Neovascular age-related macular degeneration (nAMD) is highly prevalent particularly among elderly populations worldwide, and early treatment is imperative to prevent devastating progressive irreversible vision loss and eventual legal blindness.1–3 It is important that patients‘ vision is maintained in nAMD rather than following the deteriorating course that characterises the disease. Several anti-vascular endothelial growth factor (VEGF) medications are available for the treatment of nAMD but these have differing modes of action, allow differing dosing regimens.4,5 This article reports a symposium that aimed to evaluate the use and effectiveness of aflibercept (Eylea® , Bayer, Leverkusen, Germany) in nAMD, broadly across the disease, in patient subtypes and those with disease progression. The possible impact of molecular design of aflibercept on the durability and efficacy of anti-VEGF agents was also explored.
The use of aflibercept in nAMD is supported by two large pivotal trials that showed notable and sustained efficacy.6 These studies have also explored long-term use of aflibercept and its effect on vision outcomes.6–8 More recently, data have been gathered on treat-and-extend regimens and real-world visual acuity outcomes with use of aflibercept.9,10
In nAMD, anti-VEGF treatments are given as intraocular injections which have a high treatment burden and this can negatively impact quality of life.4,11 Aflibercept allows some patients who have completed one year of on-label treatment to be moved to less frequent dosing than every 8 weeks. This does not compromise efficacy which is desirable for both patient and ophthalmologist and has the potential to reduce costs.12 Several ongoing and planned audits aim to provide more real-world evidence of the impact of aflibercept in nAMD and aim to provide data on long term outcomes. These audits also include aflibercept regimens with extended treatment intervals. When complete, the study findings may help to shape future treatment approaches in nAMD.
Strength across the disease – evidence in neovascular age-related macular degeneration
Sebastian M Waldstein
Medical University of Vienna, Austria
nAMD is the leading cause of vision loss in the elderly13 and is a leading cause of legally recognised blindness that affects 10–13% of adults aged >65 years in North America and other genetically similar populations.14 The disease affects a similar proportion of Asian and Caucasian individuals.15–17 The prevalence of nAMD increases exponentially with every decade after the age of 50 years.13 Anti-VEGF agents now provide a highly effective treatment option for nAMD and have lessened the impact of the disease.6,18,19 These treatments, however, have turned the burden of disease in nAMD into a burden of care.14
The efficacy of anti-VEGF treatments in nAMD has been demonstrated in numerous clinical trials, the largest of which were the VIEW studies (VIEW 1 in US, VIEW 2 in Europe, Asia and Australia, total n=2,457 – see end of text for study name definitions).6 Patients were randomised equally to either intravitreal ranibizumab (Lucentis®, Genentech, South San Francisco, California, US) (0.5 mg/4 weeks) or three different intravitreal doses of aflibercept: 2 mg/4 weeks, 0.5 mg/4 weeks (unlicensed dose) or 2 mg/8 weeks after 3 monthly injections. These dosing regimens were maintained for 56 weeks after which both agents were given as needed on a pro re nata (PRN) basis but had to be administered at least quarterly until week 96 with monthly monitoring. It should be noted that the treatment regimen for ranibizumab as used in the VIEW studies did not reflect its current recommended treatment frequency. The licensed posology for ranibizumab states that initially three or more consecutive, monthly injections may be needed but thereafter, monitoring and treatment intervals should be determined by the physician and based on disease activity.20 The licensed posology for aflibercept states that it should be initiated with one injection per month for three consecutive doses, followed by one injection every two months. After the first 12 months the treatment interval may be extended based on visual and/or anatomic outcomes.21
Baseline demographics in the VIEW studies were well balanced in the treatment arms and representative of typical treatment-naïve, nAMD populations in clinical trials.6 For the primary outcome, the proportion of patients maintaining vision (losing <15 ETDRS letters of best corrected visual activity [BCVA]) at week 52 was similar across all treatment arms (95% of patients treated with aflibercept 2 mg/8 weeks versus 94% of patients treated with ranibizumab 0.5 mg/4 weeks).6 At week 96 the proportions maintaining vision were 92% in both groups. In addition, mean reductions in central retinal thickness (CRT) at week 52 were also similar across treatment arms (-139 µm for aflibercept 2 mg/8 weeks versus -128 µm for ranibizumab 0.5 mg/4 weeks. At Week 96, these CRT values were -133 µm and -118 µm, respectively. Fewer injections of aflibercept were administered in the 8-weekly 2 mg aflibercept arm to week 96 compared with ranibizumab (11.2 for aflibercept 2 mg/8 weeks versus 16.5 for ranibizumab 0.5 mg/4 weeks). In both the VIEW 1 and 2 studies, aflibercept had a safety profile similar to that of ranibizumab.
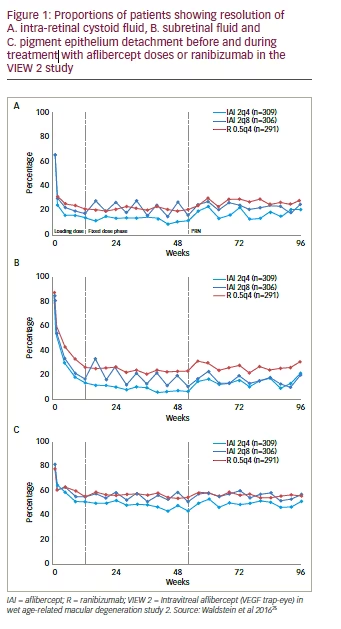
As a result of the VIEW and other anti-VEGF trials, more is known about the pathophysiology of nAMD and imaging biomarkers for disease progression. An example of such a biomarker is pigment epithelial detachment (PED). PED lesions have varying morphology but are important prognostic factors in nAMD patients. Although not primarily associated with vision loss, new onset PEDs are commonly associated with a profound subsequent deterioration in BCVA.22 In patients aged over 55 years, PED lesions often occur secondary to choroidal neovascularization.23 Approximately 50% of patients with newly diagnosed PED experience significant visual loss (>3 lines) one year from diagnosis.24
In the VIEW 2 trial, a reading-centre based grading of monthly optical coherence tomography (OCT) scans was performed. All patients underwent advanced image analysis for the characteristic pathomorphologic lesions in nAMD at all time points. Of the 1,202 patients available for intention-to-treat (ITT) analysis, 64.4% of eyes had intra-retinal cystoid (IRC) fluid, 84.1% had subretinal fluid (SRF) and 80.1% had PED lesions at baseline.14 These features are therefore very common in nAMD and each is diagnostically important. Subanalyses showed that the resolution of IRC and SRF from baseline was more pronounced with aflibercept treatment than ranibizumab. In all study arms these improvements were maintained until 96 weeks. At the end of the loading phase (12 weeks), the proportions of patients with IRC, relative to baseline were reduced by 51.2% with 2 mg aflibercept (pooled) and by 45.7% with ranibizumab, and the proportions of patients with SRF relative to baseline were reduced by 68.3% and 60.8%, respectively. There was also a more pronounced reduction of PED lesions with 2 mg aflibercept than ranibizumab and this was maintained to 96 weeks; however, PED lesions remained present throughout the study in at least 50% of patients regardless of treatment. At the end of the loading phase, relative to baseline, the proportion with PEDs was reduced by 35.6% with aflibercept (pooled) and by 28.5%, with ranibizumab (Figure 1).25
In the VIEW studies, most patients maintained good vision under PRN dosing during the second year of the trials but some experienced a sizable decrease in BCVA.26 Examination of retinal morphology showed that these patients were a defined morphologic subgroup who had PEDs at baseline and developed new IRC during the second year.14 This is likely to be a result of insufficient detection and monitoring of PEDs in patients with nAMD. In the second year patients were moved to treatment on a PRN basis and this reactive strategy may have allowed PEDs to worsen leading to secondary IRC formation and irreversible vision loss.14 PEDs should therefore be carefully monitored and PED growth proactively treated prior to the appearance of IRC and consequent retinal damage.22
PED lesions are relatively resistant to therapy. Treatment with aflibercept in the VIEW studies, showed a good efficacy on these lesions. For PEDs observed at single visits, aflibercept 2 mg/8 weeks produced a 23% relative higher rate of PED flattening compared with ranibizumab 0.5 mg/4 weeks. For PEDs observed at two or more consecutive visits (i.e. sustained PED flattening), aflibercept 2 mg/8 weeks produced a similar rate of sustained PED flattening compared with ranibizumab.27,28
An important putative subtype of nAMD is polypoidal choroidal vasculopathy (PCV). This is characterised by inner choroidal abnormal vascular networks ending in aneurysmal dilatations (polyps).29 PCVs
are most common in Asian communities (Chinese: 22.3–24.5%, Korean: 22.2–24.5%, Japanese: 23.0–54.7%, Taiwanese: 49.0%, Caucasian: 8.0– 13.0%).30 The incidence however, may be underestimated since definitive diagnosis requires indocyanine green angiography (ICGA) and this is not routinely performed in many countries.31 Further work is therefore needed to establish the true prevalence of PCV.
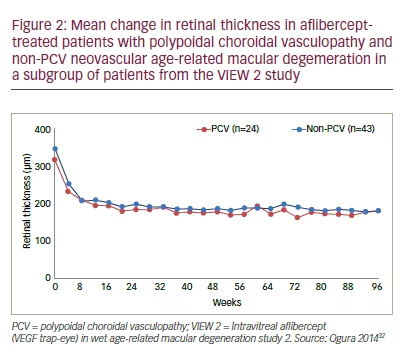
In the VIEW 2 study, a subgroup of 101 Japanese patients were examined using ICGA and 33% were found to have PCV; this was balanced in all four treatment groups. Among these patients, over 96 weeks, the mean change in CRT in all aflibercept-treated groups was similar in patients with or without PCV (Figure 2).32 This indicates the versatility of aflibercept in different types of nAMD but it should be noted that these results are from a small number of patients. In a series of PCV case studies conducted in Japan (n=16–91, total: 336), 33.3–77.7% of aflibercepttreated patients showed complete polyp regression.33–38 The ongoing Aflibercept in Polypoidal Choroidal Vasculopathy (PLANET) study (Phase 4, planned n=313) aims to collect further data from multiple Australian, Asian and European centres on the efficacy and safety of aflibercept with and without photodynamic therapy in subjects with PCV.39

Translating efficacy into real-world effectiveness
Faruque Ghanchi
Bradford Teaching Hospitals, UK
In managing nAMD it is important to strike a balance between under-treatment resulting in poor vision and over-treatment resulting in a high burden on patients and excessive costs. A treatment regimen should enable the physician to monitor, evaluate, and treat at the same time to reduce unnecessary visits. It is thus desirable to reduce the frequency of intraocular injections. For aflibercept, three treatments are initially given at monthly intervals and then the injection interval is extended to every two months for the first year.21 In the second year, the treatment interval can be further extended to 10 weeks and if the anatomic response is maintained it may be further extended to 12 weeks.6,32
In the two pivotal trials of ranibizumab in the treatment of nAMD, ANCHOR and MARINA, (n=423 and 716) injections were given monthly over 12 months, visual acuity improvements of 11.3 and 7.2 letters from baseline were reported.18,19 However, a database study of five years’ clinical experience in 92,976 treatment episodes (n=12,951 eyes of 11,135 patients) at centres in the UK did not find the same level of acuity improvement.40 At week 52 there was a 2-letter improvement in vision but by week 156 this had changed to a 2-letter deterioration compared with baseline suggesting that ranibizumab did not perform as well in real-world use as in clinical trial settings.
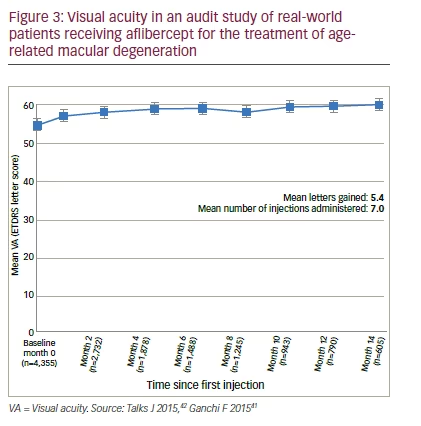
The National Aflibercept UK User Group was established to document real-world experience of aflibercept in the management of nAMD at multiple centres. This is an audit conducted at 16 centres using the regimen used in the VIEW studies. The mean patient age was 80.0 years (median: 81.0 years, 63.7% female).41,42 At baseline the mean visual acuity was 53.7; after 1 year (primary outcome) this was 58.8 (5.4 letters gained) after a mean 7.0 injections (Figure 3). Among secondary outcomes, patients with the poorest baseline visual acuity (<35 letters) gained approximately 10 letters by 1 year, a greater improvement than seen in patients with better baseline vision (35 letters – >70 letters) who improved by approximately 5 letters. In addition, the proportion of patients with legal vision for driving (BCVA >70 letters) more than doubled during treatment (17.2% of eyes at baseline increasing to 35.7% of eyes at Month 12). Approximately 92.8% of eyes maintained vision during aflibercept treatment (lost <15 letters) at 1 year,41 a comparable figure to the 95.4% maintaining vision in the bimonthly arm of the VIEW studies. The second year of the aflibercept audit study will include useful outcome data on patients who have moved to treat-and-extend regimens where possible.
Other real-world audits have also shown the rapid and robust effect of aflibercept in nAMD. In a retrospective review of records of 250 treatment-naïve nAMD patients at the Moorfields Eye Hospital, London, UK, patients received monthly injections (loading dose) followed by fixed bimonthly dosing.43 After 16 weeks, the proportion of patients with legal driving vision had more than doubled (13.6% at baseline and 34.4% at 16 weeks) (Figure 4). In addition, there was a mean gain of +7.2 after 8 weeks and +6.6 letters after 16 weeks. The proportion of patients with a CRT <300 µm at baseline increased to 89.6% after 16 weeks; and there was a mean reduction of -76.7 µm after 8 weeks and -70.2 µm after 16 weeks.
A real-world study, the ongoing RAINBOW study, aims to evaluate the use of aflibercept in nAMD in France.9,44 The study has a target of 600 retrospective and prospective treatment-naïve patients, evaluated over a 48-month period after the first aflibercept injection. Interim, 3-month results from 81 patients are positive and show a vision improvement +6.9 ± 12.1 letters BCVA from baseline which is similar to the previous findings of the VIEW 1 and 2 studies.
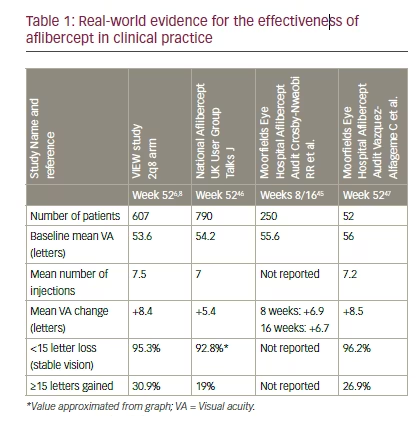
Data available to date from real-world studies and audits (n=52–790) all show substantial benefits from aflibercept treatment (Table 1) with visual acuity gains of 5.4–8.5 letters over 16 weeks to 1 year and 92.8–96.2% with stable (<15 letter vision loss) over 1 year.6,8,45–47 Further planned or ongoing Phase IV/IIIb trials are expanding the evidence base
for treat-and-extend regimens of aflibercept in nAMD. These include the clinical trials ARIES48 (planned n=268) and AZURE49 (planned n=330).
Other ongoing and forthcoming trials include: TITAN50 (planned n=225 in patients with nAMD and refractory to ranibizumab), PERSEUS51 (planned n=988 patients with nAMD) and PERSEUS-IT52 (planned n=800 treatmentnaïve patients with nAMD).
Emerging real-life evidence therefore indicates that bimonthly aflibercept is an effective treatment option in real-world management of nAMD. This treatment achieves good visual outcomes with reduced treatment burden compared with ranibizumab. The sustained efficacy of aflibercept lends itself to extended treatment intervals, helping to improve patient throughput in the clinic and reduce burden of administering and receiving intraocular injections for both clinicians and patients.

Aflibercept – designed to make a difference
Sascha Fauser
University Hospital of Cologne, Germany
Aflibercept acts by binding to key circulating factors, VEGF-A, VEGF-B and placental growth factor (PlGF) which inhibits binding of VEGF to VEGF receptors (VEGFRs) 1 and 2.53 Various important processes in the maintenance of tissue function in normal and disease conditions are therefore disrupted, including: vascular and non-vascular functions, angiogenesis, vascular permeability and lymphangiogenesis. Aflibercept is a fusion protein consisting of human VEGFR-1 and VEGFR-2 domains and human IgG1 Fc.54,55 It has a dual action and traps all VEGF-A isoforms and PIGF. Aflibercept was designed to block all members of the VEGF family; it binds in a strict 1:1 ratio and has a higher affinity for VEGF than native receptors. As a result of its different binding domains, aflibercept is able to form inert complexes with VEGF.
The pharmacokinetic profile of aflibercept provides sustained efficacy and allows long intervals between doses in many patients. A timeand dose-dependent mathematical model found that the intravitreal biological activity of a 0.5 mg dose of ranibizumab 30 days after injection is equivalent to the predicted activity of a 1.15 mg (unlicensed) dose of aflibercept after 79 days. This was also predicted to be equivalent to the activity of a 4.0 mg (unlicensed) dose of aflibercept after 87 days.56 The intravitreal half-lives of ranibizumab, bevacizumab, and aflibercept were estimated to be 3.2, 5.6, and 4.8 days, respectively.57
A study that included 83 patients with nAMD, showed that ranibizumab 0.5 mg suppressed VEGF in the aqueous humour for a mean 36.4 ± 6.7 days (standard deviation [SD]) following an injection. In individual patients this suppression time was very consistent (intra-individual stability >3 years).58 In addition, there were no rebound effects and
VEGF levels were not associated with choroidal neovascularization. In comparison, a nonrandomised prospective clinical study that included
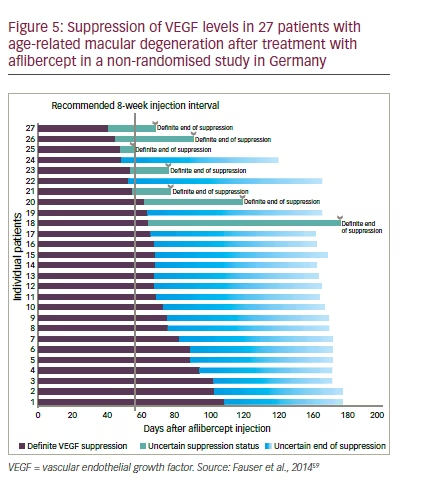
27 patients treated with aflibercept, given on an unlicensed PRN basis, showed that VEGF in the aqueous humour was suppressed for a mean 71 ± 18 days (SD) (Figure 5).59 This suggested a durable VEGF inhibition for most patients dosed with aflibercept every 8 weeks.
Despite the wide range of VEGF suppression times for anti-VEGF treatments, it is difficult to correlate these with clinical performance. VEGF is necessary but not sufficient for recurrent disease activity. Loss of VEGF suppression does not result in the immediate appearance of morphological or functional signs. Furthermore, studies which typically treat at 4-week intervals also level out some differences between anti-VEGF agents.60,61
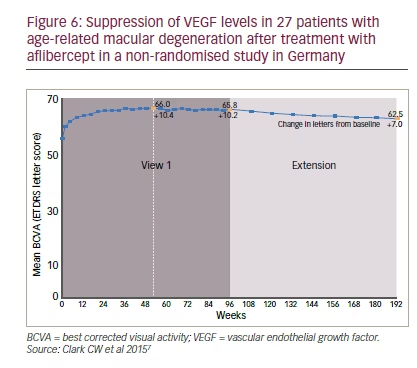
There are limited data on the sustained effect of less frequent dosing of anti-VEGF treatments in nAMD. In the VIEW studies patients who received bimonthly or quarterly aflibercept dosing from week 52 to 96 showed good maintenance of visual acuity (losing <15 ETDRS letters).6,8 During the second year of the VIEW 1 study (weeks 52–96) and its extension (weeks 96–192), approximately half of the patients were maintained on quarterly aflibercept dosing over a total of 3 years.7 During this time, mean BCVA was essentially stable, changing only from 66.0 to 62.5 letters (+10.4 to +7.0 letters compared with baseline) (Figure 6).
In the LUCAS study that compared ranibizumab with bevacizumab (n=431), patients were initially treated on a monthly basis and then the dosing intervals were extended as needed by 2-week increments up to 12 weeks.62 In order to maintain visual acuity, 37.1% of patients receiving ranibizumab could not be extended beyond 12-week intervals, 4.7% could not be extended beyond 8 week intervals and 32.9% could not be extended beyond 6 week intervals. The mean treatment interval for ranibizumab over 1 year was 6 weeks. Similar dosing intervals were necessary with bevacizumab.
The treat-and-extend regimen of aflibercept is being further investigated in the ongoing ATLAS study. This is a prospective, 2-year clinical trial in 40 patients with treatment-naïve nAMD in which, following one year of on-label treatment, dosing intervals are extended by 2-week increments up to a maximum 16 weeks provided that visual acuity is maintained and extension criteria are met. Results at 1 year showed that less frequent dosing was possible in most patients and efficacy was sustained.10 For the primary outcome, there was a mean gain of 7.5 letters (ITT population). For other outcomes, the mean number of injections during the year was 8 and over 70% of patients achieved a treatment interval of =8 weeks. Results for these endpoints at 2 years are awaited.

Discussion and conclusion
In the treatment of nAMD, aflibercept can be differentiated from other anti-VEGF agents on the basis of its innovative molecular design, which results in a robust and broad mechanism of action59,63 and provides sustained action.6,7 In a series of large clinical trials aflibercept showed favourable efficacy and this experience has been continued in completed and ongoing audits and studies of real-world clinical practice.41,45,46
In treating nAMD with anti-VEGF agents, frequent intraocular injections create a high treatment burden both for patients and ophthalmologists. Some physicians believe that using a PRN regimen when treating nAMD can expose patients to multiple disease recurrences, resulting in disease progression. Providing regular treatments at extended intervals appears to be a more advantageous route to controlling the disease. For many patients, after the initial loading doses, extending dose intervals to 8 or 12 weeks provides sustained maintenance of vision for durations up to 2 years6,32 and possibly beyond. Some ophthalmologists have suggested that treatment cessation in certain patients may be possible but identifying suitable patients is challenging.
The finding that clinical activity in nAMD returns at around half the duration of VEGF suppression may indicate that current assay methods are insufficiently sensitive to detect the low levels of VEGF that are needed to reactivate the pathological processes. It may also indicate that circulating levels of VEGF are lower or rise more slowly than those in the retina so detecting zero levels in the aqueous humour does not indicate an absence of disease activity and progression.
Collective clinical experience to date, shows that aflibercept can be used as a proactive, effective and durable treatment option in all different forms of nAMD, including patients with disease progression. Further post-marketing and real-world audits that aim to evaluate aflibercept in nAMD are in progress. These are likely to create a substantial body of evidence supporting its wider use in the maintenance of vision with minimal dosing. As the leading cause of blindness in the elderly, it is imperative that nAMD is treated in as many patients as possible. The efficacy of anti-VEGF agents and their use in extended treatment regimens may help achieve that goal.
Definitions of study name abbreviations:
ANCHOR: Antibody for the treatment of predominantly classic choroidal neovascularization in AMD; ARIES: Managing age-related macular degeneration (nAMD) over 2 years with a treat and extend (T&E) regimen of 2 mg intravetreal aflibercept – a randomized, open-label, activecontrolled, parallel-group Phase IV/IIIb Study; ATLAS: Repeated eye Injections of aflibercept for treatment of wet age related macular degeneration; AZURE: An open-label , randomized, active controlled, parallel-group, Phase 3b study of the efficacy, safety, and tolerability of 2 mg aflibercept administered by intravitreal injections using two different treatment regimens to subjects with neovascular age-related macular degeneration (mAMD); LUCAS: Lucentis compared to avastin study; MARINA: Minimally classic/occult trial of the anti-VEGF antibody ranibizumab in the treatment of neovascular AMD; PERSEUS: Investigating the effectiveness of Eylea in pateints with wet age-related macular degeneration; PERSEUS-IT: Aflibercept (Eylea) in routine clinical practice in patients with age-related macular degeneration in Italy; RAINBOW: Real life of aflibercept In France: observational study in wet AMD; TITAN: Real life of Aflibercept in France in patients refractory to ranibizumab: observational study in wet AMD; VIEW 1 and 2: VEGF Trap-eye: Investigation of efficacy and safety in wet AMD studies.







