Neurotrophic keratitis (NK), also referred to as neurotrophic keratopathy, is a rare disease characterized by trigeminal nerve dysfunction, leading to a reduction or loss of corneal sensation and subsequent degenerative changes in the cornea.1 Deterioration starts when spontaneous breakdown of epithelial cells and impaired surface-healing mechanisms increase susceptibility to corneal ulceration. Progressive thinning of these damaged corneal tissues heightens the risk of perforation.2 Clinically recognizing NK and halting its progression is crucial to preserve the integrity of the corneal surface, anterior segment transparency and, ultimately, visual function.3 This review discusses the underlying mechanisms of this corneal disorder, clinical diagnosis, current management approach and exciting prospective treatments on the horizon.
Corneal epithelium and innervation
A healthy cornea has no vascular supply and maintains complete transparency to facilitate clear vision.4 The cornea contains five sequential tissue layers with unique functions. The most superficial layer is a self-sustaining epithelium that protects the cornea from the external environment. The stroma underlies this and makes up most of the corneal structure, maintaining its shape, transparency and avascularity.3
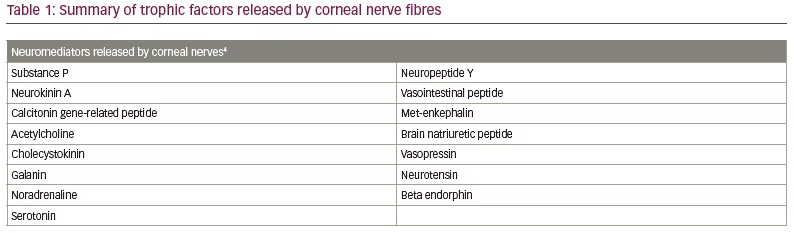
Multipotent cells at the corneoscleral limbus divide to produce both additional stem cells and differentiated epithelial cells, which migrate in centripetal fashion. These epithelial cells normally turn over at a rate sufficient to replace cells lost at the corneal surface.5 This constant replenishment is necessary to maintain proper corneal function. Trophic factors released by neighbouring corneal neurons fuels proliferation and healing of these cells (Table 1).2,4 In turn, squamous epithelial cells and corneal fibroblasts release neurotrophins and growth factors to induce the development and survival of corneal nerves.4
The cornea is extremely sensitive, stemming from its dense nerve supply. In fact, the cornea is the most richly innervated structure in the human body.4,6,7 Long ciliary and nasociliary neurons, derived from the ophthalmic division of the trigeminal ganglion, provide most of the sensory innervation to the cornea.8 These terminal nerve fibres are unmyelinated, preserving tissue transparency.4
The role of corneal nerves
Intact corneal nerve function is necessary to support ocular surface equilibrium. These nerve fibres elicit sensation to environmental stressors at the corneal surface, including chemicals, temperature and touch.4,6 External stimuli evoke protective responses, such as blinking and tear production, which provide additional nutrients and growth factors to promote healing.4,7 Diminished innervation of the cornea results in decreased tear production and a suppressed blinking reflex, which deprives the ocular surface of its natural trophic benefits. The loss of nerve function also results in poor stem-cell division at the corneoscleral limbus, which lowers the number of centrally migrating epithelial cells to replace those at the surface. This lack of appropriate turnover results in surface erosions that can progress to ulceration.9
Pathogenesis
Failure of normal nerve function is characteristic of NK. Crosstalk between the corneal epithelium and the neural network maintains a system of mutual trophic homeostasis. When nerve fibres are damaged, this hinders the release of neuromediators that help regenerate ocular surface cells. Thus, corneal nerve impairment disrupts epithelial healing and can leave the underlying stroma exposed. This increases susceptibility to stromal ulceration, melting and, ultimately, perforation of the cornea.5,9 Reduced trigeminal nerve sensation also decreases protective reflexes, including blink rate and production of tears.4
Aetiology
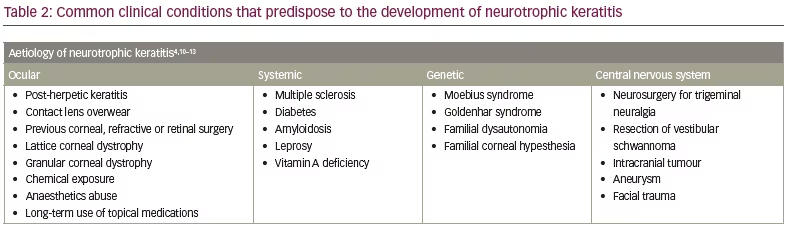
Any clinical condition that damages the trigeminal nerve at any point, from the nucleus to the terminal corneal fibres, predisposes patients to developing NK (Table 2).4,10–13 The most common conditions leading to corneal anaesthesia include herpes infections of the cornea and iatrogenic damage from neurosurgical procedures, such as for trigeminal neuralgia or resection of a vestibular schwannoma.10,11 Ocular conditions leading to NK include: overwearing contact lenses; previous corneal, refractive or retinal surgery; lattice and granular corneal dystrophies; chemical exposure; topical anaesthetic abuse; and long-term use of topical medications, such as non-steroidal anti-inflammatory drugs.1,10,12
Regarding keratorefractive surgery, NK is a rare occurrence following laser in situ keratomileusis (LASIK) and photorefractive keratectomy (PRK), but can arise from sensory nerve fibre injury during flap formation in LASIK and superficial ablation in PRK.14–16 Corneal surface innervation regenerates more slowly after LASIK compared with PRK, predisposing these patients to a longer window of impaired sensation and possible development of NK. Recovery of nerve density following LASIK may be expedited as future technology moves from mechanical microkeratome to laser-assisted flap creation.17
Other ophthalmic procedures have been linked to surface hypoesthesia, which may lead to NK. Corneal crosslinking, employed to treat keratoconus, causes a transient and significant reduction in corneal sensation.18 Certain corneal transplantation surgeries, including penetrating keratoplasty and deep anterior lamellar keratoplasty, are associated with a post-operative decrease in ocular aesthesia, while endothelial keratoplasties (Descemet stripping automated endothelial keratoplasty and Descemet membrane endothelial keratoplasty) are not.19,20 Associations have been reported between posterior-segment procedures and NK; these include surgery to repair retinal detachment and argon laser treatments.21,22
Systemic conditions can also lead to loss of corneal sensation, including multiple sclerosis, diabetes, amyloidosis, leprosy and vitamin A deficiency.1,10,12 In a patient with undiagnosed systemic disease, corneal ulceration due to NK can be the primary presenting complaint; therefore, a thorough clinical history is imperative to determine additional work-up.23 Cranial nerve palsy can occur secondary to compression from intracranial tumours or an aneurysm, facial trauma, or more rarely, congenital syndromes.12 While NK is rarer in children, when found, both congenital and acquired aetiology must be considered in the work-up. Congenital conditions that can cause corneal sensory deficit include Moebius syndrome, Goldenhar syndrome, familial dysautonomia and familial corneal hypesthesia.13 The inciting aetiology may influence the progression and prognosis of NK, depending on the severity of the corneal nerve impairment and the concomitant ocular surface integrity.10
Staging
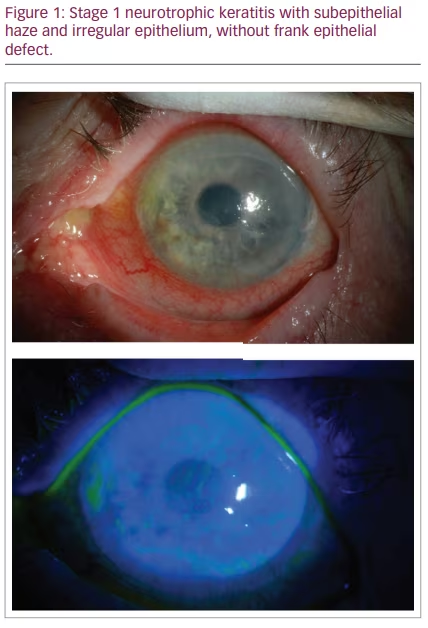
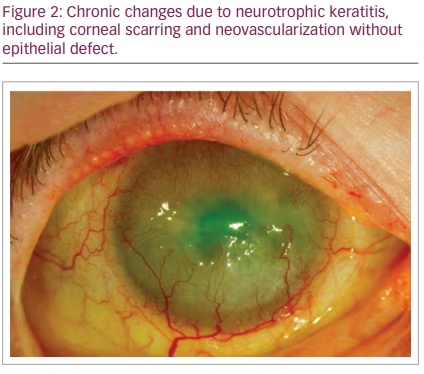
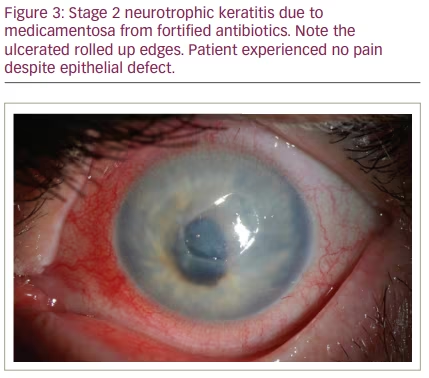
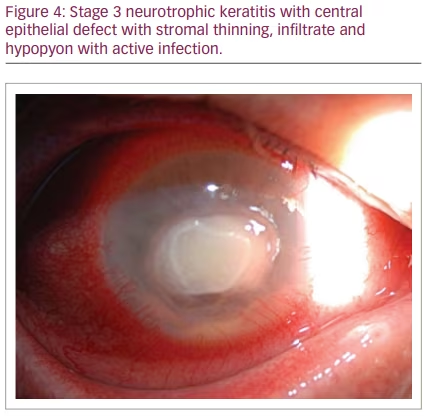
NK is classified by severity using Mackie’s classification system, proposed in 1995.24 Stage I NK is characterized by irregularity of the corneal epithelium without frank epithelial defect (Figure 1). These irregularities may present as corneal clouding, oedema, superficial neovascularization and punctate epitheliopathy with fluorescein staining (Figure 2).10,12 This stage is mild disease, with evidence of tear-film instability and reduced aesthesia in at least one corneal quadrant. Superficial damage to the inferior conjunctiva may also be discerned with lissamine green staining.5,10 Stage II, or moderate NK, is distinguished by recurrent or persistent epithelial defects without stromal involvement (Figure 3). These epithelial defects are classically round or oval shaped, with smooth rolled borders, and found most commonly in the superior corneal hemisphere.10 Rarely, this stage may be accompanied by anterior chamber inflammation.5,10 Finally, stage III NK is diagnosed when there is corneal ulceration with stromal involvement (Figure 4). In this severe stage of disease, there is stromal melting, which leads to corneal thinning and may prelude perforation.5,10,12
Diagnosis
NK is diagnosed based on the patient’s subjective history, symptoms, slit-lamp examination and corneal sensitivity tests. Corneal sensitivity must be evaluated to formally diagnose NK.5
Corneal nerve function can be tested qualitatively and quantitatively. In qualitative testing, a cotton wisp, strand of dental floss or tissue tip is used to stimulate the corneal surfaces bilaterally and the response in each eye is compared.5 Response is measured by whether the protective blink reflex is elicited. The quality of this response can be described along the range of absent to hypoesthesic to normal. Quantitative testing entails using a corneal aesthesiometer. With the Cochet-Bonnet aesthesiometer, mechanical contact is made at the corneal surface with various lengths of nylon filament. The filament is applied to the ocular surface and gentle pressure bends the filament. Greater trigeminal nerve function is evident when the patient acknowledges the filament at a longer length. The Belmonte non-contact gas aesthesiometer can also be used, which emits carbon dioxide at various flow rates and temperatures to gauge different aspects of corneal sensitivity. The severity of NK correlates with corneal nerve impairment upon testing. If the patient exhibits normal corneal sensitivity, NK is unlikely. However, if there is reduced response to ocular surface stimuli, further work-up is indicated.5
Management
It is imperative to recognize NK early and monitor it frequently to promote effective epithelial cell healing and prevent further corneal degeneration. As nerve damage decreases sensation, patients often remain asymptomatic despite NK worsening, and can progress from stage I to III quickly.10,25 The various approaches to treating NK can be broadly classified as topical medications, in-office interventions and surgical procedures (Table 3).8,10,26–30 Management in the past was stage based, depending on disease severity, with the goal of preventing further progression.25 Currently, there are more available treatments, irrespective of stage, and many new and emerging treatment options for NK.

Topical therapy encourages epithelial replenishment and healing to counteract any spontaneous surface breakdown.8 If there are recurrent epithelial defects or stromal involvement, topical antibiotics help prevent superimposed infection. Steroids may be used to combat coinciding inflammation; however, their use must be monitored as they impede healing of the epithelium and can increase the risk of perforation.10 Non-steroidal anti-inflammatory drugs should not be used, as they prevent proper epithelial healing.31 The cornerstone of topical treatment at all stages of NK is to maintain surface lubrication by frequently applying preservative-free artificial tears.8,10,25
Another topical therapy is blood-derived tears, which are formulated by centrifuging a sample of the patient’s blood and isolating the plasma or serum components.8 Plasma contains both clotting factors and platelet factors, while serum is the fluid remaining after clotting has occurred. These blood products contain high concentrations of nourishing factors, many of which are the same biologically active constituents of tears. These factors maintain epithelial cell health, promote healing, hinder proliferation of bacteria, and repress damage caused by inflammation and free radicals.32
Autologous derivatives that contain plasma rich in growth factors may be more potent than serum tears because they contain high concentrations of platelets.33 Platelet protein extract, isolated from α-granules, has high clinical efficacy for turnover of epithelial defects.34,35 Notable benefits of plasma rich in growth factors include its standardized production protocol and that it lacks pro-inflammatory cytokines, which serum tears do contain.33,34 Barriers to these treatment options include the logistical burden of a blood draw followed by preparation at a specialized facility, lack of insurance coverage, and refrigeration requirements for storage.34
A more recently developed topical drug with promising results is cenegermin (Oxervate™, Dompe US, Boston, MA, USA). This recombinant, human, nerve growth factor (rhNGF) promotes the ocular surface nerves to regenerate, improving corneal sensation, and heals epithelial defects.26,36 rhNGF is structurally identical to the naturally occurring neurotrophin produced in ocular tissues, which encourages neuronal growth and survival.37 rhNGF also binds lacrimal gland receptors to trigger the sensory-mediated tearing reflex, and stimulates epithelial cell differentiation and proliferation.8,38 Cenegermin is the first drug approved by the US Food and Drug Administration for treating NK.37,38 This recommendation was based on the conclusions of two pivotal trials; NGF0212/REPARO, performed in Europe, and NGF0214, performed in the USA. These studies showed that cenegermin is safe and effective in increasing rates of re-epithelialization in patients with NK associated with non-healing surface defects.26,36 Cenegermin is the first biological product approved for ophthalmic topical use and the first human neurotrophic factor to be implemented as drug therapy.27 If standard non-surgical interventions, including preservative-free artificial tears, therapeutic contact lenses and stopping toxic topical drugs, do not result in clinical improvement after 2 weeks, a provider may use cenegermin as a first-line regimen.36,38,39 A 2-month course of cenegermin remains clinically effective for as long as 48 months.40 Possible drawbacks to cenegermin include high cost, refrigeration requirements for storage and treatment burden, with multiple steps required to prepare individual dosage vials and application every 2 hours during the day for 8 weeks.8,27,38
A non-drug therapy is to wear contact lenses. Bandage soft contact lenses can be worn throughout the day and night, but must be removed periodically for sterilization or exchange.41 If treatment with bandage soft contact lenses is unsuccessful, scleral lenses can be trialled, as they offer a more long-term treatment option.42 Scleral lenses aim to boost epithelial healing of persistent defects and halt further thinning of underlying layers.8,10 With scleral lenses, a fluid reservoir vaults over the cornea, creating a protective hydrating layer between the lens and the ocular surface. This space can be used for sustained delivery of topical drugs, such as antibiotics and blood-derived tears.34,42 Potential disadvantages of scleral lenses include their high cost, the multiple appointments needed to obtain a custom-fitted lens, and the difficulty in applying and removing the large-diameter lens.34
An amniotic membrane can be applied in the office or operating room. A freeze-dried amniotic membrane or a cryopreserved amniotic membrane can be used to treat corneal ulceration. Both types of amniotic membrane resulted in equal levels of corneal healing approximately 30 days after application.43 For deep ulcers, surgical intervention may be employed to suture multiple layers of amniotic membrane over the defect. This method may be performed in conjunction with a contact lens or tarsorrhaphy.44 Results from the DREAM (Dry eye amniotic membrane) study support the association between amniotic membrane placement and regeneration of corneal surface epithelium.28 The amniotic membrane acts as a scaffold for re-epithelization and adds a layer of mechanical defence against the external environment while reducing inflammation and scar formation.45 Amniotic membrane tissue also contains high levels of nerve growth factor, which stimulates nerve growth and provides lasting improvements in corneal sensation.28 To sustain the beneficial effects of an amniotic membrane, the source of NK must be addressed.
Various surgical approaches can be used in refractory NK with stromal thinning. A simple procedure to shield surface defects while promoting healing is a tarsorrhaphy.46 Another method to achieve this is to induce interim ptosis by injecting botulinum toxin into the levator palpebrae superioris.47,48 Both techniques pose a cosmetic burden to the patient and compromises vision in the affected eye for the duration of treatment.10 These procedures must be considered carefully when treating children, as the sacrifice of visual stimulation can lead to developing amblyopia.49
Another surgical option is corneal neurotization, whereby an autograft or healthy-donor nerve segment is transferred to the denervated cornea to potentially re-establish sensory function. The nerve segment is often harvested from the supraorbital and supratrochlear branches of the frontal nerve, or less often, from the sural nerve.29,50 It can take weeks to months for the implanted nerve to integrate effectively and provide meaningful corneal sensation.49 This technique can promote re-epithelization and stabilize the ocular surface to allow for partial- or full-thickness corneal transplants in patients with compromised vision due to NK-induced corneal scarring.51
In addition to these treatments, there are multiple emerging approaches. The MYSTIC study of varenicline nasal spray showed promising results in improving tear-film production in patients with dry-eye disease.30 The efficacy of varenicline in improving the corneal surface in patients with NK is being investigated in the phase II Olympia study (Phase 2 clinical trial to evaluate OC-01 nasal spray in subjects with neurotrophic keratopathy; ClinicalTrials.gov identifier: NCT04957758).52 Wang et al. described complete healing of the corneal epithelium after 25 days of topical insulin (1 IU/mL, 2–3 times daily) in six patients with refractory NK.53 Tong et al. reported a case of bilateral refractory NK that showed substantial re-epithelialization 7 days after prescribing topical insulin (25 IU/mL, 6 times daily).54 Other promising therapies include thymosin beta-4 and oral nicergoline.55,56
Conclusion
NK is a rare corneal disorder but it may be more prevalent than recently assumed. When NK is suspected, objective measurement of corneal sensation is critical to establish a diagnosis. Patients must be monitored closely, as progression tends to be quick and asymptomatic. Although managing NK is challenging, the literature is expanding and provides growing evidence that will continue to guide treatment. Mainstays of treatment are protecting corneal integrity and promoting regeneration of ocular surface cells and nerve fibres. In the past, regimens were aimed at preventing further corneal damage, while new drugs show promise in targeting and reversing the root mechanism of the disease.







