Keratoconus early detection (screening) and diagnosis requires an in-deep corneal analysis with different techniques available.1 Slip lamp assessment and corneal topography/tomography are the most commonly accepted techniques in eye examination.
Corneal topography and corneal tomography are useful terms that distinguish between two different types of corneal examination, so both will coexist and be complementary.2 In fact, hybrid systems, combining Placido disk-based videokeratography and slit-scan images provide reliable corneal measurements in keratoconus assessment.3,4
The aim of this review is to provide general information about different corneal assessment technologies useful in keratoconus assessment; highlighting the different investigative techniques from its detection in the primary eye care clinic to definitive diagnosis, usually in the cornea specialist clinic. Information of most extensively available commercial devices, and the advantages and disadvantages of their use in keratoconus early detection and diagnosis, are described.
Method of literature search
We performed an extensive electronic search of the Medline and PubMed databases using individual and combinations of key words (keratoconus, keratoconus fustre, subclinical keratoconus, keratoconus treatment, keratoconus topography, keratoconus tomography, scheimpflug, keratoconus biomechanical properties, and keratoconus anterior OCT) in May 2016 to identify the relevant publications in this field. We included the references if they focused on assessment techniques of the cornea in keratoconus patients. We excluded techniques that are considered experimental, non-English publications and case reports.
Keratoconus
Keratoconus is a multifactorial disease with genetic, biochemical, biomechanical, and environmental pathophysiology,5 characterised by a thinning and steepening of the central and paracentral cornea, affecting approximately 1/2000 people in the general population.6–8 Commonly, this bilateral and asymmetric ectatic condition appears during the second decade of life and puberty and it progresses until the fourth decade of life, causing high myopia and irregular astigmatism.5–8 Keratoconus patient management requires a multi-professional approach for early detection, correct diagnosis, follow up, monitoring and adequate management that involve: primary eye care practitioners, optometrists, contact lens (CL) practitioners and ophthalmologists with the last aim to provide better care and improve patients’ quality of life.9,10
Keratoconus detection, diagnosis and classification
Keratoconus diagnosis is a challenge.5,7 Early stages of keratoconus, where clinical signs are not manifest on biomicroscopy (stromal thinning, conical protrusion, Fleischer corneal epithelial iron ring, Munson sign, Rizzuti sign, or Vogt striae)7,8 but the cornea demonstrates subtle topographic features comparable to those of clinical keratoconus receive the name of fruste keratoconus, subclinical keratoconus or keratoconus suspect.3,11–13 Therefore, distinguishing between healthy cornea and early keratoconus (in opposition to moderate or advanced stages), of subclinical keratoconus or other ectatic diseases imposes greater diagnostic challenge.5,7 It is of paramount clinical importance in primary eye care and in screening refractive surgical patients to avoid iatrogenic corneal ectasia after laser surgery.14–16
Clinical keratoconus is reliably detected with Placido disk-based corneal topography and even sometimes at slit-lamp examination.16 Other technologies, such as: corneal tomography (Scheimpflug or dual Scheimpflug devices),3,13,17 anterior segment optical coherence tomography (AS-OCT),18,19 biomechanical devices20,21 that analyse the anterior and posterior corneal surface, full corneal thickness map, epithelial mapping, or corneal biomechanical properties are necessary to complete keratoconus diagnosis.5,22
Currently, there is no clinically accepted classification allowing eyecare practitioners to clearly differentiate between healthy and keratoconus cornea (especially in early stages), and that could be used in patients’ follow-up in suspect (or diagnosed) cases. The most common classifications were the Amsler-Krumeich,23 and Collaborative Longitudinal Evaluation of Keratoconus (CLEK)24 classifications. However, both classifications fail to address current information and technological advances5 and a new classification criterion is necessary.
The Amsler-Krumeich classification proposes four different stages using refractive, topographic and biomicroscopic corneal signs. The CLEK classification proposes to use the average corneal power and root mean square (RMS) error for higher-order Zernike terms (derived from the first corneal surface wavefront) combined with clinical biomicroscopic signs. Because larger values of vertical coma have been found in keratoconic corneas, high-order corneal aberration analysis could play a relevant role in future keratoconus classification24–28 Therefore, future keratoconus classifications will be directly dependent on the accuracy and reliability of the corneal device used in patient assessment.27,29
Corneal topography
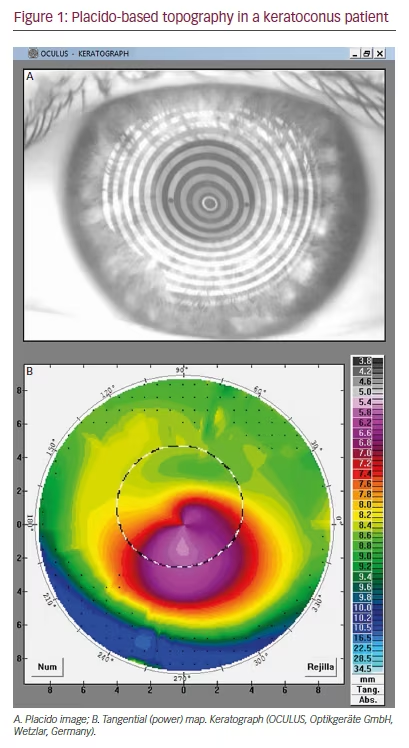
Corneal topography is a method of computer-assisted, non-invasive examination of the anterior surface of the cornea. It provides a qualitative and quantitative description of the morphology of the cornea in a topographical map after analysing the reflected image of illuminated rings (Placido disk) onto the corneal surface (Figure 1).30
Corneal topography was introduced in the mid-1980s with the developing of different algorithms to analyse the Placido photokeratoscope’s images, and has represented a true revolution in the diagnosis and management of corneal disease,2 including keratoconus. Nowadays, corneal topographers are one of the most extensively used devices in clinical practice.26,29
In fact, several mathematical indices have been developed with the aim of helping with keratoconus detection, grading the disease and monitoring its progression.26,31 For example; central keratometry (K-value)32 with different cut-off values to keratoconus suspect (>47.2 D); inferiorsuperior asymmetry (I-S value)32 with a cut-off value of 1.4 D difference between average inferior and superior corneal powers at 3 mm from the centre of the cornea; the steepest radial axes (SRAX)32 calculated with the angle between the steepest superior meridian and steepest inferior meridian; surface asymmetry index (SAI)33,34; keratoconus severity score (KSS)24 calculated with some corneal topography features (axial pattern, average corneal power and higher-order RMS) and slit-lamp signs (including scarring); keratoconus prediction index (KPI)35 calculated after a discriminant analysis of eight quantitative videokeratography indices (Simulated K1, Simulated K2, opposite sector index [OSI], centre/surround index [CSI], differential sector index [DSI], SAI, irregular astigmatism index [IAI] and analysed area [AA]); keratoconus percentage index (KISA%)36 based on K-value, I-S value, keratometric astigmatism (AST), and SRAX indices; or cone location and magnitude index (CLMI),37 calculated with the axial and tangential curvature data.
However, most of these indices depend on the topography software, but with sensitivity and specificity controversial, they may be difficult to understand38 and have not been extensively used in clinical practice.
Therefore, new criteria, easy to use and non-device dependent methods would be necessary to improve keratoconus detection, diagnosis and classification.27,29
Gas permeable (GP) CL fitting is the primary keratoconus management option.6–8,39 However, fitting of GP lenses in keratoconus patients is challenging because the irregular cornea often requires several diagnostic lenses to achieve a final acceptable GP lens fit, which prolongs practitioner and patient chair time.40–45 However, GP CL fitting could be improved with different CL fitting software that analyses Placido-based corneal topography curvature data to propose the lens parameters, mainly base optic zone radius and lens diameter.40,41,46–49 Using these software could decrease the number of diagnostic lenses necessary to achieve an acceptable CL fit and reduce the chair time in keratoconus patients.10,40,41,46
Although, corneal topography is probably the most commonly used tool for the diagnosis of keratoconus, it is accepted that this technique may lead to false negatives in the subclinical phase. That means that Placidobased videokeratographers cannot identify very mild forms of keratoconus (fruste keratoconus) that would require to be identified, assessing corneal thickness and anterior/posterior curvature measurements over the entire cornea provided with corneal tomography.22,50 Standard corneal topography could be an acceptable technique in primary care but not in speciality clinics or in screening refractive surgical patients,14–16 where a complete diagnosis is necessary and complete corneal assessment with corneal tomography.
Corneal tomography
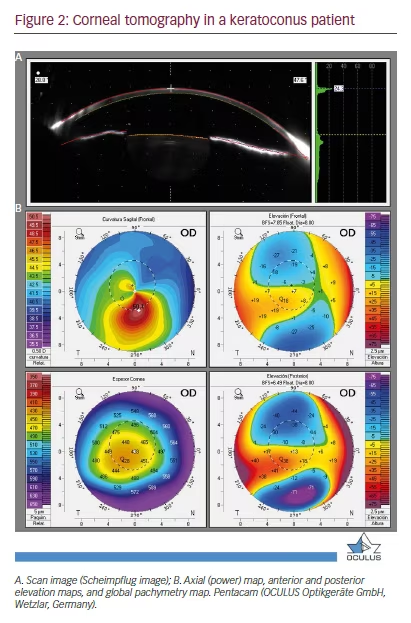
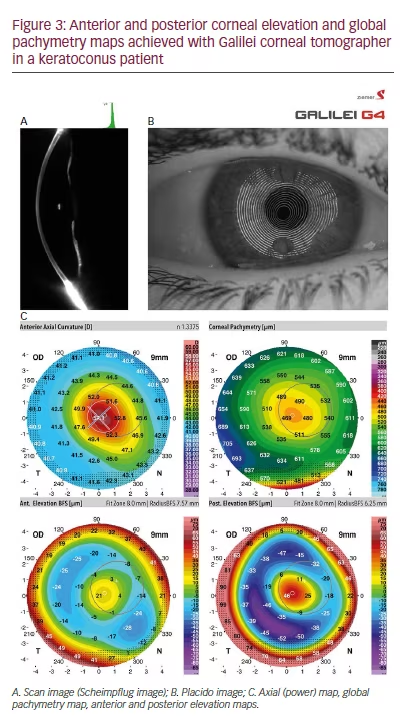
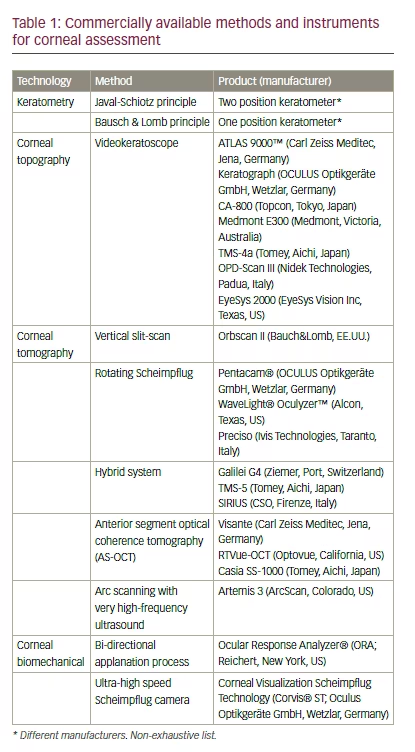
The term computed tomography is classically used in medicine, for referring to the radiographic technique for imaging a section of an internal solid organ, producing a three-dimensional image. Corneal tomography allows three-dimensional characterisation of the cornea (Figure 2) after anterior and posterior corneal surface analysis, using different slit-imaging technologies,2,51,52 such as vertical slit scanning, 53,54 rotational Scheimpflug imaging,55 arc scanning with very high-frequency ultrasound, and optical coherence tomography.56 The first device that permitted imaging of the anterior and posterior corneal surfaces, was the Orbscan II (Bausch + Lomb, New York, US).53,54 This has since been replaced by rotational Scheimpflug devices, such as: Pentacam® (OCULUS Optikgeräte GmbH, Wetzlar Germany),57 WaveLight® Oculyzer™ (Alcon, Texas, US)56 and Preciso (Ivis Technologies, Taranto, Italy). Finally, other devices combine Placido-based topography with slit-image analysis and are collectively named hybrid systems, hybrid topographers or dual Scheimpflug-Placido tomographers. Highlight Galilei G4 (Ziemer, Port, Switzerland),58 TMS-5 (Tomey, Aichi, Japan) and SIRIUS (CSO, Firenze, Italy)59 are examples of these devices. In summary, corneal tomography defines the spatial relationship between the anterior and posterior corneal surfaces and provides a global thickness corneal map (Figure 3).
Corneal tomography has been recognised as a critical diagnostic component in keratoconus patients’ assessment,5 helping in diagnosis (differentiating between fustre and clinical keratoconus) and monitoring progression, because it is necessary to confirm changes on the posterior corneal surface and corneal thickness alteration in order to diagnose (and monitor) keratoconus.5,13,50,60,61
Anterior segment optical coherence tomography
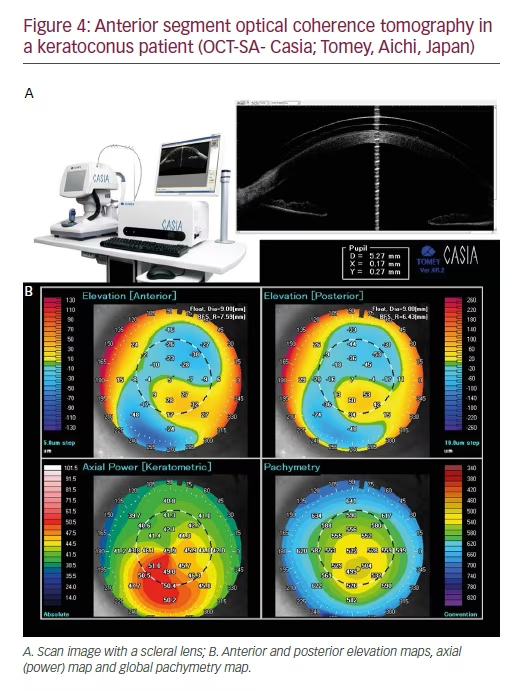
The first report of AS-OCT imaging appeared in 1994.62 OCT compares the time delay of infrared light (1310 nm) reflected from the anterior segment structures against a reference reflection, achieving a high resolution cross-sectional image of the anterior segment of the eye (from 2 to 20 ƒÊm).63
There are three commercial AS-OCT devices; Visante (Carl Zeiss Meditec, Jena, Germany), RTVue-OCT (Optovue, California, US) and Casia SS- 1000 (Tomey, Aichi, Japan).64,65 AS-OCT has been proposed to assess keratoconus patients (Figure 4);66 helping to investigate corneal thickness asymmetry,67 epithelial thickness-distribution characteristics68 and monitoring progression.19 AS-OCT could be a promise tool in keratoconus diagnosis (helping to differentiate between fustre and clinical forms of keratoconus of healthy corneas).
Biomechanical devices
Keratoconus corneas show abnormalities in biomechanical response when they are compared to normal corneas in ex-vivo studies.69,70 However, in-vivo measurement of corneal biomechanics remains a difficult task, and just two commercially available instruments have been proposed;1,71,72 the Ocular Response AnalyzerR (ORA; Reichert, New York, US) and the Corneal Visualization Scheimpflug Technology (CorvisR ST; Oculus Optikgerate GmbH, Wetzlar, Germany).
The ORA employs a dynamic bi-directional applanation process with an air-pulse, similar to that of traditional air-puff tonometers.1,71–73 The Corvis ST is a non-contact tonometer with a dual Scheimpflug, high-speed camera that takes more than 4,300 frames per second of the horizontal meridian of the cornea, and captures approximately 140 cross-sectional images of the cornea during the air-puff induced deformation.1,71,72,74 Both devices determine different corneal biomechanical metrics, mainly corneal hysteresis (CH) and corneal resistance factor (CRF) with ORA -including 37 parameters that describe the waveform of the applanation signal- and deformation amplitude respectively with CorVis. Intraocular pressure (IOP) and IOP value corrected with corneal thickness value is also provided.
Corneal biomechanical metrics are statistically significant between keratoconic and healthy corneas using both ORA71,75–77 (keratoconus showed lower CH and CRF value) and Corvis71,78–80 (keratoconus showed higher deformation amplitude), which could be useful to detect subclinical keratoconus.81
Unfortunately, data provide for these devices have not proven to be a definitive keratoconus diagnostic value (able to differentiate between keratoconus, forme fruste and normal eyes)71 because a substantial overlap exists with normal corneas,82,83 and further research is necessary to obtain valid cut-off values to use in combination with more clinical data.20,72,75,77,80,84,85 In summary, further clinical validation is necessary to inderstand the meaning of these biomechanical parameters obtainied with ORA and Corvis before they can be used in clinical practice.71
Other technologies or devices have been proposed to measure corneal biomechanical properties, for example: acoustic radiation force (ARF),86 applanation resonance tonometry (ART),87 confocal microscopy,88 optical coherence elastography,89 scanning acoustic microscopy (SAM),90 supersonic shear wave imaging (SSI)91 that must be validated in human.71
Discussion
Keratoconus early detection, diagnosis and classification are a challenge.5 Both early detection and final diagnosis require a complete eye exam and in-depth corneal assessment using different technologies, such as: corneal topography, corneal tomography, corneal biomechanics and others. However, it is necessary to differentiate between early detection of keratoconus and the final or definitive diagnosis. Early detection is of paramount importance in primary eye care, when a definitive diagnosis is not always required and referral to cornea specialist is necessary to conduct final keratoconus diagnosis. In fact, both procedures are slightly different and should follow different criteria.
Unfortunately, primary eye care service is not easy to define,92 but there is a reasonable consensus accepting that primary care is the provision of first contact care for ophthalmic conditions and the follow up, preventive, and rehabilitative care of selected eye conditions,92 in contrast with secondary or referral specialist services.93 Related with keratoconus screening and diagnosis, in primary eye care, one of the most important purposes should be the detection potential keratoconus indicator (mainly related with patient’s corneal shape) in a large population generally asymptomatic or with unspecific symptoms. This practice requires the use of cheap techniques accepted by patients and eye care practitioners with a reasonable sensitivity and specificity. The most commonly used in these clinics are Placido-based corneal topography. In future, if the price of corneal tomographers is reduced, these devices could be introduced in primary eye care clinics. A survey to optometrists in Australia showed that near of 45% of practitioners have a corneal topographic unit.94 This percentage is lower in United Kingdom (26%) and higher in Spain (60%) (Author unpublished data).
Primary eye care practitioners play a relevant role in early detection of eye disorders and pathologies.92,95–97 Keratoconus could be suspected in risk patients (Down syndrome, relatives of affected patients, ocular allergy, Asian or Arabian ethnicity, eye rubbing, floppy eyelid syndrome, atopy, connective tissue disorders [Marfan syndrome] and others)5,98,99 or when certain clinical signs are found in the eye exam, such as: scissors reflex during retinoscopy exam, “oil-droplet” reflex (Charleux sign), change in astigmatism refraction (in axe or power) or myopia increase with asymmetry between both eyes.7,100–103
In opposition, a definitive diagnose is usually done in specialist clinics, for example in cornea units, refractive surgery, etc. This practice requires establish keratoconus presence using the necessary techniques, which may be expensive but justifiable, such as: corneal tomography, AS-OCT and others devices that allow the characterisation of corneal biomechanical properties.
Conclusions
In summary, corneal topography plays a significant role in keratoconus detection in primary eye care, because anterior Placido-based corneal topographers are cheaper devices with great utility in keratoconus management (fitting GP CL) and follow up. However, a definitive keratoconus diagnosis requires anterior and posterior corneal assessment (with corneal tomography and other techniques) and global pachymetry investigation able to distinguish between healthy cornea, fustre keratoconus and keratoconus. So, corneal tomography is compulsory to conduct a definitive diagnosis or in refractive surgery patients screening.







