Cross-linking (CXL) is a technique originating from the synthetic polymer field that can be used to alter a polymer’s physical and mechanical properties. It was first proposed for corneal use by Theo Seiler from Germany in 1995 and was later applied clinically in 1998.1 A corneal ‘crosslink’ is a bond between natural polymer chains such as collagen fibrils and proteoglycan core proteins. The induction of a photochemical reaction using riboflavin (vitamin B2) and ultraviolet A (UV-A) light (370 nm) produces these cross-links, which have been proved to generate free radicals that can strengthen and rigidify the cornea. This idea was conceived after observing the CXL of proteins that occurs naturally with ageing.2
Keratoconus is a common bilateral disorder that causes progressive thinning of the cornea, transforming the normally regular dome-shaped cornea into a more irregular conical profile.3 A defect in collagen allowing enzymatic degradation and fibrillary slippage is believed to be the cause of the weakened cornea. The resulting irregular astigmatism and corneal opacities lead to visual impairment. CXL can be used to treat keratoconus by stiffening the cornea, thus preventing the progressive change in shape.
Keratoconus typically presents during adolescence; it normally progresses until approximately 40 years old, by which time natural cross-links within the cornea have become established. Keratoconus staging can be performed using the Amsler-Krumeich classification that contains four stages, ranging from mild (grade I) to severe (grade IV).3 CXL offers the opportunity to change the natural history/evolution of the disease. The aim of the procedure is to slow the progression of this disease thus maintaining vision, and to prevent advanced changes that require more invasive therapy.
Keratoconus is one of several non-inflammatory conditions that cause thinning of the cornea, termed ectasias. Corneal ectasia can also result as a rare yet serious complication of laser-assisted in situ keratomileusis (LASIK) surgery. Progressive post-LASIK ectasia can weaken the cornea’s structure causing astigmatism that is difficult to correct with lenses or further LASIK surgery. Thus, CXL may be beneficial to slow this process.4
Techniques Used for Corneal CXL
Current techniques for CXL involve the use of riboflavin drops and UV-A light exposure to the cornea. The several methods used are described in detail below.
Epithelium-Off CXL
The first attempt to cease the progression of keratoconus by using photochemical CXL was reported by Wollensak and co-workers in 2003.4 The treatment technique used has become known as the Dresden protocol (named after the founding university in Germany).5 The first step in this protocol is to remove the central 9 mm of corneal epithelium. The corneal surface is then saturated with 0.1 % riboflavin solution for 30 minutes, after which the eye is exposed to 370 nm of UV-A with an irradiance of 3 mW/cm2 over an 8 mm diameter for a further 30 minutes. Additional riboflavin drops are given every 5 minutes during UV-A exposure. Essentially, the riboflavin within the corneal stroma is activated by UV-A to produce a photochemical reaction that creates collagen and proteoglycan cross-links. The original Dresden protocol takes approximately an hour and is usually performed under topical anaesthesia. Following treatment, a short course of topical antibiotics and vitamin A eye ointment is prescribed, with further antibiotics and fluorometholone drops as necessary. Different methods of disrupting the corneal epithelium for epi-off CXL have also been described, including the use of corneal disruptor devices6 and chemical disruption using benzalkonium chloride.7
Epithelium-On CXL
Removal of the corneal epithelium in epithelium-off CXL (epi-off CXL) results in an iatrogenic corneal abrasion, causing post-operative discomfort that may last around 5 days. During this period there is an associated risk of infection, stromal haze and corneal melting. In order to reduce these post-operative complications, attempts have been made to develop collagen cross-links through an undamaged cornea (epithelium-on CXL [epi-on CXL]). This involves administering a more concentrated and altered solution of riboflavin, combined with other agents in an attempt to increase corneal epithelial permeability.8–11 In vitro studies on porcine corneas have indicated that riboflavin penetrates poorly, even when agents such as topical anaesthetics are utilised.12,13 Filippello and co-workers, however, reported clinical efficacy and reduced post-procedure symptoms and corneal opacity using an epi-on technique in a small cases series (n=20, follow-up 18 months).14 Iontophoresis is a non-invasive epi-on CXL technique that uses a weak electrical current to introduce riboflavin into the corneal stroma without the necessity of corneal epithelial removal.15–17 A generator delivers a constant current of 1 mA locally to the cornea for a set duration, usually 5–10 minutes. Irradiation is then administered as in the standard protocol. This new technique has shown promising results.16,17 With a similar efficiency to the standard protocol and the absence of side effects associated with epithelial debridement, it may become the preferred way of slowing the progression of ectatic disease. This new technique has shown promising results in terms of short-term efficacy in stabilising the progression of keratoconus, and a low side-effect profile. A comparison study is needed, however, to compare the relative efficacy of this technique with standard epi-off CXL techniques. Longer followup is also required to investigate the long-term efficacy produced by iontophoresis and epi-off CXL.
Modified Epithelium-Off CXL
Over recent years, epi-off CXL using the Dresden protocol has been widely adopted and refined to ensure the greatest efficacy and patient safety. Modifications have also been devised to increase the throughput of patients in clinic and to decrease patient anxiety during long procedures and reduce post-procedure discomfort. These include an accelerated and a pulsed technique.
Accelerated CXL
In theory, the same CXL reaction could be completed in less time using higher UV-A irradiation, assuming that the total exposure is kept constant. Technology and protocols are available for accelerated CXL (ACXL), which uses higher illumination intensities of up to 30 mW/cm2 for 3 minutes, over an 8 mm treatment zone. ACXL offers advantages to both patients and surgeons in terms of reduced treatment times. Clinical studies into the efficacy of ACXL versus standard CXL show that the results of both 6-18 and 12-month19 follow-up in patients treated with progressive keratoconus are comparable. However, recent in vitro data in porcine eyes, treated with CXL using different protocols, indicate that the CXL from ACXL may not be as much as with standard CXL treatment. Longer-term comparative follow-up studies with ACXL are required to confirm outcomes in slowing disease progression.
Pulsed CXL
Another modification that is used to increase the efficiency of CXL is using a pulsed UV-A beam. The formation of free radicals generated during the CXL process occurs at a much faster rate in the presence of oxygen; however, the oxygen within the tissues is very rapidly consumed, thus reducing the overall efficiency of the reaction.20 Therefore, the constant application of UV-A light reduces the amount of oxygen available, slowing the CXL rate and increasing the time required to complete the procedure. However, when an intermittent beam of UV-A (1 second on; 1 second off) is used, termed pulsed CXL, oxygen is allowed to diffuse into the corneal stroma when the UV-A beam is switched off. This replenishment of oxygen allows crosslinks to form at a faster rate than if the UV-A beam was continuously directed at the eye. By using pulsed illumination it is possible to achieve the same corneal stiffening effect within a shorter period of time and by delivering a lower UV-A energy dose, thereby maximising the CXL efficacy.
Combined CXL and refractive surgical techniques: ‘CXL plus’: CXL has been combined with various refractive techniques in an attempt to address not only the progressive nature of the ectasia, but also to tackle the associated corneal irregularity. It must however be noted that one of the limitations of interpreting the potential beneficial effects of these treatments is that of the relatively short follow-up in the published studies.
Excimer Laser Ablation Combined with CXL
Photorefractive keratectomy (PRK) has been carried out in combination with CXL with promising results in terms of both visual acuity (VA) and keratometric values.17,18 The Athens Protocol comprises partial PRK to a depth of 50 μm in combination with higher fluence CXL, with 3-year results on large cohorts of patients having shown improvements in uncorrected and corrected distance VA as well as reductions in keratometric values.19 Studies have also shown that there may be progressive effects on parameters such as keratometry for up to 3 years following treatment, which then seem to plateau, and this has been put forward as an argument for only partial correction of the surface irregularities, to avoid overcorrection.19,20 PRK and CXL may be performed on the same day or sequentially. However, sameday treatment appears to be more effective in terms of the visual rehabilitation of keratoconus.
Intracorneal Ring Segment Implantation Combined with CXL CXL has also been combined with the use of intracorneal ring segment (ICRS). These can be performed on the same day or sequentially in two sessions. While same-day CXL and ICRS is associated with more post-operative haze, it seems to be more effective in reducing the keratometric values on topographic examination. Both techniques appear similar in terms of uncorrected and corrected distance VA and refractive error. Same-day topography guided-PRK and CXL has also been studied in patients having had previous ICRS implantation for keratoconus.21 Zeraid and colleagues have shown significant improvements in VA and keratometry, as well as refractive sphere and astigmatism in keratoconus patients with low to moderate astigmatism.22 Similar results have also been achieved by various other groups, with good safety outcomes.23–25
CXL and Phakic Intraocular Lens Implantation
CXL can also be combined with the use of a phakic intraocular lens (PIOL) implantation, of which there are several types including iris fixated, angle supported and posterior chamber. Several reports have been made supporting the safety and efficacy of such lenses in combination with CXL in the visual and refractive outcome of patients with keratoconus.26,27
Despite riboflavin-based CXL becoming the conventional treatment for progressive ectasias, other collagen CXL therapies such as aliphatic betanitro alcohols, bacteriochlorophyll derivative WST11 with near infrared light and rose Bengal with green light are also being investigated.
Riboflavin Preparations Used for Corneal CXL
In the Dresden protocol, 0.1 % riboflavin in 20 % dextran is applied. Once riboflavin is applied to corneal surface, it diffuses into the stroma forming a concentration gradient (0.04 % at 400 μm after 30 minutes). The concentration within the stroma needs to be sufficient for the CXL reaction to occur. It is also important for blocking the UV light from penetrating deeper into the globe, potentially harming the endothelium and deeper structures from both UV and excessive free radical formation. It is preferable to ensure that the riboflavin has penetrated through the corneal stroma into the anterior chamber (visible as yellow staining) before UV-A application.28 The minimum recommended residual corneal thickness for CXL is 400 μm following epithelial removal to reduce the risks described above. However, a significant proportion of eyes with established keratoconus are below this level. In addition, there is progressive thinning of the cornea throughout the application of the standard riboflavin.29 Thus, hypotonic solutions have been developed in an attempt to reduce thinning and cause swelling prior to UV-A irradiation.
Raiskup and co-workers have shown that hypo-osmolar riboflavin can be used safely in keratoconus patients with thinner corneas.30 They showed that in 32 eyes treated this way, the mean pre-operative corneal thickness was 382.3±41.9 μm; following epithelial debridement was 337.0±51.9 μm, and increased to 451.8±46.7 μm after hypo-osmolar 0.1 % riboflavin for 30 minutes. Clinical results demonstrated stable vision and keratometry at 12 months, without corneal scarring or other safety concerns.
Interestingly, Vetter and co-workers applied various iso-osmolar and hypo-osmolar riboflavin preparations to porcine corneas to investigate their effect on the central corneal thickness, in an attempt to determine a composition that would cause moderate corneal swelling.21 This experiment demonstrated a range of outcomes (in terms of percentage thickness of the corneas pre-treatment), ranging from 66 % using the standard Dresden protocol solution (riboflavin 0.1 %/dextran 20 %) up to 151 % in other preparations. They however noted that the final thickness was not significantly correlated to osmolality; being also influenced by other factors such as pH and chemical composition. In support of this observation, in another study, iso-osmolar riboflavin (0.1 %) in hydroxypropyl methyl cellulose (HPMC) has been shown to maintain corneal thickness during the course of treatment compared with the thinning observed with dextran.31
From the literature already available, it is clear that further studies are still needed to clarify the optimal composition of riboflavin preparation(s) to achieve safe but effective CXL, particularly in thin corneas. This is especially significant with the increased adoption of accelerated CXL with higher UV-A intensity.
When to Cross-link the Cornea in Keratoconus
Not every patient with keratoconus requires CXL. The current indication for treatment is a proved progression of the disease due to the associated risks of the procedure. A defined criterion for disease progression has not yet been fully established, and the parameters used to show progression and the time frame in which these parameters are observed varies throughout the literature.
In the Dresden studies, progression indicating the necessity for treatment was based on an increase in Kmax at the apex of keratoconus of 1 diopter (D) in 1 year, deterioration of VA or the need for new contact lens fittings more than once in 2 years.5 Vinciguerra and co-workers defined keratoconus progression as a change in either myopia and/or
astigmatism of ≥3 D in the previous 6 months, a mean central K-reading change of ≥1.5 D observed in three consecutive topographies during the preceding 6 months or a mean central corneal thickness decrease of ≥5 % in three consecutive tomographies performed in the previous 6 months.23
Taking into account that current topographic methods have intrinsic variability, as a rule of thumb, the rule of 1s may be useful while monitoring patient topography results in order to identify candidates for CXL: a change in Kmax of ≥1 D, a change in Kmean of ≥1 D, a change in Kmax–Kmin of ≥1 D OR a change of ≥1 D cylinder on refraction, which may indicate progression. However, topographic measurements do have an inherent variability, and are affected by contact lens wear among other factors. Topography is also less accurate in the initial period (especially within 2 weeks) following a CXL procedure.
The UK’s National Institute of Health and Care Excellence (NICE) guidelines published in September 2013 advise that there is sufficient evidence for epi-off CXL but papers on the newer techniques used in epithelium-on CXL are lacking. Therefore, it is encouraged that outcomes are audited in order to compare the two techniques. When selecting patients for research, it is advised that corneal thickness and the likelihood of progression are noted so that they can be taken into account. It is important that corneal specialists only carry out CXL and that patients are aware of the longterm implications of treatment. Currently, there is also an on-going US Food and Drug Administration review of corneal CXL.
Evaluating the Success of Corneal CXL
There are several means of measuring the success or failure of CXL (see Table 1). These include morphological measures (such as keratometry), functional outcomes (such as uncorrected and corrected VA and improvement in quality of life), depth of riboflavin penetration (as confirmed by anterior segment optical coherence tomography) and the rate of complications. The availability of newer clinical instrumentation such as the ocular response analyser now also enable us to determine the extent of corneal stiffening by CXL.24
Adverse Effects and Complications of Corneal CXL
While CXL is a relatively safe procedure, adverse effects are known to occur, most of which are transient:
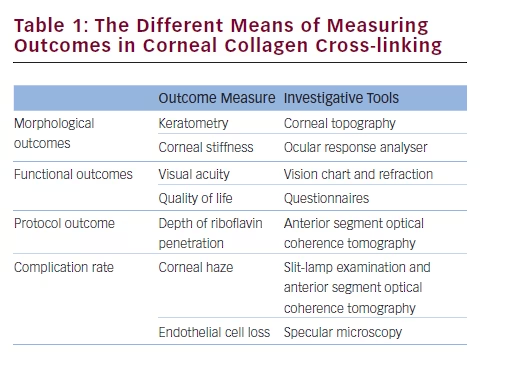
Corneal Haze
Temporary corneal haze is a common adverse effect of CXL (see Figure 1), and has been reported in 9.8 % of cases by Caporossi and
colleagues,25 with a mild degree of haze observed in all patients studied by Wittig-Silva et al.26 This tends to resolve gradually with topical steroid therapy. Permanent corneal haze has also been documented to be one of the more frequent observed side effects of CXL in the literature.27,32
Corneal Oedema
Transient stromal oedema has been observed on slit-lamp examination in 70 % of patients in the first 30 days following treatment.25 Unresolving corneal oedema, although less frequent, has also been described in up to 2.9 % of patients in a retrospective case series of 350 patients with a mean follow-up of 14 months.33 Gokhale and colleagues34 reported a case of severe transient stromal oedema in a patient despite a corneal thickness of greater than 400 μm. Possible causes of endothelial damage in patients with pre-operative corneal thickness greater than 400 μm undergoing CXL include inadvertent delivery of excessive doses of energy, intraoperative corneal thinning, insufficient instillation of riboflavin and improper calibration or focusing of the UV-A source. Measures to reduce such complications have been suggested, such as frequent intraoperative pachymetry with administration of hypotonic solution if corneal thickness falls below 350 μm, removal of the lid speculum and holding the eyelids closed as much as possible during instillation of riboflavin drops to prevent excessive corneal thinning secondary to evaporation and frequent calibration of the equipment.33,34 For the majority of cases in whom guidelines for CXL are adhered to, however, endothelial cell loss does not appear to be accelerated in comparison with the physiological reduction in endothelial cell density reported in the literature, at around 2 % per year.25
Damage to Extra-corneal Structures
UV exposure can itself cause direct damage to the ocular tissues, causing photokeratitis, cataract and thermal or photochemical damage in the retina. A homogeneous exposure to UV irradiation is necessary to avoid focal damage from hotspots, even if the average irradiance is 3 mW/cm2. The use of riboflavin together with UV-A irradiation decreases the cytotoxic irradiance level to 10 times lower than that of UV-A irradiation alone. Adhering to certain criteria, such as removal of the epithelium to facilitate diffusion of riboflavin throughout the cornea, application of riboflavin for 30 minutes prior to UV exposure and ensuring a minimal corneal thickness of 400 μm helps to reduce the risk of such damage to the ocular tissues.28 For these reasons, some protocols use preoperative pilocarpine drops to constrict the pupil and reduce the potential risk to retro-irideal structures. The long-term implications of CXL on the limbus and its stem cells have also not been completely determined and some propose the use of limbal protection during CXL with circular sponges.
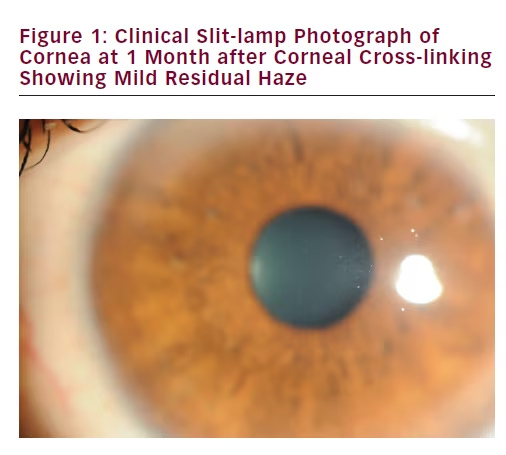
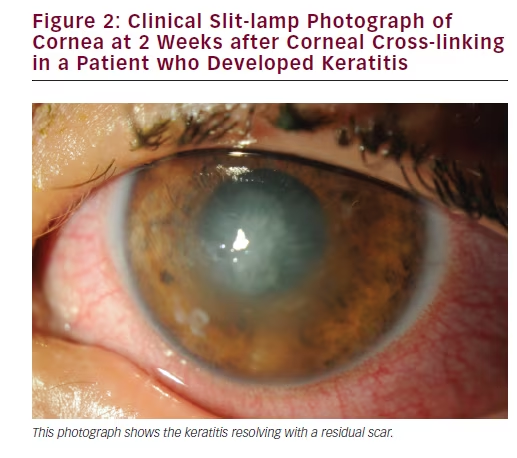
Keratitis
Keratitis is one of the more serious potential complications following CXL, which may be either infectious or sterile (see Figure 2). Patients with keratoconus who are known to have poor epithelial healing or active ocular diseases/inflammation (including atopic conjunctivitis) are in fact contraindicated for CXL due to the increased risk of infection. Other serious complications are rare and include corneal melting and perforation, and stromal scarring, while other minor complications such as pain are present in most to all patients treated with CXL.35
Major Studies of Corneal CXL in Keratoconus
There are several clinical studies to note that highlight the efficacy of epi-off CXL. The field does however lack formal clinical trials and data on the non-epi-off CXL methods. Notable studies for epi-off CXL are described below and summarised in Table 2.
Dresden Study
This is a longitudinal study with consecutive recruitment. The strength of the Dresden study is a large sample size but the study weaknesses are that it is a retrospective study, there is the lack of a control group and the sample size at 1 year is small. Patients recruited into the study were over 18 years of age with progressive keratoconus and a corneal thickness of at least 400 μm. Progression of keratoconus was defined as an increase in Kmax of 1 D in 1 year, a subjective deterioration of VA and the need for new contact lens fitting more than once in 2 years. The Dresden protocol was used for the treatment of all patients and patients were evaluated daily until re-epithelialisation, then at 1 month, 6 months, 12 months and annually thereafter. The study has 241 eyes with ≥ 6 months’ data and 33 eyes with ≥ 3 years’ data post-CXL. Fiftythree per cent of patients at 1 year (142 eyes) showed an improvement in best-corrected VA (BCVA) and 20 % showed stabilisation. The K value of the apex decreased by a mean of 2.68 D in 62 % eyes and remained stable within 0.5 D in 17 %. The 3-year data from 33 eyes showed 58 % had an improvement in BCVA and it remained stable in 29 % of eyes.

The K value of the apex also decreased by a mean of 4.84 D in 78 % and remained stable in 2 %.
Siena Eye Cross Study25
This study reports the long-term results of CXL in 44 progressive keratoconic eyes. The strengths of this study are that it is a prospective study with a minimum follow-up of 48 months. In addition, data were collected for both treated and non-treated fellow eyes. A significant weakness of the study is its poor definition of progression, lack of a formal control group and a small sample size at 4 years. The 44 treated eyes revealed a significant reduction of mean keratometry values by 1 year (–1.96 D) and 4 years (–2.26 D); uncorrected VA (UCVA) improved by a mean of +2.41 Snellen lines after 12 months and +2.75 after 24 months; and BCVA improved by a mean of +1.34 Snellen chart after 12 months and +1.93 D after 24 months. Data also showed a reduction in coma aberration. Keratoconus stability was detected in all 44 eyes after 48 months. Fellow eyes (the control group), on the other hand, showed a mean increase of +1.2 D after 1 year and 2.2 D after 2 years. Non-treated eyes also revealed a mean progression of 1.5 D in more than 65 % after 24 months.
Australian Study26
This study is a prospective unmasked randomised control trial. The other strengths of this study are its good sample size, 3 years’ data and the age range of between 16 and 50 years old. Patients with progressive keratoconus were defined over 12 months with an increase of at least 1 D in Kmax, an increase in astigmatism as determined by manifest subjective refraction of at least 1 D or a minimum of 0.1 mm decrease in the back optic zone radius of the best-fitting contact lens. A modified Dresden protocol for treatment was used. One hundred eyes were randomised into CXL or control groups, and results were reported from 48 control and 46 treated eyes. Treatment resulted in Kmax flattening by 0.72 D at 12 months, by 0.96 D at 24 months and by 1.03 D at 36 months. The mean change in UCVA was –0.15 logMAR at 36 months and BCVA –0.09 logMAR at 36 months. By contrast, control eyes revealed an increase in Kmax by a mean of 1.2 D at 12 months, 1.7 D at 24 months and 1.75 D at 36 months; and mean change in UCVA measured +0.10 logMar at 36 months. In conclusion, at 36 months, there was a sustained improvement in Kmax, UCVA, BCVA after CXL while the control group demonstrated further progression of keratoconus.
Conclusion
CXL is a successful technique that is able to slow the progression of keratoconus and other forms of corneal ectasia. At the moment the epi-off CXL technique (Dresden protocol and/or accelerated protocol) is the most widely used technique for CXL.







