Ocular allergy is one of the most frequently encountered conditions in clinical eye care—epidemiologic reports have shown that it affects up to 60 million US citizens.1 The eye, being continually exposed to the external environment, is a common site of allergic inflammation. The classic clinical signs of this inflammation are seen in the conjunctiva. Allergic conjunctivitis (AC) manifests as a immunoglobulin E (IgE) hypersensitivity reaction that occurs when the ocular surface is exposed to external antigens.2–4 These antigens include airborne pollen, animal dander, and other environmental antigens.3,5 Even though these environmental antigens are not always threats to the ocular surface, they elicit an inappropriate adaptive immune response in some individuals, leading to this hypersensitivity reaction.3,6,7
Ocular allergy comprises clinically distinct forms, ranging from the milder, relatively benign variants such as seasonal and perennial AC (SAC and PAC, respectively) to the more serious sight-threatening variants, such as vernal and atopic keratoconjunctivitis (VKC and AKC, respectively). Of these types, SAC and PAC are the most common and together, represent 95 % of all ocular allergy cases in the US.8 Both SAC and PAC develop as a result of an immediate type 1 IgE hypersensitivity response.6 Signs and symptoms associated with these types of conjunctivitis include ocular and periocular pruritus, redness, chemosis, watery discharge, burning, stinging, and photophobia. The main goal of treatment is to reduce inflammation early and to minimize further ocular surface irritation and discomfort.9,10
SAC is most commonly caused due to exposure to pollens from grasses, trees, ragweed, or other seasonal plants. It therefore tends to occur frequently in spring and autumn, which are seasons associated with higher levels of these airborne allergens.11 While the most obvious solution would be avoidance of the causative agents, this may not always be practical. The most commonly used nonpharmacologic interventions include cold compresses, lubrication such as artificial tears, and the daily disposal of contact lenses in patients who are wearers.12 Saline eye drops have been described as effective in reducing signs and symptoms in 30–35 % of cases.13,14
If these options are not successful, multiple pharmacologic treatment modalities are available for SAC, of which antihistamines, mast cell stabilizers, dual-acting mast cell stabilizers-antihistamines, nonsteroidal anti-inflammatory drugs (NSAIDs), and topical ocular corticosteroids are commonly used. These drugs are available in varying doses, act at different levels of the allergic response, and offer different rates of efficacy. While a number of studies address the efficacy or mechanisms of action of individual drugs within these classes, there is a gap in the literature in comparing the efficacy of dual-acting agents to corticosteroids based on mechanism of action. This article reviews the mechanisms of action of antihistamines and mast-cell stabilizers both as stand-alone agents, and as dual-acting mast cell stabilizers-antihistamines and corticosteroids.
The Allergic Inflammatory Response
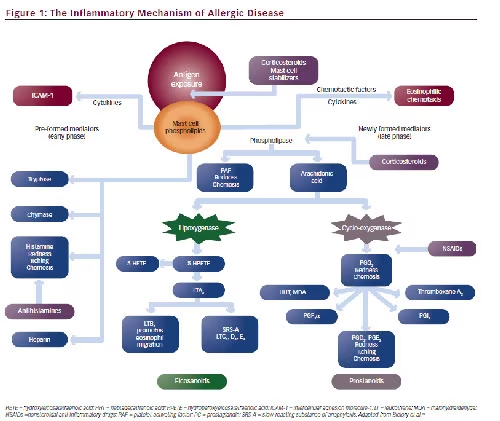
The allergic inflammatory response occurs in three phases (see Figure 1): a sensitization phase, an early phase, and a late phase.15 The major events that occur in each phase of the reaction are as follows:
Sensitization Phase
The sensitization phase occurs when the ocular surface encounters aeroallergens such as pollen. The allergens are processed by antigenpresenting cells (APCs) present on the epithelium of the conjunctival mucosa and presented on the surface of these cells as peptide fragments in association with the major histocompatability complex (MHC) II molecule. This allergen/MHC activates naïve T helper (TH) cells, resulting in the maturation of the naïve cells into mature type 2 TH cells, which, in turn, results in the production of cytokines, such as interleukin (IL)-4 and IL-13, which interact with naïve B cells. Antibody switching occurs in these B cells to stimulate the production of IgE, which then binds to receptors on the surface of the basophils and mast cells.15 Each mast cell has over a million high affinity receptors, FcεRI for IgE.
Early Phase
Upon encountering the same allergen again, the sensitized eye responds by triggering a type I hypersensitivity response, during which the allergen
binds to mast cell-linked IgE antibodies and facilitates the cross-linking of these antibodies. This leads to changes in the outer surface of the mast cell, making it more permeable to calcium ions. The crosslinking of a critical mass of IgE antibodies causes the mast cells to degranulate and release a variety of primary inflammatory mediators that are stored in their preformed granules. These include histamine, serotonin, lipid-derived mediators, such as leukotrienes C4, D4, and E4 and prostaglandins D2 and E2, platelet-activating factor, proteases such as tryptase and chymase, heparin, and other eosinophil and neutrophil chemoattractants.15 Histamine, the classic mediator of allergic inflammation, is a primary amine that plays a pivotal role in the pathogenesis of all allergic diseases. It is released from mast cells and basophils after a specific allergen binds to the IgE present on the cell surface. It binds to specific receptors and stimulates the increased release of multiple proinflammatory cytokines and arachidonic acid metabolites, resulting in a vasoactive effect.4,16 The signs and symptoms of the allergic reaction are primarily attributed to the activation of H1 receptors, leading to an increase in vascular permeability and a sensation of itching and pain.17 In addition, other mediators such as leukotrienes, prostaglandins, platelet-activating factor, and eosinophil chemoattractants released by mast cells add to the symptoms of the early-phase response; this usually begins within seconds of exposure to the allergen, and lasts for up to 40 minutes after exposure.3,15,18 The leukotrienes have activities similar to those of histamine, but are more than 100 times more potent on a molecule-for-molecule basis. Overall, the signs and symptoms of the early-phase response include itching, increased chemosis from increased vascular permeability and injection/ vasodilation, burning, and foreign body sensation.
Late Phase
The late-phase reaction—characterized by migration and infiltration of eosinophils, neutrophils, and basophils to the ocular surface— begins 4–12 hours after the release of the mast-cell mediators and perpetuates the allergic inflammation of the conjunctiva.4,15 The release of chemotactic factors in the early phase of the allergic reaction triggers a secondary cascade of inflammatory reactions marked by the release of newly formed leukotrienes and prostaglandins; this secondary cascade is responsible for the late phase of the allergic reaction.19 Mast cells produce newly formed cytokines that prolong the allergic response. These secondary messengers are IL-4 and IL-5, tumor necrosis factor alpha (TNFα), and platelet-activating factor; they stimulate the production of intercellular adhesion molecule 1 (ICAM-1) and vascular cell adhesion molecule 1 (VCAM-1), and thereby provide signals to initiate a second wave of inflammatory reactions. Additionally, cell adhesion molecules stimulate conjunctival inflammation with a prevalence of eosinophils, neutrophils, and lymphocytes with a type 2 TH cell profile of cytokine production and mucosal hyperreactivity.18
Mechanism of Action of Antihistamines in Ocular Allergy
As is the case with many allergic responses, antihistamines are a reasonable therapeutic option for ocular allergy and work primarily by H1 receptor antagonism.16 In most allergic conditions, oral H1 antihistamines are one of the drug types of choice. However, in the case of allergic conjunctivitis, topical antihistamines are often preferred to their oral counterparts. This is because the conjunctiva is easily accessible, allowing topical drug delivery to provide rapid action combined with ocular surface hydration.4 Because histamine is a major contributor to the signs and symptoms of the immediate reaction, topical antihistamines play an important role in providing symptomatic relief.4
Antihistamines used to treat SAC include levocabastine, emedastine, antazoline, and pheniramine; vasoconstrictors are commonly co-administered with these drugs to improve symptomatic relief.4,20–22 A study showed that 0.1 %, 0.2 %, and 0.3 % chlorpheniramine; 0.3 % dexbrompheniramine; 0.2 % pyrilamine; and 0.4 % and 0.5 % pheniramine were all effective in relieving the itching and conjunctival injection associated with topically applied histamine.23 In a study involving 40 SAC patients, Pipkorn et al. observed that levocabastine use resulted in significant reduction in symptoms compared with placebo.20 Levocabastine has also shown better efficacy than sodium cromoglycate, antazoline/naphazoline, and oral terfenadine.24
Allergic conjunctivitis may be accompanied by rhinitis (rhinoconjunctivitis); in these cases, oral or nasal antihistamines may be prescribed. However, systemic antihistamines are reported to produce side effects such as ocular surface damage from decreased tear volume and tear flow and sedation.25,26
Despite the advancements in drug delivery and receptor action, antihistamines are still unable to block all manifestations of allergic inflammation because histamine is not the only mediator released by mast cells during the allergic response. Other mast cell-derived mediators such as proteases, lipid-derived mediators, and cytokines also initiate a cascade of events that culminates in conjunctival vasodilation, increased conjunctival vascular permeability, and migration and infiltration of inflammatory leukocytes to the ocular surface.16 Because the mast cell is the primary site of release of these mediators, an alternative therapeutic approach was adopted, resulting in the development of a drug class known as mast cell stabilizers.
Mechanisms of Action of Mast Cell Stabilizers in Ocular Allergy
The mast cell is pivotal in the inflammatory cascade in allergic eye disease. Inhibition of mast cell degranulation is an important target for reducing the acute symptoms of the active disease.27 Mast cell stabilizers offer sustained and prolonged efficacy, possibly via the inhibition of mast cell cytokinemediated events on proximal cells such as the upregulation and expression of adhesion molecules and ocular epithelial cell-mediated chemokine release.16 Mast cell stabilizers were initially thought to inhibit mast cell degranulation by inhibiting calcium influx; further research has suggested they may also act by inhibiting eosinophil, neutrophil, and monocyte activation as well as blocking the release of pro-inflammatory cytokines from inflammatory cells.28 By preventing the degranulation of mast cells and the release of preformed and additional newly formed mediators, they potentially impact both the early- and late-phase reactions.8 Common mast cell stabilizers such as lodoxamide, cromolyn sodium, and nedocromil sodium have proven efficacy in the treatment of SAC.29–33 While all mast cell stabilizers reduce hyperemia, itching, and irritation, they may vary in their efficacy in relieving such signs and symptoms of SAC.8,34
Mast cell stabilizers offer the theoretical advantage of inhibiting the release of all mediators at the cellular level; however, they may not

completely inhibit all mast cell-mediated reactions. Further, in some circumstances, they may not be as effective as antihistamines for symptomatic relief as their onset of action is relatively slow and complete effects may not be seen for up to 3 weeks.35 In a comprehensive review of ocular allergic disease, Bhargava et al. state that given their mechanism of action, mast cell stabilizers would be most effective given prophylactically.36 This is attributed to the need for stabilization of mast cells prior to the degranulation triggered by the allergic reaction; they require a long loading period and must therefore be applied routinely for several weeks before expected exposure for optimal benefit—this may lead to decreased patient compliance.8
Mechanism of Action of Dual-Acting Agents in Ocular Allergy
Pure antihistamines and mast cell stabilizers have limitations when prescribed alone—antihistamines target H1 receptor-specific activity, while mast cell stabilizers have a slow onset of action and work best prophylactically. Dual-acting mast cell stabilizers-antihistamines, however, provide both mast cell stabilizing and antihistaminic activity and are therefore more commonly used in the treatment of signs and symptoms of SAC. These drugs work by stabilizing mast cells and antagonizing the H1 receptor; thus, the mast cell stabilizing action provides an added benefit over pure antihistamines.12 This dual-acting class includes drugs such as bepotastine, azelastine, epinastine, ketotifen, alcaftadine, and olopatadine (see Figure 2).
Phase III trials of bepotastine 1.5 % solution have demonstrated its efficacy and tolerability in the treatment of the signs and symptoms associated with allergic conjunctivitis.37,38 Pooled data from these two placebo-controlled, conjunctival allergen challenge (CAC) trials that randomized 157 patients to receive either bepotastine (n=78) or placebo (n=79) were also analyzed.39 Ocular itching, the primary efficacy measure, was significantly reduced in all patients receiving bepotastine compared with those receiving placebo (p<0.0001 for all time points). Furthermore, the reported adverse events in both trials were generally mild—with no patient experiencing a serious or severe adverse event, and no event considered related to the drug. After a CAC, bepotastine 1.5 % solution produced statistically significant reductions in individual nonocular symptoms including nasal congestion, rhinorrhea, ear or palate pruritus, and nasal pruritus. Each of these symptoms were quantified on a 0–4 scale (0=none; 4=severe), and a summed nonocular composite symptom score was calculated on a 0–16 scale. These reductions in symptoms were observed soon after instillation lasting for at least 8 hours, with the greatest reduction seen for nasal congestion and rhinorrhea (p≤0.05).40
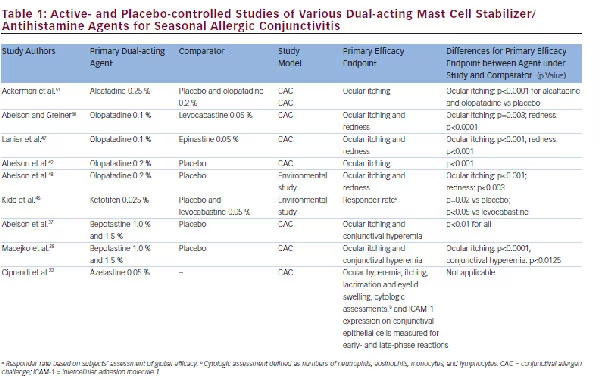
The efficacy of azelastine has also been shown in a randomized, double-blind, placebo-controlled, two-part CAC clinical trial.22 Azelastine use also caused a dose-dependent inhibition of the development of itching of the eyes and conjunctival redness in 24 patients.41 Similarly, trials have also demonstrated the efficacy of other dual-acting mast cell stabilizer-antihistamine formulations versus placebo in the treatment of allergic conjunctivitis by using CAC models.38,42 Table 1 provides details on a number of active- and placebo-controlled trials on dual-acting agents. These trials either use CAC paradigms or are studies based on environmental models. The CAC model accurately reproduces the signs and symptoms of allergic conjunctivitis by replicating the natural disease process. Environmental allergic conjunctivitis models, on the other hand, are less precise and results may be confounded due to high variability in pollen levels, subject exposure time to the environment, and patient study medication compliance.
Ketotifen, a benzocycloheptathiophene agent, has been approved in the US for the treatment of ocular itching associated with allergic conjunctivitis.43 It is a noncompetitive, H1 receptor antagonist with multiple mechanisms of action: it stabilizes mast cells, inhibits platelet activating factor, and serves as an eosinophil inhibitor. While ketotifen has no effect on serotonin 5HT, adrenergic, or dopaminergic receptors, it has demonstrated a direct inhibitory effect on eosinophil oxidative metabolism.44,45 In a study comparing the efficacy and safety of ketotifen 0.025 % to that of levocabastine 0.05 % in the treatment of SAC, 519 patients with SAC were randomly assigned to ketotifen, placebo, or levocabastine twice daily in each eye for 4 weeks. Patients were assessed at followup (days 5–8) and end of study (days 25–31) visits.46 The responder rate, based on the subjects’ assessment of global efficacy at the follow-up visit, was the primary efficacy variable. Ketotifen produced significantly better outcomes than levocabastine for relief of the signs and symptoms of SAC at both the follow-up and termination visits (p<0.05). Olopatadine, another dual-acting mast cell stabilizer-antihistamine, has been approved by the US Food and Drug Administration (FDA) for the treatment of the signs and symptoms of allergic conjunctivitis. The safety and efficacy of olopatadine has been shown in active- and placebocontrolled trials.47–49 The efficacy of olopatadine 0.1 % was compared with levocabastine 0.05 % in a CAC study.50 Subjects were bilaterally challenged with an allergen 27 minutes after topical drug administration, and the first post-challenge assessment was carried out 3 minutes later, or 30 minutes after instillation of the drug. Ocular symptoms were graded on a standard 0–4 scale. Ocular itching severity for olopatadine 0.1 % was significantly lower than that for levocabastine 0.05 % at 3 minutes and 10 minutes post-challenge (p<0.001). Further, grading of ocular redness severity significantly favored olopatadine (p<0.0001). Olopatadine has also been shown to demonstrate statistically significant improvement compared with epinastine in a study using the CAC model.47 In a placebo-controlled CAC study evaluating the efficacy of olopatadine 0.2 % in reducing the signs and symptoms of allergic conjunctivitis in 90 patients,49 olopatadine 0.2 % was found to be significantly more effective than placebo in the treatment of ocular itching at 15 minutes and 16 hours (p<0.001 for both). A 10-week, randomized, placebo-controlled trial to determine the efficacy and safety of 0.2 % olopatadine QD in the treatment of allergic conjunctivitis or rhinoconjunctivitis was conducted in 260 patients.48 This environmental study assessed ocular signs and symptoms in terms of frequency (whole-unit scale from 0 to 5) and severity (half-unit scale from 0 to 4); grass pollen counts were also assessed daily. Analyses of ocular itching and redness showed that when grass pollen counts were high, a respective 21 % and 14 % of patients in the olopatadine 0.2 % group assessed the frequency of ocular itching and redness as >grade 2 compared with 47 % and 31 % of patients in the placebo group (p<0.001 for ocular itching; p<0.003 for redness, respectively). The FDA approved alcaftadine 0.25 % in 2011 as a dual-action agent for the prevention of itching associated with allergic conjunctivitis. In a phase IV double-masked, placebo, and active controlled head-to-head CAC study, alcaftadine 0.25% was compared with olopatadine 0.2 %. The primary efficacy measure was ocular itching evaluated by the subject at 3, 5, and 7 minutes post-challenge. Duration of action was measured at 16 and 24 hours postinstillation of the study medications. One hundred and fifteen subjects completed the study. For the primary measure of ocular itching, alcaftadine 0.25 % and olopatadine 0.2 %, were statistically superior to placebo at all three measured time points for both the 16- and 24-hour measures (p<0.0001). Eyes treated with alcaftadine 0.25 % had numerically lower mean ocular itching scores than eyes treated with olopatadine 0.2% at every time point, and this difference was statistically significant at the 3-minute time point 16 hours postinstillation (p=0.026). Also it was noted that only alcaftadine 0.25% provided statistically significant relief in chemosis at every time point in the 24 hours post instillation.51
Animal studies have also highlighted potential advantages of alcaftadine compared with olopatadine. Ono and Lane compared the abilities of each agent to modify epithelial cell changes associated with allergic conjunctivitis during late-phase reactions in a modified CAC model.52 Assessments were carried out for acute-phase (15 minutes) and late-phase (24 hour) responses in sensitized mice challenged with topical allergens and treated with either drug. Allergy symptoms (redness, itch, tearing, and edema) and conjunctival mast cell numbers were measured, along with eosinophil numbers and tight junctional protein expression for the late phase. While similar efficacy profiles and mast cell numbers were observed for both treatments, alcaftadine-treated mice had significantly lower conjunctival eosinophil infiltration. Allergen challenge caused a significant decrease in junctional protein expression; this decrease was prevented by alcaftadine but not by olopatadine. The authors postulated that olopatadine might not be as effective as alcaftadine. Another animal study also showed that topical ophthalmic olopatadine use may result in significantly decreased tear volume, an important factor for consideration for practitioners choosing a topical medication for patients with SAC.53
Mechanism of Action of Corticosteroids in Ocular Allergy
In contrast to mast cell stabilizers and antihistamines, corticosteroids exert an effect at a point upstream in the arachidonic acid pathway and therefore may provide broader therapeutic effects. Corticosteroids exhibit a number of nonspecific anti-inflammatory effects. They interact with the cytosolic
glucocorticoid receptor (GR) and create a ligand-receptor complex. This complex migrates into the cell nucleus and upregulates the expression of anti-inflammatory regulators such as mitogen-activated protein kinase phosphatase (MKP)-1,54 while simultaneously repressing the expression of pro-inflammatory proteins including inflammatory cytokines, and matrix metalloproteinases (MMPs) by inhibiting transcription factors such as nuclear factor κB (NF-κB) and activator protein 1 (AP-1); these actions affect the entire allergic inflammatory process.36,55,56 Corticosteroids also modulate mast cell production by affecting various immune mediators including IL-3, IL-4, and ICAM-1.18 Further, the inhibition of phospholipase A2 prevents the induction of the arachidonic acid cascade (see Figure 1). This stops the formation of subsequent inflammatory mediators such as prostaglandin E2 (PEG2).28,57 Corticosteroids have been shown to inhibit leukocyte migration and adherence, stabilize lysosomal membranes, modulate lymphocytic activities, and inhibit histamine synthesis and arachidonic acid release. They also decrease capillary permeability, and fibroblast and capillary permeation.57,58 Additionally, corticosteroids are responsible for directly inducing eosinophil apoptosis, suppressing the synthesis and release of eosinophil survival factors, and stimulating their engulfment by phagocytic cells.59 The reason corticosteroids are efficacious is that they block most inflammatory pathways—both GR-mediated and non-GR-mediated—in the allergic reaction.
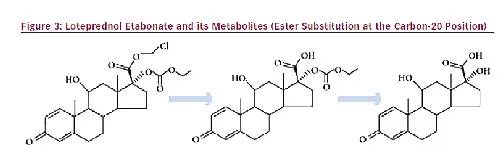
Conventionally, the prescription of corticosteroids has been reserved for more serious or advanced forms of allergic conjunctivitis.5 This is possibly because of concerns that prolonged corticosteroid use may result in adverse effects such as increased intraocular pressure (IOP) or cataract formation.60 Corticosteroids may have either a ketone or an ester in the carbon-20 (C- 20) position.12 Conventional corticosteroids such as dexamethasone and prednisolone have a ketone group in the C-20 position; this ketone group is believed to be associated with cataract formation.61 The substitution of this ketone group with a chloromethyl ester moiety at the C-20 position led to the development of loteprednol etabonate (LE), the only ester corticosteroid currently approved by the FDA for the treatment of the signs and symptoms of SAC.12 LE is a product of retrometabolic drug design; an inactive metabolite is first converted into an isosteric/isoelectronic analog with structural modifications. After eliciting the desired therapeutic effect, this analog undergoes rapid, predictable metabolism back to the original inactive metabolite.62 Because of this retrometabolic design, LE has several advantages over the C-20 ketone-based corticosteroids. LE is rapidly converted into its inactive metabolites by circulating esterases after exerting its therapeutic effect, resulting in fewer ocular side effects than older C-20 corticosteroids such as prednisolone (see Figure 3).63 This unique metabolic profile is particularly important, as numerous studies have shown that LE use results in fewer clinically significant increases in IOP.14,64,65
Cataract formation is another concern associated with extended corticosteroid use. Corticosteroid-induced cataract formation occurs due to nonenzymatic formation of Schiff base intermediates between the steroid C-20 ketone group and nucleophilic groups, such as ε-amino groups of lysine residues of lens proteins. This Schiff base formation is followed by a Heyns rearrangement of the adjacent C-21 hydroxyl group, resulting in stable anime-substituted adducts.61 However, as LE was designed with a C-20 ester rather than a C-20 ketone, it is unable to form covalent adducts via this mechanism, thereby decreasing the likelihood of cataract formation, although other mechanisms of cataractogenesis may occur.
Clinical trials have demonstrated the safety and efficacy of LE suspension 0.2 % and 0.5 % in the treatment of the signs and symptoms of SAC.19,66,67 A placebo-controlled, parallel-group study of 0.2 % LE in patients with SAC examined the efficacy of LE versus placebo in 133 patients receiving either LE or placebo bilaterally four times daily for 42 days.66 Patients were evaluated on days 2 or 3, 7, 14, 28, and 42. The primary efficacy parameters measured were ocular itching (0–4 scale) and bulbar conjunctival injection (0–3 scale). A reduction in severity was seen in both the LE and placebo groups for itching (–3.5 vs –3.1 units) and bulbar conjunctival injection (–1.3 vs –0.9 units) over the first 2 weeks. The LE treatment effects for both itching (0.6) and bulbar conjunctival injection (0.5) were significant (p<0.001). Complete resolution of signs and symptoms at visit 4 (day 14) strongly favored LEtreated patients over placebo-treated patients (54 % and 38 % for itching, and 31 % and 9 % for injection, respectively). No patients experienced an elevation in IOP of ≥10 mm Hg during the course of this study. A similar 42-day QID placebo-controlled trial including 135 patients with SAC also showed the safety and efficacy of LE in the treatment of SAC.67 As in the previous study, primary efficacy variables were ocular itching and bulbar conjunctival injection. Ocular signs and symptoms were measured as supportive efficacy variables. A reduction in severity of ocular itching (–3.4 vs –3.0 units) and bulbar conjunctival injection (–1.5 vs –1.0 units) was observed over the first 2 weeks for the LE and placebo groups, respectively. The treatment effect by these measures was 0.4 and 0.5 units in favor of LE (p≤0.008). Complete resolution of itching at day 14 was observed in 58 % of patients in the LE group and 38 % in the placebo group, whereas 36 % (LE) and 15 % (placebo) of patients experienced complete resolution of bulbar conjunctival injection. Only one patient in each treatment group had an IOP elevation ≥10 mm Hg or greater during the 6 weeks of treatment.
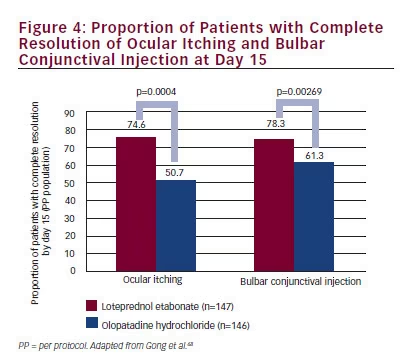
A 0.5 % LE suspension was also found to be effective in the prophylaxis of SAC; this trial also used ocular itching and bulbar conjunctival injection as the primary efficacy parameters.19 In this study, 293 subjects with a history of SAC were treated with either 0.5 % LE or placebo four times daily from the beginning of the allergen season and continuing for a period of 6 weeks. The primary efficacy measure was the composite severity of itching and bulbar conjunctival injection. Secondary efficacy outcomes included investigator global assessments and a secondary composite evaluation, which was the sum of tearing, erythema, chemosis, and discomfort. The primary efficacy endpoint was reached by 94 % and 78 % of patients in the LE and placebo groups, respectively (p=0.001). Similar results were observed for the investigator global assessment (86 % vs 64 %; p< 0.001) and the secondary composite evaluation (77 % vs 68 %), although the differences for the secondary composite signs and symptoms evaluation were not statistically significant (p=0.092). None of the LE-treated patients had an increase in IOP of ≥10 mm Hg, whereas two placebo patients did experience such an increase. Results from these trials suggested that 0.5 % LE was safe and more efficacious than placebo in the treatment of SAC. More recently, the safety and efficacy of 0.2 % LE has been compared with that of a dual-acting antihistamine-mast cell stabilizer. Gong et al. compared the efficacy and safety of 0.2 % LE to that of olopatadine for the treatment of the signs and symptoms of SAC.68 This multicenter, parallelgroup clinical trial randomized 293 Chinese patients to receive either LE suspension 0.2 % QID (n=147) or olopatadine solution 0.1 % BID (n=146) in accordance with the label dosing instructions. Changes from baseline in ocular itching and bulbar injection, the primary efficacy parameters, were evaluated for 15 days. At day 15, the mean (standard deviation [SD]) itching severity in the LE and olopatadine groups was 0.26 (0.469) and 0.72 (0.908) and injection was 0.23 (0.457) and 0.40 (0.539). Mean change from baseline (CFB) differences for ocular itching, −0.46 (95 % confidence interval (CI) −0.59, −0.27) and bulbar injection, −0.19 (95 % CI −0.27, −0.08), were significant in favor of the LE suspension (p≤0.0006). Complete resolution of ocular itching was observed in 74.6 % and 50.7 % patients in the LE and olopatadine groups, respectively, while 78.3 % and 61.3 % patients experienced complete resolution of bulbar conjunctival injection; Figure 4 demonstrates that both outcomes were significantly better with 0.2 % LE suspension compared with olopatadine (p≤0.00269). No major safety concerns were reported, and no patients experienced a clinically significant increase (≥10 mm Hg) in IOP. Results from this study indicate that LE suspension is as safe as olopatadine solution, and possibly more effective in the treatment of SAC.
These results are consistent with the aforementioned placebo-controlled trials for SAC, as well as those reported in a retrospective chart review.69 Ilyas et al. examined safety data from 397 patients being treated with LE 0.2 % for SAC and PAC; 159 of these patients had been using LE continuously for more than 12 months. The review reported that there were no recorded steroid-induced adverse effects in LE-treated eyes, such as posterior subcapsular opacification; additionally, no incidences of steroid-induced IOP increases of ≥4 mm Hg were recorded. While providing good efficacy in the treatment of SAC, the substitution of the C-20 ketone group with an ester results in smaller and fewer rises in IOP and decreased cataractogenic potential, and may alleviate the major concerns associated with corticosteroid use. Newer corticosteroids such as LE appear to maintain the benefits of corticosteroid therapy, while significantly reducing several of the risks associated with traditional corticosteroid use, such as cataract formation and increased IOP. However, isolated cases of increased IOP with LE 0.2 % may still occur, in particular in patients with long axial length, a known risk factor for steroid-induced IOP elevation.70,71
Development of Selective Glucocorticoid Receptor Agonists in Ocular Allergy
While newer corticosteroids such as LE substantially reduce the risk for increased IOP and cataractogenesis, there is a continued need for molecules that can provide the potent anti-inflammatory effects of corticosteroids without having the associated side effects. A greater understanding of the underlying molecular mechanisms of GRs has led to the development of a new class of drugs—selective GR agonists (SEGRAs). SEGRA molecules, although quite diverse, are structurally distinct from steroids and NSAIDs. Available pre-clinical data in the literature support the concept that antiinflammatory effects versus corticosteroid-associated side effects could be functionally separated with appropriate GR ligands.72–78 The hypothesis is that SEGRAs bind to GR and impose a unique conformation to the GR— different from that imposed by corticosteroids. The altered conformation results in a different profile of GR-mediated effects on intracellular signaling pathways. This leads to the targeting of anti-inflammatory pathways, while demonstrating low potential for effecting pathways associated with adverse events observed with corticosteroids. SEGRAs therefore provide an entirely different and more targeted mode of action to steroids with a more favorable safety profile. The clinical utility of a SEGRA compound, mapracorat, is under investigation for the treatment of skin and ocular inflammatory conditions, including allergic conjunctivitis.
Conclusion
The available treatment options for AC vary based on their mechanisms of action. Antihistamines, while offering good efficacy, exert their effects only at the level of the H1 receptors. Mast cell stabilizers are an effective alternative; however, their efficacy is realized when they are used prophylactically prior to the exposure to the offending allergen. Dual-acting mast cell stabilizer-antihistamines are preferred, and studies have shown they are more effective than either agent is alone. Corticosteroids exert effects on the entire inflammatory cascade, and are the most potent option available for the treatment of the signs and symptoms associated with SAC. Some clinicians reserve the use of corticosteroids for severe allergy owing to concerns about their potential to increase IOP. LE, a C-20 ester corticosteroid, provides similar potency to that of ketone corticosteroids with less effect on IOP. In one currently published environmental study, LE offered better efficacy than the dual-acting agent olopatadine.68 Further comparative studies are warranted. Despite the wide range of treatments available, it is difficult to identify which drug or drug class is superior to another due to differences in study designs and outcome measures. However, we conclude that compared with antihistamine-mast cell stabilizer agents, corticosteroids produce a more comprehensive inhibition of the allergic inflammatory cascade. The C-20 ester structure of LE minimizes the potential for the ocular side effects of most concern—increased IOP and c ataract formation—and therefore may be a better choice. Finally, preliminary results from studies with SEGRAs are promising and suggest that these molecules may emerge as the treatment of choice for AC; however, data from large multicenter trials are awaited.







