Presbyopia is a progressive, age-related condition that reduces the eye’s ability to focus on near objects, due to gradual lens thickening and decline in its elasticity and accommodative capacity, and represents the first stage of the dysfunctional lens syndrome.1–3 Presbyopia typically affects individuals aged >40 years and is highly prevalent, affecting nearly 2 billion people worldwide and 128 million individuals in the USA alone.4 Globally, the prevalence of presbyopia is expected to increase further as the population grows and life expectancy rises.4 Although the American Optometric Association had classified presbyopia based on the phase of decline in accommodation, as premature, incipient, functional, absolute or nocturnal,5,6 no specific qualitative or quantitative measures were provided. More recently, a consensus-based classification of presbyopia into mild, moderate or advanced severity using characteristics such as age, behaviours and distance-corrected near visual acuity (DCNVA) has also been proposed.6
Loss of near visual acuity can negatively impact a person’s daily activities, quality of life and emotional well-being.7–12 Moreover, progressive depletion of the remaining accommodative ability in individuals with advanced or absolute presbyopia impairs their intermediate vision, which can further hinder daily activities and social interactions.5 Uncorrected presbyopia not only results in difficulty performing daily activities requiring near (e.g. reading, writing) and intermediate (e.g. computer work, cooking) vision, but can also cause symptoms such as headaches and eye strain,8,13 and cause a sense of diminished accomplishment.7–10,12 Standard treatments for presbyopia include corrective glasses or contact lenses, as well as corneal- or lens-based surgical procedures.14 However, none of these methods can fully restore the dynamic range of accommodation of the lens.1 Cost, risks and benefits, and patient goals should be measured for each available modality.
Until recently, no pharmacological treatments were approved for presbyopia. In October 2021, pilocarpine hydrochloride (HCl) ophthalmic solution 1.25% (Vuity™; Allergan, an AbbVie Company, North Chicago, IL, USA) was approved in the USA for the treatment of presbyopia in adults.15 This once-daily treatment is available in a proprietary vehicle using pHast™ technology that allows rapid equilibration to the physiological pH of the tear film.15–18 This article reviews the mode of action of pilocarpine HCl in the treatment of presbyopia, the preclinical and clinical evidence supporting the therapy, and its importance as the first and, at the time of writing, only topical agent approved for the treatment of presbyopia. Also discussed are the value of pilocarpine HCl ophthalmic solution 1.25% to individuals with presbyopia in providing improved functional vision, without detriment to their distance vision, as well as the inconvenience of glasses, contact lenses or reliance on coping mechanisms.
Presbyopia: Condition burden and unmet need in the treatment landscape
In addition to the humanistic burden, presbyopia incurs a substantial societal cost, particularly in lower- and middle-income countries, where rates of correction have been reported to be as low as 6%.9,19,20 In fact, the burden of uncorrected and undercorrected presbyopia on global gross domestic product was estimated at US$11,023 billion in 2011.19 Fortunately, appropriate correction of presbyopia can dramatically increase productivity,21 and it has been projected that universal correction of presbyopia to levels similar to those observed in parts of Europe (96%) could reduce the financial burden by almost 90% (i.e., US$1,390 billion).19
In developed nations, broad accessibility of corrective devices such as glasses means that optimal correction of presbyopia is often overlooked, and potential alternative treatment options may not be considered.1 Consequently, management of presbyopia remains suboptimal due to a lack of treatments that are effective and yet convenient, non-invasive and relatively risk free. In addition to the fact that non-pharmacological treatments fail to restore natural accommodation of the lens, corrective glasses are often considered inconvenient due to their appearance, psychosocial impacts (e.g. feeling/looking older) and/or impact on people’s daily activities (among others).13,22 During concept elicitation interviews with individuals who used prescription or over-the-counter reading glasses for presbyopia management (n=20), participants most commonly reported inconvenience related to forgetting their glasses and being unable to read important documents at work, for example.13 The same interviewees also reported that glasses caused strained or tired eyes and limited their vision at varying distances.13 Contact lenses can also cause symptoms such as dry or tired eyes, itching and blurring,23 and may limit daily activities.22 In addition, certain types of glasses are associated with blurred peripheral vision and impaired depth perception, which may increase the risk of falls or accidents,14 while contact lenses have the potential risk of ocular surface infections.24,25 Surgical interventions are not without limitations and may not be suitable for some patients. These limitations include continued requirement for spectacles for some patients/activities and the potential for complications such as impaired intermediate or distance vision, corneal haze, dysphotopsia, decreased contrast sensitivity and infections.24,26–28
Given the above limitations, it is perhaps not surprising that, when asked to rank different treatments, individuals affected by presbyopia have expressed a preference for eye drops over glasses, contact lenses and surgery.13 Various topical miotics such as pilocarpine and carbachol have been studied for their potential to provide a glasses-free, minimally invasive alternative to presbyopia management.24 However, anatomical and physiological barriers to effective delivery have presented a challenge.17 Furthermore, their use has been historically associated with side effects such as ocular discomfort, redness, inflammation, headache and vision blur.17,24,29,30 The current literature thus highlights an unmet need in the presbyopia treatment landscape.
Pilocarpine hydrochloride 1.25%: Mechanism of action and pHast™ technology
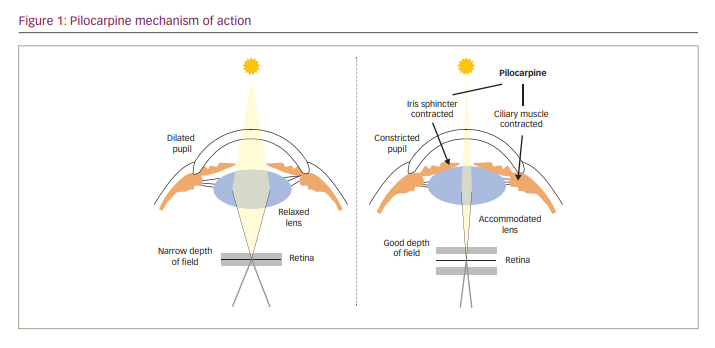
Pilocarpine is a cholinergic muscarinic receptor agonist that acts through M3 muscarinic receptors located on smooth muscles, such as the iris sphincter and ciliary muscle.31 Activation of these receptors leads to contraction of the iris sphincter, constricting the pupil (miosis) and creating a pinhole effect (Figure 1). Reduction of the pupil diameter increases the depth of focus and allows for greater ranges in uninterrupted near and intermediate vision that cannot be achieved using spectacle lens correction.18,32 Meanwhile, contraction of the ciliary muscle changes the lens thickness, stimulating accommodation to improve near vision.18,32–34 Pilocarpine HCl has long been used to decrease intraocular pressure in patients with glaucoma, but its use in this indication has declined steadily due to side effects such as brow ache, headaches and vision blur, along with the emergence of other (more effective) glaucoma medications.35 More recently, however, pilocarpine HCl has been investigated as a treatment for presbyopia, due to its ability to enhance both depth of focus and accommodation.18,32
Pilocarpine HCl ophthalmic solution 1.25% is an optimized formulation in a proprietary vehicle that relies on pHast™ technology. The product was designed specifically for the treatment of presbyopia (based on the unmet need for a non-invasive pharmacological eye drop) and reformulated to improve bioavailability and tolerability.13,17
A low pH (~4) is required to stabilize pilocarpine in generic ophthalmic solution, due to a high susceptibility of pilocarpine to hydrolysis at physiological pH (~7).17,36 However, pilocarpine is protonated (or ionized) at low pH, leading to poor transcorneal permeation and, consequently, limited bioavailability at target tissues.17,37 The low pH of generic pilocarpine ophthalmic solution may also cause some of the adverse effects experienced by patients with glaucoma.36 With this in mind, a re-engineered ophthalmic solution was designed to equilibrate rapidly to the pH of the ocular surface to improve bioavailability and reduce side effects such as ocular discomfort and vision blur.17
Preclinical and phase I studies
In vitro studies and a phase I clinical trial evaluated the properties of the pHast™ technology-based formulation of pilocarpine HCl ophthalmic solution 1.25%, compared with a generic formulation of pilocarpine HCl 1.0% commercially available for the treatment of glaucoma.17 The first in vitro study compared the time required for the two formulations to equilibrate with the pH of simulated tear fluid and found that the pHast™ technology-based formulation underwent faster pH equilibration, achieving a final pH of 6.43 within 1 minute (starting pH: 4.35).17 In contrast, the generic formulation, initially at pH 4.41, increased to pH 4.44 by the end of the 10-minute period.17 In a separate study using cultured human corneal epithelial cells, increasing the pH from 4.3 to 7.96 enhanced corneal permeability to pilocarpine, which was attributed to an increase in the non-ionized proportion of pilocarpine on the ocular surface at physiological pH.38 Faster pH equilibration of the pHast™ technology-based formulation was thus expected to improve bioavailability in clinical studies and translate into greater or longer iris sphincter contraction and/or ciliary muscle activation, and increased depth of focus.38
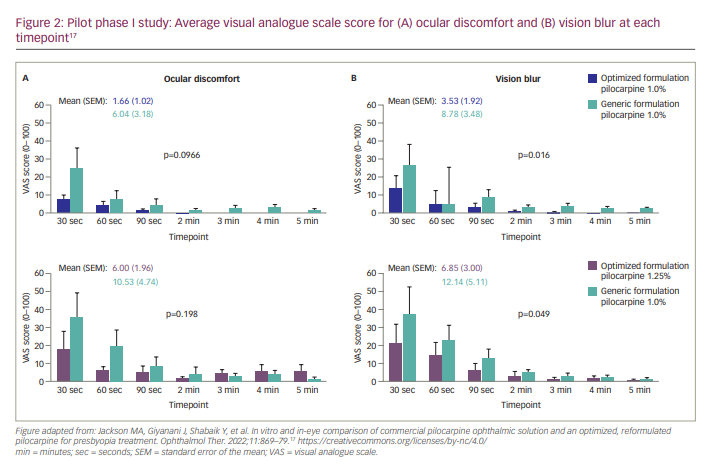
A phase I clinical study (Table 1) evaluated the pHast™ technology-based and generic formulations in five healthy adults and found that, compared with the generic formulation, the pHast™ technology-based formulation demonstrated numerically less ocular discomfort (p=0.0966) and statistically significantly less vision blur (p=0.016) over 5 minutes following administration (Figure 2).17 Only one adverse event (AE; eye pressure/pain) was reported with the pHast™ technology-based formulation, compared with eight AEs with the generic formulation (most commonly blurred vision).17 Based on these findings, the pHast™ technology-based formulation was expected to improve comfort, cause less vision blur and provide a well-tolerated alternative to generic pilocarpine ophthalmic solutions.17
Phase II studies
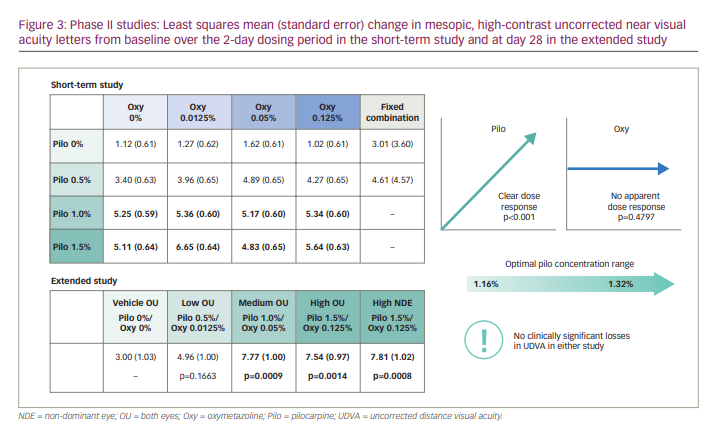
To determine the optimal concentration of pilocarpine HCl for improvement of near vision in individuals with presbyopia, two concurrent dose-ranging phase II studies (one short-term and one extended study; Table 1) were conducted in emmetropic individuals affected by presbyopia.32 Both studies were randomized, vehicle-controlled trials that evaluated various combinations of pilocarpine HCl and oxymetazoline (a selective α1-adrenergic receptor agonist that causes pupil dilation and vasoconstriction), as it was hypothesized that the addition of oxymetazoline might prolong the duration of action of pilocarpine HCl and potentially reduce the rate and severity of AEs.32
In the short-term study, 163 participants, stratified by baseline uncorrected near visual acuity (UNVA; ≤20/80 versus >20/80), were randomized 1:1:1:1 to one of four treatment groups based on the pilocarpine HCl concentration (0%, 0.5%, 1.0% or 1.5%).32 Each group received their assigned pilocarpine HCl eye drop in the non-dominant eye and a concomitant oxymetazoline eye drop of increasing concentration (0%, 0.0125%, 0.05% and 0.125%) in four 2-day dosing periods (each separated by a 7- to 21-day washout).32 A fifth 2-day dosing period using a fixed combination of pilocarpine HCl 1.0%/oxymetazoline 0.125% followed.32 Treatment was administered once daily in the non-dominant eye, with vehicle (placebo) administered in the dominant eye.32 The primary endpoint was the average change from baseline in mesopic UNVA in the non-dominant eye over the 2-day dosing period.32
In the extended study, 151 participants, stratified by age and iris colour, were randomized 1:1:1:1:1 to one of four fixed combinations of pilocarpine HCl/oxymetazoline treatments (0%/0% [vehicle], 0.5%/0.0125%, 1.0%/0.05%, 1.5%/0.125%) used once daily for 28 days in both eyes,32 while a fifth group used fixed-combination pilocarpine HCl 1.5%/oxymetazoline 0.125% in the non-dominant eye and vehicle in the dominant eye.32 The primary endpoint was the mean change from baseline in mesopic, high-contrast UNVA letters in the non-dominant eye at day 28.32
The short-term study demonstrated a statistically significant dose-dependent response to pilocarpine HCl (i.e. increase in average mesopic, high-contrast UNVA from baseline; p<0.001), and polynomial regression modelling determined the optimal pilocarpine HCl concentration to be between 1.16% and 1.32%.32 Both the pilocarpine HCl 1.0% and 1.5% groups had a similar mean change in mesopic, high-contrast UNVA letter gain of approximately five letters across all oxymetazoline concentrations (Figure 3). In contrast, the oxymetazoline concentration did not have a significant impact on the aforementioned outcome (p=0.4797),32 indicating that oxymetazoline did not contribute to the unfixed combination’s efficacy. Results from the extended study supported these findings (Figure 3), demonstrating robust efficacy of the fixed combination containing pilocarpine HCl 1.0%, with a statistically significant percentage of participants maintaining a ≥3-line improvement in mesopic, high-contrast UNVA from baseline for up to 8 hours.32 Pilocarpine HCl concentrations of up to 1.5% had acceptable safety and tolerability profiles in both studies, and addition of oxymetazoline did not improve safety, compared with pilocarpine HCl alone.32 The most common treatment-related treatment-emergent AE (TEAE) reported in the extended study was headache, which was observed in all treatment groups and was mostly mild in severity. Most cases of headache started approximately 1 hour post-dose and had resolved by hour 3. Notably, there were no reports of clinically significant losses in uncorrected distance visual acuity (UDVA) in either study.32 Based on these results,32 together with preclinical and phase I findings,17 the optimized formulation of pilocarpine HCl 1.25% in the pHast™ technology-based formulation was evaluated in phase III studies.
Phase III studies
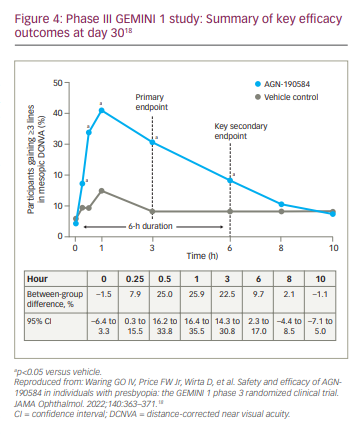
GEMINI 1 and GEMINI 2, two multicentre, double-masked, randomized, controlled, parallel-group, phase III studies, evaluated the efficacy and safety of pilocarpine HCl ophthalmic solution 1.25% in individuals with presbyopia (Table 1).18,39 Eligible participants were aged 40–55 years and in good general health, and had objective and subjective evidence of presbyopia. Additional eligibility criteria are summarized in Table 1. Participants were randomized 1:1 to pilocarpine HCl 1.25% or vehicle administered once daily in both eyes for 30 days. The primary and key secondary endpoints were the proportion of participants gaining ≥3 lines in mesopic (10–11 lux at the target), high-contrast, binocular DCNVA on day 30, hour 3 and hour 6, respectively, without losing >5 letters in corrected distance visual acuity (CDVA).18,39 Achievement of ≥3-line improvement is a rigorous efficacy endpoint required by the US Food and Drug Administration (FDA) for measuring the effect of new compounds on visual acuity.18,40
GEMINI 118 met the primary and key secondary endpoints; on day 30, the percentage of participants who gained ≥3 lines in mesopic, high-contrast, binocular DCNVA without losing >5 letters CDVA was 30.7% with pilocarpine HCl 1.25% and 8.1% with vehicle at hour 3 (between-group difference, 22.5%; 95% confidence interval [CI], 14.3 to 30.8; adjusted p<0.001), and 18.4% and 8.8%, respectively, at hour 6 (between-group difference, 9.7%; 95% CI, 2.3 to 17.0; adjusted p=0.01) (Figure 4). Improvements were observed across all prespecified subgroups based on age, baseline binocular DCNVA, iris colour and emmetrope status, and statistically significant improvements in the overall study population were observed as early as 15 minutes following administration of pilocarpine HCl 1.25%. Although the between-group difference in ≥3-line improvement in mesopic DCNVA did not remain statistically significant at hour 8, pilocarpine HCl 1.25% demonstrated statistical superiority over vehicle in other clinically relevant efficacy measures up to hours 8 or 10 on day 30, including changes from baseline in mesopic DCNVA and photopic distance-corrected intermediate visual acuity (DCIVA) letters.18 The secondary endpoint of functional vision was also met, with up to 93% of participants achieving 20/40 or better in photopic, high-contrast, binocular DCNVA, which is equivalent to being able to read 6-point fonts from a distance of 14 inches (~35 cm).18
No participants with a mesopic DCNVA gain of ≥3 lines at hour 3 lost >5 letters in mesopic, high-contrast, binocular CDVA, indicating no adverse impact of pilocarpine HCl 1.25% on distance vision, consistent with phase II studies.18 Moreover, treatment was well tolerated, with most treatment-related ocular AEs being mild in severity.18 The most common TEAE was headache, which was mostly (87%) mild and transient, and no participants discontinued the study due to headache.18 Evaluation of pilocarpine pharmacokinetics in 22 of the GEMINI 1 study participants indicated low systemic exposure and rapid clearance of pilocarpine HCl 1.25% from the circulation, without accumulation.41 Systemic concentrations of pilocarpine were lower than observed with other pilocarpine eye drops available on the market.41
Pooled analyses of GEMINI 1 and GEMINI 2 have been performed to evaluate the efficacy and safety of pilocarpine HCl 1.25% and related patient experience in a larger population sample. In total, 750 participants were randomized across the two trials: 375 to pilocarpine HCl 1.25% and 375 to vehicle. Similar to the individual GEMINI 1 study,18 pilocarpine HCl 1.25% demonstrated statistical superiority over vehicle in terms of proportion of participants gaining ≥3 lines in mesopic, high-contrast, binocular DCNVA without losing >5 letters of mesopic CDVA at hour 3.39 Moreover, a statistically significantly greater proportion of participants treated with pilocarpine HCl 1.25% experienced a clinically meaningful improvement of ≥2 lines in mesopic, high-contrast, binocular DCNVA (compared with vehicle) at all timepoints up to 10 hours.42 In this pooled analysis, additional endpoints evaluating near and intermediate vision were also statistically significantly improved with pilocarpine HCl 1.25%, compared with vehicle.39,43 Among those with moderate or advanced presbyopia, a statistically significantly higher proportion of participants experienced ≥2-line and ≥3-line gains in photopic DCNVA and DCIVA with pilocarpine HCl 1.25% than with vehicle.44 Additionally, 74–94% of participants treated with pilocarpine HCl 1.25% achieved functional vision (20/40 or better in photopic, high-contrast, binocular DCNVA) on day 30, hour 1, depending on presbyopia severity at baseline (20/50, 20/63 and 20/80).45 As expected, participants with less severe presbyopia (20/50) were more likely to achieve functional vision (94% at day 30, hour 1).45 Furthermore, up to 42% of participants with mild or moderate presbyopia treated with pilocarpine HCl 1.25% achieved 20/20 or better in DCNVA without losing >5 letters of CDVA on days 14 and 30.46
Pilocarpine HCl 1.25% was well tolerated, with no serious TEAEs and few (n=6, 1.6%) TEAEs leading to treatment discontinuation.39 Headaches were mostly mild and transient (84%), requiring no treatment, and there were only two discontinuations due to headache TEAEs in the pilocarpine HCl 1.25% arm.39 Overall, these findings suggest that pilocarpine HCl 1.25% is a well-tolerated and efficacious eye drop for presbyopia, improving functional near and intermediate vision throughout the 30 days, without compromising distance vision.18,39
Real-world safety data
Although no serious ocular AEs (e.g. retinal detachment)47 were reported in the phase III trials, safety data were reported for only 30 days, and eligibility criteria excluded patients with a higher risk of retinal detachment, such as those with a history of cataract surgery or pre-existing retinal pathology.18 In terms of real-world data, retinal detachment was recently reported in two adults who received pilocarpine HCl ophthalmic solution 1.25% for presbyopia.47 The first was a 47-year-old male who was prescribed pilocarpine HCl 1.25% ophthalmic solution for presbyopia following a routine ocular examination by his eye care provider. At that visit, his refraction was +0.25+1.00×180 in the right eye and +0.25+0.75×010 in the left, with an add of +1.50D in both eyes. One month later he presented with flashes and floaters, which he had first noticed 3 days after pilocarpine treatment was initiated. The individual had a history of cataract surgery in the right eye (performed 3 months earlier) but otherwise reported no trauma or ocular history. Vision was 20/20 in the right eye and 20/25 in the left at presentation. Dilated examination revealed an inferotemporal retinal detachment and retinal tear in the right eye. The left eye also demonstrated a retinal detachment with a horseshoe tear. He underwent staged retinal detachment repair with vitrectomy in the left eye, followed by a scleral buckle with vitrectomy in the right eye 1.5 weeks later, and best-corrected vision was 20/20 in both eyes at the most recent follow-up. The second individual was a 46-year-old male with a history of dry eye and contact lens use. He was prescribed pilocarpine HCl 1.25% after a routine eye examination in which his best-corrected visual acuity was 20/25 and 20/30 in the right and left eyes, respectively. The manifest refraction was –4.00+1.50×161 in the right eye and -3.25+1.00×180 in the left. The patient was phakic in both eyes, with no vitreous detachment in either eye. Dilated fundus examination revealed cobblestone retinal degeneration in the temporal periphery of both eyes. Five weeks after starting pilocarpine HCl 1.25%, the individual noticed a nasal visual field defect in his left eye that progressed overnight to include his central vision. Dilated examination revealed a superior retinal detachment from the 11 to 3 o’clock positions, with subretinal fluid extending into the macula. He subsequently underwent retinal detachment repair with a pars plana vitrectomy and, during surgery, was found to have a retinal tear at the 1 o’clock position.
While the authors could not confirm that pilocarpine HCl 1.25% was the cause of retinal detachment,47 these cases do warrant further investigation. Rates of retinal detachment in individuals receiving pilocarpine HCl 1.25% are likely to be rare; however, if there is a causal link, widespread use of this treatment could lead to significant numbers of people being affected. The US label for pilocarpine HCl 1.25% does include a warning on the risk of retinal detachment in susceptible individuals and those with pre-existing retinal disease.15 However, the authors of the case report suggested that physicians should consider the use of a dilated examination before prescribing pilocarpine HCl 1.25% to determine whether individuals (particularly those who are myopic) are at a higher risk of retinal detachment due to risk factors such as lattice degeneration, asymptomatic holes, tears and other retinal pathologies.48,49
Across the GEMINI 1 and 2 trials, 80 participants had previously received laser-assisted in situ keratomileusis,50 and, although their level of correction pre-treatment with pilocarpine HCl 1.25% was not reported, these participants may have been highly myopic. However, because they underwent dilated funduscopic examination and their retinas were deemed healthy, they were allowed to continue in the trial and had no AEs during the 30-day safety reporting period. In addition to undergoing a dilated examination, recipients should be informed of these potential AEs and instructed on how to recognize symptoms such as flashes, floaters and visual field loss.47 Physicians should also ensure that discussions with the patient on the risks, benefits and potential AEs are documented on the appropriate clinical chart.
Patient value
Despite the detrimental impact of presbyopia on quality of life, until recently no validated patient-reported outcome (PRO) instruments were available to assess presbyopia-specific symptoms, impacts, coping behaviours and treatment satisfaction.51 Therefore, two PRO instruments were subsequently developed in accordance with FDA standards, and validated for use in the phase III GEMINI 1 and GEMINI 2 trials: (1) the Near Vision Presbyopia Task-based Questionnaire (NVPTQ), which evaluates vision-related reading ability (performance domain) and satisfaction with vision-related reading ability (satisfaction domain);51 and (2) the Presbyopia Impact and Coping Questionnaire (PICQ), which assesses the impact of presbyopia on non-reading tasks and activities (impact domain), and use of coping behaviours such as squinting or increasing font size (coping domain).52
In GEMINI 1, differences in mean changes from baseline statistically significantly favoured pilocarpine HCl ophthalmic solution 1.25% over vehicle at day 30, hour 3 for all four PRO domains mentioned above.18 Least squares mean (95% CI) differences in change from baseline (pilocarpine HCl 1.25% minus vehicle) were 0.8 (0.6 to 1.1) for NVPTQ performance, 0.8 (0.5 to 1.1) for NVPTQ satisfaction, -0.3 (-0.4 to -0.1) for PICQ impact and -0.5 (-0.6 to -0.3) for PIQC coping scores (adjusted p=0.01 for all). These findings suggest that participants receiving pilocarpine HCl 1.25% experienced greater ability and satisfaction for near-vision reading, lesser presbyopia impact, and a reduction in the use of presbyopia coping mechanisms.18
The patient experience when using pilocarpine HCl 1.25% was also assessed in a pooled analysis of GEMINI 1 and GEMINI 2, wherein participants were invited to complete an online follow-up survey after trial completion.53 Participants in the pilocarpine HCl 1.25% group reported statistically significantly higher mean satisfaction scores (3.3) than those receiving vehicle (2.4; p<0.05), with 89% reporting that they were satisfied with pilocarpine HCl 1.25% treatment. Most participants who received pilocarpine HCl 1.25% were able to stop using their glasses for at least part of the day (79%) or during the entire 10-hour treatment window (76%), and 84% responded that they were likely or very likely to use the eye drop if it became commercially available.53
Summary and conclusion
Pilocarpine HCl ophthalmic solution 1.25% is the first and, at the time of writing, the only FDA-approved topical agent to treat presbyopia in adults. In randomized, controlled clinical studies this non-invasive treatment significantly improved near and intermediate vision without impacting distance vision, and was well tolerated.18,32,39 The proprietary vehicle using pHast™ technology allows rapid equilibration to the physiological pH of the tear film, greater comfort and tolerability, and minimal vision blur, and the increased bioavailability resulting from improved transcorneal permeation may contribute to the duration of action.17
With a once-daily administration, up to 94% of individuals with presbyopia in the phase III trials achieved improved functional vision, and almost one-third achieved the gold standard endpoint of a ≥3-line gain in mesopic, high-contrast, binocular DCNVA (without losing >5 letters in CDVA).18,39,45 Effects were seen within 15 minutes, even in dim lighting conditions, with improved visual performance observed for up to 6–8 hours.18,39 Compared with the vehicle group, participants receiving pilocarpine HCl 1.25% were more satisfied with their near-vision reading ability and experienced a clinically meaningful reduction in use of presbyopia coping mechanisms.18
Overall, the availability of pilocarpine HCl 1.25% is an exciting breakthrough for the millions of Americans with presbyopia, particularly those who want to reduce their dependence on spectacles or contact lenses and do not wish to undergo surgery. The addition of an eye drop to the existing presbyopia treatment landscape means that there are now multiple surgical and non-surgical options that can be used as a single solution or in combination as a multipronged approach, affording individuals the opportunity to find the best customized solutions for their individual needs. Given the increasing global burden of presbyopia, more individuals around the world may eventually be able to use this treatment in the future.







