The main risk factor for glaucoma damage is an intraocular pressure (IOP) above an undeterminable threshold value for an individual patient. The IOP is not constant but is subject to multiple physiological and pathological fluctuations. These can vary in magnitude and duration, for example, the pulsation of retinal vessels, Valsalva manoeuvre, changes from upright to supine position, and circadian rhythm. The reference standard to determine IOP is Goldmann applanation tonometry (GAT), which is usually performed only once and seldom multiple times during office hours. This only gives information of a few seconds out of 86,000 seconds in a day, and as such decreases the value of information gained. Measurements in sleep laboratories are cumbersome. In addition to this there is still critical discussion about weather fluctuations pose an additional independent risk factor for glaucoma progression.1,2
Multiple IOP measurements take time and the repeated topical anaesthetic used with GAT may damage the cornea. An alternative would be multiple or alternating IOP measurements with non-contact or other tonometers, which however are not the reference standard. Similar to noninvasive 24-hour electrocardiogram (continuous) or blood pressure measurements (intermittent), continuous IOP measurement would perhaps provide additional important information. This idea was seemingly realised with the contact lens sensor (CLS) Triggerfish® (SENSIMED AG, Lausanne, Switzerland). This medical device received CE certification in 2008 and American Food and Drug Administration (FDA) approval in 2016.
The review describes the current state of knowledge about this medical device, especially discussing the question whether an application in daily clinical or office use for diagnosing and therapeutic monitoring of glaucoma patients is necessary and/or recommended.
Contact lens sensor Triggerfish – product description
The theory published by Leonardi et al., was, that IOP could be measured indirectly by a strain gauge embedded in a contact lens.3 The basis for this theory was that the corneal curvature in the limbal area (diameter of the strain gauge at 11.5 mm) changed with variation in IOP, and that this could be measured. Two circular strain gauges made from platinum-titanium (7 μm) and a loop antenna made from gold (30 μm) for data transfer and a microchip (50 μm) for wireless data management were embedded in a silicone contact lens (diameter 14.2 mm), through a complicated manufacturing process. For better patient tolerance it was coated with an oxygen plasma. Power is supplied by the recorder and an antenna which is stuck around the orbit. Measurements are taken at 5 minute intervals for 30 seconds, within these 30 seconds 10 times with 300 datasets. The gained data are transferred wirelessly via this antenna to a recorder which is worn around the waist. During the 24-hour measurement period spectacles with metal frames must be avoided, but all other normal daily activities should be performed and documented in a diary. This is with the exception of activities that include moisture and water (e.g. swimming, showering etc) and
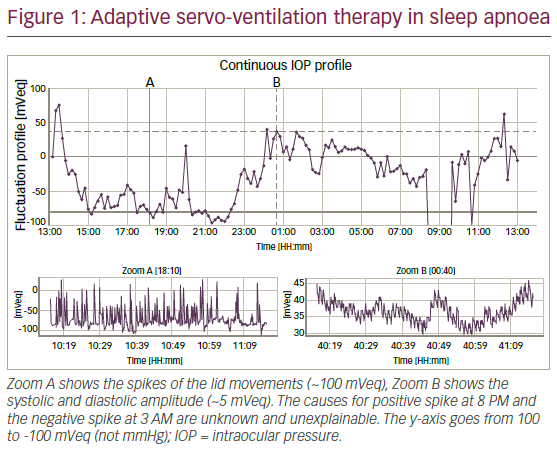
excessive body movements (sport). Moisture/water would be a safety issue and excessive movement may break the wire contacts, which leads to information loss. The result is a 24-hour profile consisting of 288 individual measurements, which can be viewed in detail (Figure 1, Zoom A, Zoom B). The x-axis provides the time, the y-axis initially provided an arbitrary unit measurement, which was replaced by a new software to milliVolt (mV) equivalent ([mVeq] not mmHg) (Figure 1). High spikes (usually around 100 mVeq) caused by blinking are filtered from the noise signals. The CLS can be used only once.
In-vitro studies
Leonardi et al. used enucleated pigs’ eyes and applied a prototype of the CLS, fluctuated the pressure by 10 mm Hg every 50 seconds, which showed a curve that corresponded well with manometric measurements.3 Subsequently Leonardi et al. fluctuated the pressure every 4 seconds between 11 and 14 mmHg, simulating a typical ocular pulse amplitude of 3 mmHg.4 Additionally the IOP was increased by 1 mmHg every 10 seconds (starting at 19 mmHg up to 30 mmHg and back to 19). The linear profiles gained by the CLS once again correlated well with the manometric pressure profiles. The mean change per mmHg was 113 μV. In all of the initial experiments the pressure was only changed within seconds, never recorded longer than 1 minute.
Faschinger et al. repeated these experiments with a human eyeball and much longer intervals (30 minutes) between stepwise (5 mmHg) increases of IOP from 10 to 30 mmHg.5 They could not find a correlation between the manometric and CLS profiles. The reason for this could not be determined. It is possible, that the strain gauges of the CLS respond better to short inputs than to longer gradual changes.
First applications in humans
Leonardi et al. successfully recorded the IOP, additionally the artificial increase and decrease of the IOP and ocular pulsations in five healthy subjects.6 After gaining CE certification in 2008, the CLS was provided to various clinics for clinical testing of a “24-hour monitoring of IOP”, looking predominantly at safety and tolerability during the 24-hour period. Both parameters were described as good.7,8 In rare cases the CLS had to be removed prematurely or caused more marked corneal epithelial changes.9 The main adverse events were blurred vision (82%), conjunctival hyperemia (80%) and superficial punctate keratitis (15%).10
The subsequent publications involved patients with various types of glaucoma, such as open-angle glaucoma10 and normal tension glaucoma.11 Surprisingly, the gained CLS profiles lead to therapy changes in two-thirds of patients.9 Patients with normal tension glaucoma11 were monitored with the CLS off and on therapy. On the one hand these showed very unique profiles, on the other, three out of five patients showed CLS values that were generally higher in the treated session than in the control session, while IOP levels were lower in all patients in the treated session. Hollo et al. also found no correlation between the CLS profile and the GAT in patients with ocular hypertension and openangle glaucoma, who were investigated without therapy and later with a prostaglandin analogue.12
Reproducibility and pressure patterns in 24-hour profile
Investigations at a 1-week intervals showed fair to good reproducibility of the CLS profile.10 In 77.2% of patients (glaucoma suspects or established glaucoma) two typical patterns of the CLS profile were found: the majority (62.9%) had a sinus curve with a peak at night between 01:00 am and 06:00 am (acrophase), while 14.3% showed a flat curve. In 17.1% the acrophases did not show much correlation between sessions. The acrophase is ascribed to the supine position during sleep. A weakness of this study was the inclusion of patients on and off therapy, because the therapy might have had an influence on the profile.13 Mottet et al. examined 12 healthy subjects in a sleep laboratory repeatedly with the CLS (on three different days) and hourly non-contact tonometry (NCT, on four different days) to evaluate the rhythm reproducibility and symmetry between the eyes.14 The reproducibility between CLS and NCT showed poor to fair correlation. These results suggest that the difference obtained between the different techniques were related to technical methods. Liu et al. examined 30 subjects with healthy eyes to determine whether the time of the acrophase measured either with CLS or pneumotonometer (PT; every 2 hours, sitting while awake and supine while asleep) correlates.15 They showed synchronicity and no significant difference between the two measurement methods: with CLS the peak of the acrophase was at 04:44 am (±210 min), with the PT it was at 04:11 am (±120 min). The linear regression analysis showed no correlation of the variations (“amplitude”, i.e. half distance between the cosinor-fit maximum and minimum) between the two methods (CLS 99.8 ± 65.5 mVeq versus PT 3.1 ± 1.4 mmHg). Realini et al. demonstrated that the repeatability of measurements in IOP is not a given, since the IOP of healthy individuals did not manifest a sustained and reproducible diurnal pattern when measured by GAT.16 In this study, however, measurements were only taken from 8:00 am to 8:00 pm. They found an overall agreement for absolute IOP values of fair to good, but the overall agreement of IOP change was uniformly poor.
Validity
Measurement methods have a high validity when their results correlate with empirical measurement results. For the CLS this would mean that its values would correlate with those of GAT (or manometric measurements). Since the CLS, however, does not measure mm of Hg, but rather an electronic value given in mVeq this comparison is not possible. The CLS profile, however, should behave similar to the IOP profile, taken by GAT, e.g. during physiological increase of IOP due to changes in head position. The wake-sleep-slope is attributed to this change of the head position.13 A comparison of the CLS profile with an IOP profile of the fellow eye (measured with PT) showed a very good correlation of values (correlation coefficient R2+0.914).17 Other working groups could not demonstrate this correlation. Faschinger et al. found no correlation between CLS profiles and GAT profiles in the fellow eye taken in upright, supine and head-down positions, even after repeated measurements.18,19 Similar experiments were performed by Sunaric-Megevand et al., where IOP was artificially increased with a ring and acquired by CLS and GAT profiles.20 The correlation coefficient was statistically not significantly different from zero. A multicentre, randomised, clinical study including 59 patients showed CLS readings did not correlate, and was no predictor of change in IOP as measured either GAT or iCare pro.21
Possible explanation of discrepancies between GAT and CLS profiles
The CLS results are based on the expansion of the strain gauges, changing their resistance, and on the assumption that the IOP might change the corneal curvature at the corneoscleral junction (11.5 mm diameter). This was reported by Greene and Gilman in experiments with rabbits and strain gauges in contact lenses in 1974, showing a resistance change of 0.08 Ohm per mmHg IOP change.22 Epithelial side strain, epithelial thickness and central corneal radius of curvature during changes of IOP were measured using digital imaging processing in human enucleated globes.23 Central corneal radius of curvature during an IOP increase from 2 to 22 mmHg changed by 0.05 mm (= 2.5 μm per mmHg) and with a further IOP increase from 22 to 100 mmHg by 0.08 mm (= 1.0 μm per mmHg, not linear). A similar value of 3.07 μm per mmHg was described by Lam and Douthwaite 24 during an IOP change of 6.5 mmHg in a headdown posture (30 degrees). Both publications measured the changes of the central corneal curvature, but the strain gauge of the CLS lies in the periphery of the cornea. Finite element models could provide more exact measurements of curvature changes in the corneal periphery. After all, this is the meeting point of two different tissues (sclera and cornea) with different thickness, elasticity, rigidity and hydration. Since the IOP is not directional power but rather a scalar one, the location (central or peripheral) of the measurement should make no difference in identical tissues. However, a difference in two different tissues is expected. In summary, the CLS provides values based on electrical signals that, despite being related to IOP, do not measure the same phenomenon.27
Investigations with a PT in one eye and a CLS in the fellow eye showed good correlation of respective profiles,17 although less pronounced. Again, the reason might be that the two device units (mmHg and mVeq) are not directly comparable.
Artifacts
Leonardi et al. listed factors that could have a possible influence on the CLS results: movements of the eye, blinking of the eyelids, movement of the contact lens, corneal thickness, changes in stromal hydration throughout the day and night, biomechanics, keratometry, corneal diameter, axial length, scleral thickness and rigidity.4 Hollo et al. found an increasing trend to the wearing time in all CLS profiles.12 De Smedt et al.8 and Mansouri et al.9 described a characteristic short-term signal drop correlating well with the exposure to sunlight. Faschinger et al. investigated the influence of light on the CLS profile. Absolute darkness showed no change in signal power; however, light showed slight changes.26 There was, however, no correlation between light intensity and the amplitude of the fluctuation. It is possible that these variations were caused by temperature changes due to the light source and room temperature. The role of temperature on the CLS signals was demonstrated by Faschinger et al., showing that an increase in temperature lead to a decrease of the signal.27 Strain gauges out of metal possess electrical and thermal conductivities that are interdependent. In principle, changes in temperature should be neutralised by the construction of the strain gauges (Wheatstone bridge) and by a second passive strain gauge (as they are in the CLS). Another factor to take into account is that silicone is a good insulator, but the thickness of the CLS, where the strain gauge is positioned, is far from the centre and might therefore be influenced by temperature changes.
Contact lens wear might limit the oxygen concentration in the cornea, leading to oedema. In a study of 20 patients the thickness of the central and peripheral cornea (approximately 6 mm from pupil centre) were measured. During a 14-hour wear period (pre-to-post sleep, mean duration 9.9 ± 0.7 hours) the cornea remained statistically unchanged when compared to the fellow eye (ultrasound pachymetry and Pentacam). In the eye with the CLS, the thickness changed from 523 μm pre study to 537 μm post study (swelling of 14 μm).28 No statistical differences were found in the horizontal and vertical radius of the cornea in either eye. The study by Tojo et al. showed a similar increase from 526 μm ± 29 to 533 ± 27 μm.29 Hubanova et al. demonstrated a mean maximal increase of 24.3 μm ± 9.4 (measured with the Visante Anterior Segment Optical Coherence Tomography [Carl Zeiss Meditec, Inc., Dubline, California, US]).30 In another study the thickness decreased from 560.3 μm before application of the CLS to 548.0 μm after 24-hour wear (statistically significant).15,17 The use of the CLS and correlations to the biomechanics properties of the cornea have not been published yet. The result of the CLS profile should not be influenced by open eyelids during wake and closed eyelids during sleep.17
Subtle analyses of the Triggerfish profile
De Moraes et al. investigated 40 patients with treated open-angle glaucoma to determine if there was a correlation between the CLS profile and a progression rate of the visual field loss.25 The patients were classified as either fast, slow or minimal progressors. The authors defined six main outcome measures: number of large peaks (peaks with a height of 90 mVeq or more), mean peak ratio (mean peak height to time-to-peak), wake-to-sleep slope (1 hour before and 1 hour after the time the patient went to sleep), amplitude of the cosine curve (overall estimate of the magnitude of the signal oscillation), variability of the mean (amount of fluctuations) and area under the receiver operating characteristic curve (reflects the magnitude of the signals and the duration at which they remained above a reference line). Several parameters were associated significantly with faster progression (e.g. number of large peaks while awake, mean peak ratio while asleep, wake-to-slept slope, etc). There are several uncertainties in the study: it was not mentioned what kind of topical therapy the patients were receiving (beta blocker in the morning? or prostaglandin analogue in the evening?), that might have influenced the pattern of the CLS profile; it was not mentioned, if any of the patients had cataract surgery during the study period that might have improved the mean defect (MD)-value; in the graph showing the average CLS output profile the starting values differed in a large extent from the endpoint, without an explanation (start and end point should be at the same level); the much higher standard deviation at the end of the 24-hour profile was not explained; the value of 90 mVeq or more for a “large” peak was chosen without an explanation (maybe because the standard deviation was about 90 mVeq?). This arbitrary threshold demonstrated a mean peak amount of only 0.4 (during sleep) and 1.8 (during wake [within 24 hours]).
Tojo et al. described “fluctuations” of the IOP in 11 eyes with pseudoexfoliation syndrome and 11 normal eyes and found, that the range of fluctuations was significantly higher in the pseudoexfoliation group.29 All healthy eyes had their maximum value during the nocturnal period, but only 7 out of 11 eyes with pseudoexfoliation. In this study, fluctuations were defined as the difference between the maximum and minimum value (in mVeq) during the course of 24 hours, and the mean was 294 mVeq in healthy eyes versus 522 in pseudoexfoliation eyes. Additionally, the fluctuations of the diurnal (mean 270 versus 477) and nocturnal (mean 179 versus 272) period were interpreted separately.Possible explanation of discrepancies between GAT and CLS profiles The CLS results are based on the expansion of the strain gauges, changing their resistance, and on the assumption that the IOP might change the corneal curvature at the corneoscleral junction (11.5 mm diameter). This was reported by Greene and Gilman in experiments with rabbits and strain gauges in contact lenses in 1974, showing a resistance change of 0.08 Ohm per mmHg IOP change.22 Epithelial side strain, epithelial thickness and central corneal radius of curvature during changes of IOP were measured using digital imaging processing in human enucleated globes.23 Central corneal radius of curvature during an IOP increase from 2 to 22 mmHg changed by 0.05 mm (= 2.5 μm per mmHg) and with a further IOP increase from 22 to 100 mmHg by 0.08 mm (= 1.0 μm per mmHg, not linear). A similar value of 3.07 μm per mmHg was described by Lam and Douthwaite 24 during an IOP change of 6.5 mmHg in a headdown posture (30 degrees). Both publications measured the changes of the central corneal curvature, but the strain gauge of the CLS lies in the periphery of the cornea. Finite element models could provide more exact measurements of curvature changes in the corneal periphery. After all, this is the meeting point of two different tissues (sclera and cornea) with different thickness, elasticity, rigidity and hydration. Since the IOP is not directional power but rather a scalar one, the location (central or peripheral) of the measurement should make no difference in identical tissues. However, a difference in two different tissues is expected. In summary, the CLS provides values based on electrical signals that, despite being related to IOP, do not measure the same phenomenon.27 Investigations with a PT in one eye and a CLS in the fellow eye showed good correlation of respective profiles,17 although less pronounced. Again, the reason might be that the two device units (mmHg and mVeq) are not directly comparable. Artifacts Leonardi et al. listed factors that could have a possible influence on the CLS results: movements of the eye, blinking of the eyelids, movement of the contact lens, corneal thickness, changes in stromal hydration throughout the day and night, biomechanics, keratometry, corneal diameter, axial length, scleral thickness and rigidity.4 Hollo et al. found an increasing trend to the wearing time in all CLS profiles.12 De Smedt et al.8 and Mansouri et al.9 described a characteristic short-term signal drop correlating well with the exposure to sunlight. Faschinger et al. investigated the influence of light on the CLS profile. Absolute darkness showed no change in signal power; however, light showed slight changes.26 There was, however, no correlation between light intensity and the amplitude of the fluctuation. It is possible that these variations were caused by temperature changes due to the light source and room temperature. The role of temperature on the CLS signals was demonstrated by Faschinger et al., showing that an increase in temperature lead to a decrease of the signal.27 Strain gauges out of metal possess electrical and thermal conductivities that are interdependent. In principle, changes in temperature should be neutralised by the construction of the strain gauges (Wheatstone bridge) and by a second passive strain gauge (as they are in the CLS). Another factor to take into account is that silicone is a good insulator, but the thickness of the CLS, where the strain gauge is positioned, is far from the centre and might therefore be influenced by temperature changes. Contact lens wear might limit the oxygen concentration in the cornea, leading to oedema. In a study of 20 patients the thickness of the central and peripheral cornea (approximately 6 mm from pupil centre) were measured. During a 14-hour wear period (pre-to-post sleep, mean duration 9.9 ± 0.7 hours) the cornea remained statistically unchanged when compared to the fellow eye (ultrasound pachymetry and Pentacam). In the eye with the CLS, the thickness changed from 523 μm pre study to 537 μm post study (swelling of 14 μm).28 No statistical differences were found in the horizontal and vertical radius of the cornea in either eye. The study by Tojo et al. showed a similar increase from 526 μm ± 29 to 533 ± 27 μm.29 Hubanova et al. demonstrated a mean maximal increase of 24.3 μm ± 9.4 (measured with the Visante Anterior Segment Optical Coherence Tomography [Carl Zeiss Meditec, Inc., Dubline, California, US]).30 In another study the thickness decreased from 560.3 μm before application of the CLS to 548.0 μm after 24-hour wear (statistically significant).15,17 The use of the CLS and correlations to the biomechanics properties of the cornea have not been published yet. The result of the CLS profile should not be influenced by open eyelids during wake and closed eyelids during sleep.17
Subtle analyses of the Triggerfish profile De Moraes et al. investigated 40 patients with treated open-angle glaucoma to determine if there was a correlation between the CLS profile and a progression rate of the visual field loss.25 The patients were classified as either fast, slow or minimal progressors. The authors defined six main outcome measures: number of large peaks (peaks with a height of 90 mVeq or more), mean peak ratio (mean peak height to time-to-peak), wake-to-sleep slope (1 hour before and 1 hour after the time the patient went to sleep), amplitude of the cosine curve (overall estimate of the magnitude of the signal oscillation), variability of the mean (amount of fluctuations) and area under the receiver operating characteristic curve (reflects the magnitude of the signals and the duration at which they remained above a reference line). Several parameters were associated significantly with faster progression (e.g. number of large peaks while awake, mean peak ratio while asleep, wake-to-slept slope, etc). There are several uncertainties in the study: it was not mentioned what kind of topical therapy the patients were receiving (beta blocker in the morning? or prostaglandin analogue in the evening?), that might have influenced the pattern of the CLS profile; it was not mentioned, if any of the patients had cataract surgery during the study period that might have improved the mean defect (MD)-value; in the graph showing the average CLS output profile the starting values differed in a large extent from the endpoint, without an explanation (start and end point should be at the same level); the much higher standard deviation at the end of the 24-hour profile was not explained; the value of 90 mVeq or more for a “large” peak was chosen without an explanation (maybe because the standard deviation was about 90 mVeq?). This arbitrary threshold demonstrated a mean peak amount of only 0.4 (during sleep) and 1.8 (during wake [within 24 hours]).
Tojo et al. described “fluctuations” of the IOP in 11 eyes with pseudoexfoliation syndrome and 11 normal eyes and found, that the range of fluctuations was significantly higher in the pseudoexfoliation group.29 All healthy eyes had their maximum value during the nocturnal period, but only 7 out of 11 eyes with pseudoexfoliation. In this study, fluctuations were defined as the difference between the maximum and minimum value (in mVeq) during the course of 24 hours, and the mean was 294 mVeq in healthy eyes versus 522 in pseudoexfoliation eyes. Additionally, the fluctuations of the diurnal (mean 270 versus 477) and nocturnal (mean 179 versus 272) period were interpreted separately.
Liu et al. described a few “uncharacterised factors” that may cause data variations: unknown relation between IOP and CLS signal at the beginning of wearing the CLS, the effect of unilateral CLS fitting on the sleep posture and IOP, and the signal baseline drift.15
Changing the sales pitch and summary
The CLS has no history of serious adverse events and can be used without any safety concerns. When the CLS was launched in 2009 it was marketed for “continuous monitoring of the IOP fluctuations” and the data were displayed in arbitrary units as a “fluctuation profile of continuous IOP profile”. Nowadays this product is promoted as recording a “24-hour profile of ocular dimensional changes”. The initial assumption was that IOP changes independently lead to changes in corneal curvature. Other factors may also have an influence on corneal curvature, such as variable filling of the choroid and other yet unknown mechanisms which may affect the volume of the globe. Possible artefacts (see above) have not been adequately defined. There is also no consensus on the definition of a “fluctuation” (peak, size of the mVeq change between two points versus multiple measuring points). The predominantly good demonstration of the ocular pulse (in 86.5%, ocular pulse frequency corresponds to the systole and diastole of the cardiac cycle17), which is usually a small value of 2–5 mVeq, proves that this device can measure some corneal curvature change. Other potential influences on the profile derived from the total of single measurements must be evaluated by further baseline experiments. As long as these studies are not performed and published, the interpretation of the CLS profiles, at least, cannot be taken at face value, making it inappropriate for daily clinical use.
Cost effectiveness
It is not known which countries have a billing code for the “(pseudo) continuous” IOP measurement. The cost for the CLS is not inexpensive, and the usefulness for the diagnosis and management of glaucoma patients has not been proven. George Spaeth (Wills Eye Hospital, US) posed the following question at the 2016 European Glaucoma Society Meeting in Prague: “Is there any evidence that a patient with glaucoma would suffer harm without the knowledge of a 24-hour IOP profile?” The answer is missing.
Despite the outstanding technical construction of the CLS it is possible that in future implantable IOP measurement devices, which use capacitive sensors, and work completely independently of stretching ocular tissues, would provide more reliable data.
So what do we know?
• The CLS Triggerfish is a contact lens with embedded strain gauges at a diameter of 11.5 mm, which should measure changes of the corneal curvature that might be due to changes in IOP.
• The result is a profile of 288 measurements in mVeq (equivalent with an unknown reference, not mV. The relationship between CLS output in mVeq and mmHg is unknown).
• The majority of the profiles show a nocturnal acrophase (are those without acrophase eyes at a greater risk for glaucoma or progression?)
• Safety and tolerability are good, minor adverse events resolve spontaneously within a day.
• Reproducibility is fair to good, but due to physiological changes of the IOP during a day or a week not a primary importance.
• Validity in terms of comparability to an established empirical test is missing.
• Definition of “fluctuations” is missing (within 24 hours or 24-hours maximum-minimum).
• Interpretation of profiles is not standardised and definition of artefacts missing.
• Change of the marketing massage from “24-hour IOP profile” or “continuous monitoring of IOP fluctuation” to “24-hour profile of ocular dimensional changes”.
• No billing codes so far.







