 antigen (a cell-cell adhesion protein) and Tie-2, a receptor for angiopoietin-1.24,30 Further promotors of corneal angiogenesis include FGF, platelet-derived growth factors (PDGF), angiopoietins, matrix metalloproteinases (MMP-2, -9, and -14) and inflammatory mediators such as interleukins (IL-1, -6, and -8), tumour necrosis factor (TNF-α), and transforming growth factor (TGFβ).30
Assessment and imaging of corneal neovascularisation
Clinical evaluation and colour photography
CoNV are often visible on slit-lamp biomicroscopy, and attempts have been made to characterise CoNV based on clinical appearance and anterior segment photography.31,32 Features studied have included the source of vessels (conjunctival, limbal or iris), depth of vessels, branching patterns (straight loops, tortuous or arborising) as well as the presence of leakage causing lipid exudation and corneal oedema, against which a scoring, as well as staging systems for CoNV have been devised.
Clinical observation has shown that afferent vessels are narrower, straighter and situated more posteriorly within the corneal stroma. Clinically they appear to be light red in colour, and the passage of blood through these vessels can occasionally be seen to be rapid and pulsatile. This is in contrast to efferent vessels, which are of larger calibre, more tortuous and darker red in colour. When observed under high magnification red blood cells within these vessels can be seen to tumble or roll more slowly.32
The use of angiography in assessment of corneal neovascularisation
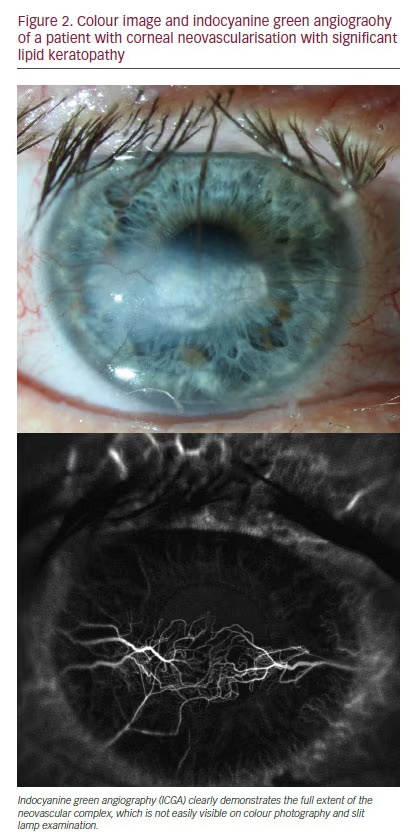
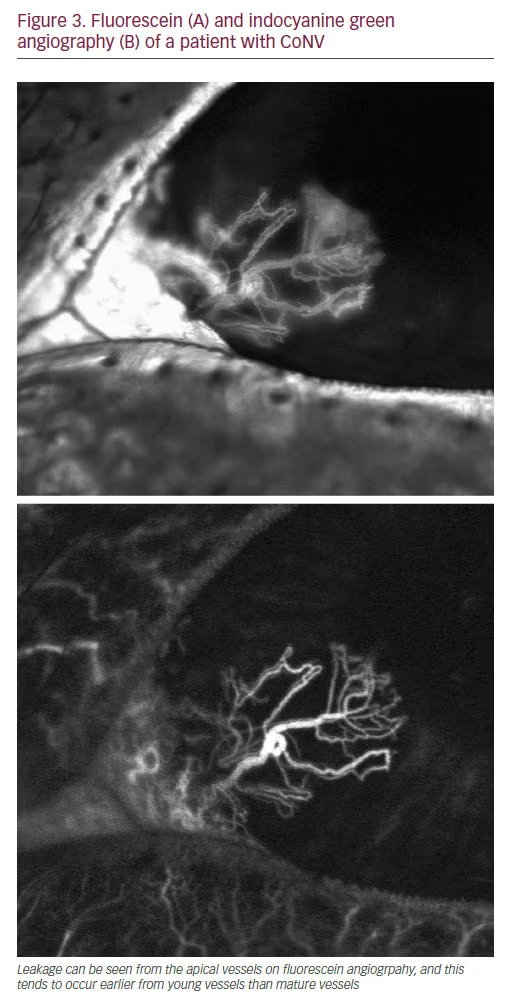
Clinical observation in isolation, however, has various limitations. It is often not possible to assess the full extent of a neovascular complex, and determining the age of vessels can also be challenging.2,33–34 Furthermore, it may be extremely difficult to differentiate afferent from efferent vessels, particularly in large neovascular complexes with numerous vessels crossing the limbus. Corneal angiography with both fluorescein (FA) and indocyanine green (ICGA) has been shown to be extremely useful in this respect.33–35 Publications describing the use of angiography for the assessment of corneal disease date back to the late 1960’s and early 1970’s, however these techniques were not adopted for many years.36 Angiography has been shown to clearly delineate CoNV which are not apparent clinically due to the presence of corneal scarring, and is significantly better at detecting the total area of vascularisation than colour photography (see Figure 2). FA is particularly helpful in establishing the age of CoNV, whereby vessels younger than 6 months tend to leak from the apical vessels significantly earlier than more mature vessels (see Figure 3).33 This can be helpful in guiding treatment as younger vessels may be more responsive to certain treatments such as anti-VEGF.37 Examining the early videography of the angiograms is also very useful to differentiate the afferent from the efferent vessels in a neovascular complex, of which there are normally 1–2 per neovascular complex, thereby enabling the targeting of treatment with cautery.38
Digital subtraction analysis of ICGA and late FA together with in vivo confocal microscopy (IVCM) have been used to further characterise CoNV, and have detected novel presumed lymphatic vessels, which were found only in patients with active CoNV. This work has also been useful in demonstrating the presence or absence of red blood cell traffic in CoNV, and demonstrating that even when no cellular elements are seen within the vessels, the CoNV are still perfused. This may challenge current thinking regarding the relevance of inactive CoNV in corneal transplantation. These methods, however, are technically challenging and require improvements due to difficulties with image alignment.39
En face optical coherence tomography angiography
Ang and colleagues have adapted the use of the AngioVue OCTA system for corneal optical coherence tomography (OCT) angiography.40,41 In their work they have been able to image the normal corneolimbal vasculature as well as aberrant blood vessels in the setting of anterior segment pathology such as early graft rejection, superficial corneal scarring and lipid deposition. Advantages of this technique of imaging CoNV include its rapid and non-invasive nature as well as a relatively shallow learning curve. Furthermore, it provides information on the depth of vessels, which may help guide targeted fine needle diathermy (FND) treatment
of the vessels. In a pilot clinical study, it has shown to be comparable to ICGA for the measurement of CoNV.42 It is limited, however, in that the current technology may produce artifacts that may mimic abnormal vessels. Also, the field of view is limited and it gives no information on vessel leakage. Larger prospective studies with standardisation of the technique of anterior segment OCT angiography are needed.
In vivo confocal microscopy
The use of in vivo confocal microscopy (IVCM) for imaging corneal vessels was first described in 1998 in rabbits,43 and later to study lymphatic vessels in live corneas in rat models.44 IVCM has also been applied to the examination of human corneal blood vessels and lymphatics, and provides useful real-time information about the presence or absence of blood cell traffic, cell densities and cell characteristics in vivo.39,45 IVCM provides useful adjunctive information regarding the activity of
corneal blood vessels, as well as the opportunity to perform serial in vivo assessments in the same patient, thereby enhancing our understanding of the processes involved in the corneal immune response.
Management of corneal neovascularisation
Treatment for CoNV can be broadly divided into medical and surgical, and can be challenging. Modalities employed in the 1950’s to 1970’3 included cryotherapy, surgical/diathermic peritomy, lamellar grafts, scar tissue barrier and ionizing rays. These were of limited efficacy and often fraught with complications.46
Pharmacological treatment
Corticosteroids
Topical steroids are widely used for the treatment of CoNV and currently represent the mainstay of medical treatment for suppression of actively proliferating vessels.47,48 Although not fully understood, the antiangiogenic properties of steroids are thought to derive from their broad anti-inflammatory properties including inhibition of pro-inflammatory cytokines (IL-1, IL-6 and TNF-α) and cell recruitment,49–52 inhibition of prostaglandin synthesis and arachidonic acid release,53 inhibition of plasminogen activator54 and inhibition of budding and sprouting of new vessels.47 While steroids are effective in controlling the early phase, inflammation-induced angiogenesis, the suppressive effect on CoNV may be incomplete and full regression of established vessels may not be achieved.30 Potential side effects including glaucoma, cataract formation, corneal thinning and increased risk for infections need to be considered particularly with prolonged use of topical corticosteroids.46
Nonsteroidal anti-inflammatory drugs
Several nonsteroidal anti-inflammatory drugs (NSAIDs) including flurbiprofen, indomethacin, ketorolac, diclofenac and nepafenac have been reported to inhibit CoNV in experimental studies.46 NSAIDs inhibit the enzyme cyclooxygenase (COX-2 isoform) expressed by new angiogenic endothelial cells that converts arachidonic acid to prostaglandins (E2). Eicosanoids produced by COX-2, amongst other factors, mediate VEGF expression in inflammation-associated angiogenesis.55–57 However, the variable efficacy and risk of corneal melting and perforation associated with its long-term use curtail the clinical utility of NSAIDs for the treatment of CoNV58 and these agents are therefore not considered as first line agents.
Cyclosporine A
Cyclosporine A (CsA), an immunomodulatory agent with inhibitory effects on several T-cell functions, has been shown to inhibit CoNV in in vitro studies and animal models, presumably by inhibiting the effects of IL-2 and the inhibition of COX-2, the transcription of which is activated by VEGF in primary endothelial cells.59–61 The role of CsA for the treatment of CoNV in human corneas, however, remains highly controversial as several studies have provided conflicting results on the efficacy of systemic and topical CsA, especially in the context of corneal graft rejection.62–64 Bock et al. reported no significant effects of high-dose subconjunctival CsA implants on CoNV after high-risk penetrating keratoplasty and concluded that local CsA has negligible antiangiogenic effects in the human cornea, at least in the transplant setting.65 Moreover, Gottsch and Akpek reported a stimulatory effect of topical CsA on CoNV in a case series of patients with sterile rheumatoid central corneal ulcers.66
VEGF inhibitors
Vascular endothelial growth factors (VEGFs) have been recognised as the most important regulators of corneal angiogenesis67,68 and the development of anti-VEGF compounds for targeting angiogenesis has introduced new prospects in the management of CoNV in recent years.69 Promising results have been reported for the use of anti-VEGF agents including pegaptanib, bevacizumab, ranibizumab and aflibercept in both experimental animal models and clinical human trials.30,70–71 Pegaptanib is a 28-base ribonucleic aptamer that specifically binds VEGF165, an isoform of the VEGF-A family, with high affinity. The selective binding of pegaptanib may explain its limited antihaem- and antilymphangiogenic efficacy when compared to ranibizumab and bevacizumab, which are capable of pan-isoform suppression.72–73 Bevacizumab is a full-length, recombinant humanised murine monoclonal antibody (molecular weight 149kD) that binds to and inactivates the biological activity of all five human isoforms of VEGF-A.74 Ranibizumab is the Fab fragment from the same monoclonal antibody used to create bevacizumab, which has been affinity-matured to optimise its VEGF-A binding potential. Ranibizumab is approximately one-third the size of bevacizumab (48kD), theoretically allowing for better corneal penetration compared to bevacizumab.30 Aflibercept is a VEGF-trap molecule that was created by combining the second domain of VEGFR-1 and the third domain of VEGFR-2 with human IgG Fc fragment. It acts as a receptor decoy for all isoforms of VEGF-A and has the highest affinity and longest duration of action of all VEGF blockers currently studied.75,76 The fact that it also binds placental growth factor (PIGF) -1 and PlGF-2 may potentially enhance its antiangiogenic response.71
Bevacizumab and ranibizumab have the capability of partially inhibiting or reversing CoNV, either by topical or by subconjunctival administration, although the reported clinical responses varied in different animal models and human studies.77–83 A recent review and meta-analysis by Papathanassiou et al.70 found a significant improvement and reduction of CoNV upon bevacizumab treatment in animal and human eyes. The overall reduction of neovascular area from baseline was reported to be 36% in human eyes. The effectiveness of topical and subconjunctival bevacizumab for CoNV is comparable,70,77–78 although topical administration may require higher doses than subconjunctival injections for equal treatment responses.84 However, subconjunctival administration of bevacizumab may have better outcomes on graft rejection rates as demonstrated in the mouse model.71 No significant differences have been reported comparing the efficacy and safety profile between bevacizumab and ranibizumab for both topical and subconjunctival administration.30,85 Although topical ranibizumab and bevacizumab both effectively reduced corneal neovascular area, vessel caliber and invasion area in two prospective studies on human eyes, ranibizumab may be favourable in reducing CoNV and have an effect earlier in the course of treatment than bevacizumab due its smaller molecular size and better penetration through the corneal epithelium.30,86 Subconjunctival injections of ranibizumab and bevacizumab have been shown to result in significantly less CoNV in rat models compared with placebo, with no significant difference between these two anti-VEGF agents.85 Most clinical studies suggest that the topical or subconjunctival use of bevacizumab and ranibizumab is a relatively safe and welltolerated option for the treatment of CoNV with no reported serious side effects.70–71,87–88 Intrastromal injections of anti-VEGF might be an efficient and safe alternative89 and may be considered particularly for intrastromal neovascularisation and in the presence of lipid keratopathy.90–91 Ahn et al. used intrastromal ranibizumab in a case of CoNV refractory to bevacizumab treatment and found satisfactory results.92
Bevacizumab treatment in human trials usually consisted of topical drops ranging from 0.5–1.25% given 2–5 times daily and/or single or repeated subconjunctival injections of 1.25–2 mg and intrastromal injections of 0.25 mg.70,89 In the few studies using ranibizumab on human eyes the drug was usually applied topically as 1% drops four times daily.30,82 The optimal dose and frequency of anti-VEGF agents to reach the therapeutic concentration and achieve maximal effect in the cornea as well as the optimal duration of treatment have yet to be clarified in future trials.
Whether VEGF blockage mainly inhibits active or also stable CoNV remains controversial. Some studies reported better outcomes when treating more immature vessels in the active early phase of corneal angiogenesis.37,93 Previous investigations have demonstrated increased expression of VEGF and its receptors in actively growing vessels79 and it is known that anti-VEGF agents have an inhibitory effect on mural cell recruitment and thus vascular maturation,94 which may explain the more potent effect of anti-VEGF on more immature vessels. Pericyte recruitment to vessels maintains the stability and maturation of new vessels, and the endothelial cells become less dependent on VEGF at this stage, which may explain the limited anti-angiogenic efficiency of anti-VEGF for later stage CoNV.37,95 In addition to this favourable response to anti-VEGF in active CoNV, other studies have reported that stable CoNV could also be treated effectively and reduced significantly by anti- VEGF treatment.70
Aflibercept may hold promise as an effective modality for use in patients with CoNV. Two recent studies have demonstrated an inhibitory effect of topical and subconjunctival aflibercept on CoNV in animal models.96,97 Gal-Or et al. reported significant inhibition of CoNV after subconjunctival application of 25 mg/ml aflibercept in rats compared to controls and better efficacy compared to equivalent doses of subconjunctival bevacizumab.96 Park et al. treated rabbits with topical aflibercept in two different doses (0.01% and 0.1%) and compared the inhibitory effect on CoNV with topical bevacizumab (0.1%). Treatment using both concentrations of aflibercept seemed to be effective and the differences from the control were statistically significant. No superior effect of 0.1% topical aflibercept, however, compared to an equivalent bevacizumab dose was found.97 Further studies including clinical trial on human eyes are needed to evaluate the efficacy, dosage, and safety of aflibercept for the treatment of CoNV.
Aganirsen (GS-101)
Aganirsen (GS-101) is an antisense oligonucleotide that inhibits the mRNA expression of insulin receptor substrate-1 (IRS-1) proteins, thereby controlling engagement and overexpression of IRS-1, which is thought to be an important mechanism in corneal angiogenesis.98,99 The anti-angiogenic and angio-regressive effects of topical aganirsen have recently been demonstrated in eyes with CoNV following bacterial, viral or traumatic keratitis in a randomised, controlled phase III study.100 The relative neovascular area was significantly reduced by 26.20% after 3 months of treatment at a daily dose of 86 mg compared with placebo and an improvement of the neovascular area of 26.67% could still be observed three months after discontinuation of aganirsen. Furthermore, the need for transplantation was significantly decreased in a small subgroup of patients with viral keratitis and central CoNV. Topical application of aganirsen was safe and well tolerated. However, no positive effect of aganirsen on visual acuity compared with placebo was observed and further studies are necessary to support and confirm the clinical benefit of this novel agent.100
Surgical and parasurgical treatment of CoNV
Laser treatment
Argon laser uses light-induced thermal damage to ablate vessels and is an established treatment for retinal neovascularisation. Haemoglobin has a very high absorption rate of argon energy, which can therefore coagulate haemoglobin-filled corneal vessels. Argon laser beams can be used at the slit lamp and be precisely focused, adding a high degree of specificity to the procedure.101 Despite successful results being reported in cases of lipid keratopathy (reduction in extent of CoNV by 62%, in density of CoNV by 49% and improvement in visual acuity [VA] by 48%),102 different limitations of this technique have been highlighted including technical difficulty because the vessels are deeper, narrower and have rapid pulsatile flow;103 impermanence of the occlusive effect104,105 and vessel recurrence;106 and thermal destruction of vessels with increased production of inflammatory mediators and up-regulation of VEGF.107 In addition it must be taken into account that the high laser energy predisposes to complications such as iris atrophy, corneal thinning, pupillary ectasia, peripheral corneal haemorrhage99,108–109 and necrotising scleritis.110 There are also a few studies reporting that the vasocclusive effects of yellow laser are comparable to argon laser.111 The hypothesis behind the choice for yellow laser over argon blue–green laser was on the basis that yellow was absorbed more by oxyhaemoglobin and reduced haemoglobin, therefore requiring less total energy.112 Another option is Nd:YAG (1064 nm), however in a comparative study with argon and yellow lasers (570 nm) authors noted that the Nd:YAG requires tissue necrosis to occlude the vessels, thus making it a potentially poorer choice.113 Neither of these lasers are routinely used in the clinic.
Photodynamic therapy
Photodynamic therapy (PDT) has been used successfully in the treatment of choroidal neovascularisation. A porphyrin derivative such as verteporfin is administered systemically, and accumulates in abnormal blood vessel endothelial cells. When activated by non-thermal red light laser, cytotoxic oxygen free radicals are produced resulting in damage to the vascular endothelium by necrosis and apoptosis, resulting in thrombotic occlusion of the target vessels.114 PDT has been applied to the treatment of CoNV and has been shown to be safe and efficacious in various animal models in reducing the number and length of blood vessels clinically and on histopathological examination.115 PDT for treatment of CoNV in humans was first described in 2003 on a single patient with CoNV in a penetrating keratoplasty.116 Subsequently, Yoon and colleagues conducted a prospective interventional case series of verteporfin PDT (6 mg/m2) on 18 eyes with refractory CoNV. They found that at 12 months, 77.8% of patients had a reduction in CoNV detected on anterior segment photography, with complete occlusion in 50%, and a significant mean reduction in area of neovascularisation. There were no significant complications other than mild stromal haze in one patient.117 Al-Torbak also presented similar findings in his retrospective review of 33 eyes treated with PDT with complete occlusion of CoNV in 42.4% and partial occlusion in 24.2% of eyes, again with no systemic or ocular complications. He reports that corneas with extensive neovascularisation may require several treatments in order to achieve adequate results. Furthermore, response to PDT was significantly lower in eyes with deep corneal neovascularisation.118 PDT with verteporfin has also been combined with other treatments including subconjunctival bevacizumab, achieving a slightly higher rate of complete occlusion of 66.7%,119 and so called triple therapy with subconjunctival bevacizumab as well as triamcinolone acetonide in a single case, which required retreatment.120 PDT has been found to be effective if applied to a noninflamed cornea where the dye is retained within the CoNV, such that the photo oxidation reaction is confined to the blood vessel. When applied in this scenario, PDT is relatively selective for CoNV, with little damage to surrounding tissue, making multiple treatments possible for recalcitrant vessels. However, in an inflamed cornea, the vessels have increased permeability, leaking dye into the stroma. In this situation the laser energy exacerbates the inflammatory reaction and is more likely to cause reperfusion.121 Furthermore, recurrence, high cost of the dyes and of the dedicated diode lasers, as well as potential local and systemic complications have limited its acceptance.100,122
Fine needle diathermy
FND occlusion of CoNV has been described as an effective and relatively easy technique.37 The potential adverse effects of FND should be taken into account and diathermy should be limited to the afferent vessels (selective FND).123,124 Some corneal changes after FND have been welldescribed, whereas others such as cellular changes are less understood. Feldman et al showed that radial thermokeratoplasty causes damage to the corneal endothelium beneath and surrounding the coagulation site.125 The corneal heating modifies corneal curvature.126,127 Collagen shrinkage with potential damage to adjacent corneal stroma has been reported.127 Long-term effects of diathermy to the cornea are not yet clear. The process of corneal diathermy itself may be a stimulus for further CoNV by secondary release of proangiogenic factors.128 It would, therefore, be reasonable to try and minimise the application of FND to the cornea. Cursiefen et al. reported that arterioles tend to comprise less than 1% of CoNV. One option, therefore, would be to treat only the afferent vessel(s) of the CoNV complex.129 It has been shown that once the afferent vessel or vessels have been identified on angiography, it then makes their identification on slit-lamp biomicroscopy more evident and reliable. Whilst one third of patients required retreatment with FND, this may reflect identification of vessels on angiography that are not apparent on colour images.38,130
At present, angiographic guidance is the best option for planning FND, as targeting the afferent vessels is effective in reducing the area of CoNV, whilst minimising the amount of diathermy applied to the cornea. Antiangiogenic factors such as VEGF may be the better choice if lessmature or immature vessels predominate89, as identified from time to leakage on angiography.131
Possible future options
Current therapeutic options in the treatment of CoNV may be associated with numerous side effects and are unable to provide sustained benefits. The immune-privileged nature and accessibility of the cornea make it an attractive target for gene therapy, an alternative to pharmacological treatment that could provide non-toxic and long-term benefits. Additionally, progress of gene therapy treatment on the cornea can be monitored visually. To date, most studies of gene therapy for CoNV are still in the pre-clinical experimental stages, using trauma-induced CoNV on animal models. In the future, recently developed transgenic mouse models of CoNV may become the mainstream assay for angiogenesis research.132 Gene therapy seems to be effective in animal studies, though its clinical utility might be limited by safety issues arising from the vectors and overexpression of transgenes. A growing number of target genes have been identified and numerous gene-specific anti-angiogenic agents are under investigation in experimental settings, some with promising results.
VEGF pathway
The main target so far investigated has been the principal stimulus for angiogenesis, VEGF and its receptors. While some of these treatment approaches have already been discussed above, solid evidence for efficacy of others is still emerging.
Gene silencing by small interfering RNA (siRNA) allows effective control over posttranscriptional gene regulation. In particular, VEGF pathwayspecific siRNAs have showed a significant inhibitory effect on CoNV.133,134 Using a similar approach, a significant inhibitory effect on corneal haemand lymphangiogenesis was observed after subconjunctival injections of Prox1 siRNA in a rat model of alkali burn-induced inflammatory CoNV.135 The use of siRNA targeting hypoxia-inducible factor 1α (HIF-1α), a potent proximal proangiogenic regulator of corneal VEGF expression, has been reported in a mouse model of CoNV.136 Upregulation of some micro-RNAs (miRNAs) including miR-184137 and miR-132138 has been reported in rodent models of CoNV, also offering a potential new treatment target.
Everolimus is a derivative of rapamycin that inhibits human kinase mammalian target of rapamycin (mTOR) and indirectly decreases the expression of angiogenic growth factors such as VEGF, TGF-a and PDGF-b.139 It has been approved by the Food and Drug Administration (FDA) as a chemotherapeutic agent in the treatment of malignant solid tumours and as an immunosuppressant after renal cell transplantation.140,141 Sunitinib is a multi-target VEGF receptor tyrosine kinase inhibitor that targets VEGF and PDGF receptors and inhibits angiogenesis.142 Sunitinib has been approved for the treatment of kidney cancer, pancreatic neuroendocrine tumors and gastrointestinal stromal tumors by the FDA.143 Both agents have recently been shown to effectively reduce VEGFR-2 levels and inhibit CoNV in a rat model.144
Other target genes
An alternative approach to targeting VEGF and its receptors is to exploit the mechanisms the host itself has to limit the extent of VEGF induced angiogenesis. This mechanism uses the Robo4 (R4) receptor, a member of the axon guidance receptor family which is expressed on angiogenic endothelial cells.145,146 Upon interaction with its ligand, R4 generates a negative signal in the cell that diminishes downstream VEGF signaling. Local administration of soluble Robo4 to wild type herpes simplex virus-1 (HSV-1) infected mice with CoNV significantly diminished the extent of corneal angiogenesis.147
Recent investigations implicate netrins in the pathogenesis of CoNV.148,149 Netrins are secreted molecules involved in axon guidance and angiogenesis. Han and co-workers found that netrin-4 functions similarly as netrin-1 in angiogenesis, affecting human umbilical vein endothelial cell tube formation, viability and proliferation, apoptosis, migration, and invasion in vitro, and reversed corneal neovascularisation in vivo.150,151
Over the past few years, endostatin (ES) has drawn some attention for its ability to treat CoNV in the experimental setting.152 ES is an endogenous inhibitor of endothelial cell proliferation and angiogenesis, specifically inhibiting vegetative vascular endothelial cell growth. However, its feasibility is limited by a very short half-life due to chemical instability.153 Subconjunctival injection of polysulphated heparin (PSH)-modified ES with increased chemical stability was reported to significantly suppress CoNV with superior potency compared to ES.154 Similarly, Fc-endostatin (FcE) was developed by fusing endostatin to the Fc region of an IgG molecule, which significantly decreased corneal neovascularisation in a rabbit model of VEGF-induced neovascularisation.155
Tumor necrosis factor (TNF)-α has also been studied as a potential pharmacological target in the treatment of CoNV.156 TNF-α is a well-known pro-inflammatory cytokine, which acts on the vascular endothelium by promoting vasodilatation, edema, and leukocyte recruitment, all commonly associated with the development of CoNV. It has been demonstrated that infliximab, a recombinant human antibody to TNF-α is effective in reducing haemangiogenesis and lymphangiogenesis in different animal models of CoNV.157,158
The reduction of oxidative stress using H2-enriched irrigation solution has been suggested as an effective immediate measure to prevent CoNV in a mouse model for alkali burns.159 As a potential regulator of cellular reactive oxygen species (ROS) metabolism, the small GTPase RhoA and its downstream effector ROCK in the RhoA/ROCK signal pathway has recently been shown to play a critical role in angiogenesis by protecting endothelial cells from inflammatory damage via suppression of nuclear factor kappa B signaling160 and reduced ROS production.160–1 As a potent inhibitor of Rho-kinase, fasudil has been demonstrated to inhibit alkali burn-induced CoNV.162 Likewise, FK506 binding protein 12 (FK506BP), a small peptide with a single FK506BP domain that is involved in suppression of ROS and the immune response, significantly inhibited alkali burn-induced corneal inflammation in rats.163
To date, the only gene therapy option for corneal angiogenesis that has reached the clinical trial stage is Gene Signal-101 (GS-101), which is discussed above. With more target genes and biocompatible vectors being developed, more studies are needed to develop safe gene therapy that can not only prevent, but also regress on-going CoNV without the need for frequent and invasive administration.
Conclusion
CoNV is a common sequela in the setting of corneal pathology including inflammation, infection and trauma, amongst others. It has potentially devastating effects on vision due to secondary scarring and lipid deposition, as well as important implications on corneal transplant rejection and survival. Treatment options for CoNV are numerous, highlighting the limited efficacy and/or associated complications of the various different modalities. Recent advances in the assessment of CoNV have given new insights into our current knowledge of CoNV, and proven helpful in guiding and evaluating treatment. Furthermore, improved understanding of the pathogenesis of CoNV, along with animal and human studies of the various pharmacological treatments help guide the decision between medical and surgical management, often depending on the maturity of CoNV. Future options may include gene therapy, which may prove to be a non-invasive option with long-term benefits, although further studies are required to determine the safety and efficacy of these treatment options.







