Avascularity of the cornea optimises its optical performance and also helps confer it immunological privilege.1 Loss of this immune privilege leads to poor survival of corneal grafts.2 Corneal new vessels (NVs) can also lead to oedema, lipid deposition, inflammation and scarring leading to reduced vision.3 Maintenance of corneal avascularity is an active process dependent on the interplay between angiogenic and antiangiogenic factors in the tissue.4 Pathological environments such as hypoxia, inflammation, degeneration, limbal stem cell deficiency, etc. can destabilise this balance and lead to corneal lymph and blood vessel formation, sprouting from the adjacent limbal vasculature. Our understanding of angiogenesis and lymphogenesis come from the early description in 1974 of a corneal angiogenesis model by Gimbrone et al.5 and much later of an animal model of lymphangiogenesis dissociated from angiogenesis.6 Although the presence of blood vessels in the cornea has been well recognised, the role of lymph vessels has only more recently been defined by the work of Collin et al. who used an animal model using radioactive iodine labelled albumin.7 These studies have shown that lymphangiogenesis occurs earlier in the stage of corneal new vessel formation.
It is estimated that the incidence of corneal NVs in the US is 1.4 million.8 Until recently the mainstay of treatment has been using topical steroids, the long-term use of which is restricted by glaucoma and cataract formation.9 Other immunomodulators such as cyclosporine or methotrexate,10,11 laser photocoagulation,12 fine needle diathermy13 and photodynamic therapy (PDT)14 have been described in the treatment of corneal NVs. These treatments, however, have had variable or inconsistent success in addition to the side effects. As the molecular mechanisms of angiogenesis are being better understood and with the advent of antiangiogenic treatments for exudative age-related macular degeneration, the prospect for corneal new vessel management looks brighter. Vascular endothelial growth factor (VEGF) inhibitors, by targeting the angiogenic process at a molecular level can limit or cause regression of corneal NVs with minimal side effects. A recent meta-analysis found that both topical and subconjunctival bevacizumab achieved significant reduction in the area of corneal NVs.15 With the introduction of new treatment modalities comes the challenge of evaluation of effectiveness. Accurate comparison requires adequate quantification of corneal blood vessels.
In Vitro Corneal New Vessel Imaging
Several animal studies have described different in vitro methods of quantification that have been insufficiently validated. Proia et al. used India ink enhancement of blood vessels on histological samples.16 Later, immunofluorescent staining of vascular endothelial cells was utilised to quantify corneal NVs on animal models.17,18 While these experimental in vitro animal models have broadened our understanding of the pathology, they are of little assistance in the day-to-day management of a patient with corneal NVs.
In Vivo Corneal New Vessel Imaging
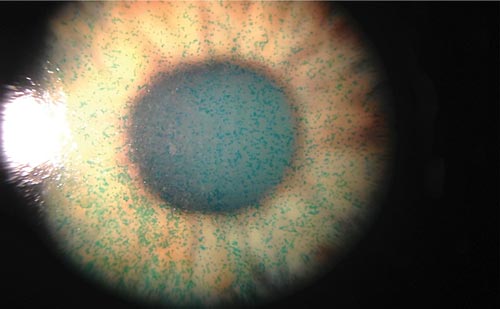
The simplest method of documenting corneal NVs is colour-coded drawings. The reproducibility of this method is limited, especially if different assessors are involved. The traditional method of imaging corneal NVs for documentation purposes is colour photography (see Figure 1). This qualitative approach is prone to inter-observer variation in interpretation. Several studies have attempted to circumvent this problem by devising quantitative analysis of digital colour photos. Hashemiam et al., in their study of comparison of subconjunctival bevacizumab with topical corticosteroids, counted the major thick walled vessels originating from the limbus up to the scar.19 The authors suggested that this was more accurate compared with determination of the area of NVs in a 2D photograph of a dome-shaped structure. There were, however, no standard definitions of a thick walled vessel nor was the matter of reproducibility addressed. Conrad et al. utilised the contrast between blood vessels and stroma of cornea on digital photographs utilising monochromatic light.20 The vessel area was calculated by pixel counting on a montage of non-overlapping adjacent images. Falkvoll described measurement of corneal NVs on still mode video pictures colour images on a mouse angiogenesis model.21 Validity of the method was shown by the high correlation between the morphometric measurements and computerised values on the same vessel structures. Becker described an in vivo method of corneal NVs quantification of both the morphology of and the haemodynamic using intra-vital fluorescence microscopy in New Zealand white rabbits utilising fluorescent rhodamine 6G markers.22 Parke et al. used negative image analysis in a mouse model. Serial image analysis enabled them to ascertain the rate and half-life of the regression of corneal NVs area.23 While quantification of corneal NVs in these animal models have been used effectively in individual studies, the methods have not been reproduced or adapted to the clinical context.
An easily adaptable quantification process of corneal NVs in a clinical scenario was described by Pillai et al.13 They evaluated NVs in terms of the number of quadrants involved, the depth of vessels and whether they were active or quiescent. No details of how the differentiation between activity and quiescence was made are provided. Vassilev also described a similar method of quantification by analysing corneal photographs and determining the number, size and centricity of vessels. This analysis was performed by the same non-blinded reviewer.24 Lai et al. described a grading system of corneal NVs.25 They graded NVs as mild when cornea showed one to six vertical neovascularisation areas, moderate where there are more than 15 vertical vessels and none. There was, however, no standardisation of vertical vessels and hence was prone to subjective interpretations. Importantly, these clinical grading systems may not take into account blood vessels obscured by corneal scar tissue.
Semi-automated Evaluation of Corneal New Vessels
The development of computer software technology led to the evolution of computer-aided methods of quantification of corneal NVs at the turn of the century. Computer-aided semi-automated approaches of analysing retinal blood vessels have been described before.26 However, the noise levels, presence of bright reflections and variations in background illumination of corneal NVs require a different approach to retinal blood vessels. Also, the random pattern of these pathological vessels does not allow pattern recognition. Wu et al. in their study of the anti-angiogenic effect of somatostatin on a mouse corneal angiogenesis model used a semi-automated computer program to quantitatively evaluate the area of neovascularisation.27 The outline of the area of neovascularisation was traced manually on a computer image and the area calculated with image analysis software (Enhance 3.0; MicroFrontier, Des Moines, Iowa) in a blinded strategy. You et al. studied the effect of different doses of subconjunctival bevacizumab on 29 patients with corneal NVs.28 The effect of treatment was quantitatively analysed using Image Pro Plus (Media Cybernetics, Silver Spring, Maryland) software program. This involved manually outlining the area of corneal NVs on a TIFF format digital image, followed by estimation of area. This quantification was carried out in a blinded manner. By comparing the area of corneal NVs before and after treatment, they were able to demonstrate a doserelated regression of corneal NVs.
Quantification of corneal NVs was further refined by Dasterdji et al. in their study on the effect of topical 1 % bevacizumab on corneal NVs in 10 patients.29 They used three parameters to analyse corneal NVs using graphics editing software (Photoshop CS2, Adobe Systems Inc. Berkeley, California) on digital colour images after manually delineating the neovascularisation area. In addition to determining new vessel area, the authors also measured vessel calibre, described as the approximate diameter of a vessel. The third metric described was the invasion area defined as the fraction of corneal area affected by NVs.
In an attempt to circumvent the variability associated with manual interpretations of colour photographs, Tatham et al.30 devised a semiautomated computer software program. This method utilised National Institutes of Health (NIH) image software Image J (http://rsb.info.nih.gov/ ij/). The PC-based software program is designed to be used on any highresolution corneal photograph. The process involves five steps entailing edge detection utilising the Canny edge detection filter,31 automatic detection of limbus using Hough transform search,30 masking and filtering to allow analysis of cornea, region growing by hue to quantify scarred tissue and colour segmentation to identify and trace corneal NVs. This is followed by calculation of the percentage of cornea that is opaque and/or vascularised by pixel counting. This novel technique was tested on 20 patients and compared with the independent assessments of three experienced ophthalmologists. There was a good correlation between clinicians and the software program.
While the latter had better reproducibility compared with clinicians, quality of images, pupil size, density of scar and poorly defined limbus as in vascularisation affected the ability of the software program to perform the analysis. Small vessels were also not detected by the software due to the minimal pixel difference.
Another approach to a semi-automated method of corneal NVs was described by Otoum and Edirisinghe32 utilising the Contourlet Transform principle that takes into account the shape of cornea. Their technique involves manually fixing a limbus on a digital colour image followed by contrast enhancement, noise removal, segmentation of blood vessels and determination of the total area occupied by the blood vessels by pixel counting.
These early attempts at devising a semi-automatic method for corneal NVs quantification have the potential to be a useful aid in clinical practice to monitor response to treatments. A major drawback of these methods is that there is an underestimation of the NVs in presence of scarring as we have shown in our study.33
Fluorescein- and Indocyanine Green Angiography-aided Evaluation of Corneal New Vessel
Research that involves comparing different treatment strategies for corneal NVs requires a more rigorous technique of recognition of blood vessels followed by quantification.
Four decades ago, Easty and Bron34 described using anterior segment fluorescein angiography (FA) to delineate all corneal NVs. Their work on 250 patients has revealed useful insights into the nature of corneal NVs. Similar to our study they also noticed significant differences in the complexity of blood vessels on a FA image compared with clinical examination or colour photographs. Leakage of fluorescein from the pathological blood vessels of cornea was a feature noticed by Easty as well in our study. The physiological blood vessels of retina and the iris did not leak fluorescein dye. Pathological blood vessels have weaker endothelial junctions leading to leaking of the dye.
Fossarello et al. described using fluorescein and indocyanine green angiography (ICGA) to describe the regressive effects of corneal NVs treated with PDT.35 There was no attempt at quantification. We have described a technique of documenting corneal NVs using FA and ICGA.33
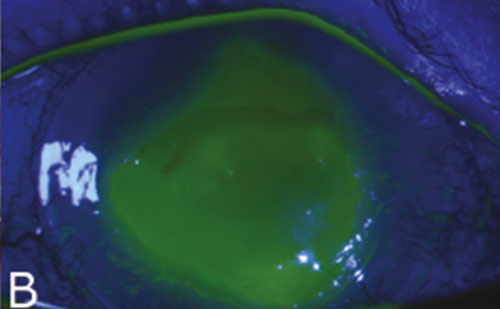
After obtaining digital photographs using standard settings, intravenous indocyanine dye was injected into a peripheral vein as for retinal angiography. Single-frame ICGA photographs were taken every 3 to 5 (now every second) seconds for 3 minutes utilising a scanning laser ophthalmoscope (HRA2; Heidelberg Engineering, Heidelberg, Germany). The process was repeated after injecting intravenous fluorescein. Compared with clinical, colour and FA images, ICGA showed welldelineated vessels even in the presence of scarring (see Figure 2 and 3).
The fluorescence in FA was partially blocked in presence of scarring (see Figure 3). Also, the leakage of fluorescein obscured vessel details. Indocyanine dye being more protein bound is better retained in blood vessels compared with fluorescein dye. Therefore, we used ICGA images for describing vessel parameters. The area of corneal NVs and geometric properties (length, diameter and tortuosity) were determined using a computer program written in numerical computing language (MatLab R14; The Math Works Inc., Natick, Massachusetts). The process involved defining the limbus manually, followed by estimation of pixel resolution.
The corneal vessels were defined manually and enhanced through a Gaussian filter to remove noise, followed by application of selective enhancement filters as originally described by Li et al.36 This was followed by estimating the area by automated pixel counting compared with a predetermined fixed level. Applying this technique, Kirwan et al. have shown that changes in the area of neovascularisation, vessel diameter, distribution of vessel diameters and, in some cases, vessel tortuosity on FA and ICGA provided reliable measures of change following treatment.37 Stereo images of corneal ICGA have the capability of indicating depth of NVs (see Figure 4). Though invasive, with a small possibility of allergic reactions, ICGA-aided quantification of corneal NVs has the potential to be a robust research tool.
Confocal Evaluation of Corneal New Vessels
Confocal imaging of cornea in health and disease has evolved in the last decade although there is a paucity of studies describing corneal blood vessels. This could be due to the technical and interpretation challenges described by Wells et al.38 Yayali used confocal technology to map out early events of cell migration from the limbal microvessels in an angiogenesis model.39 Peebo et al. used confocal technology to image blood and lymph vessels in vivo followed by histopathological correlation of these vessels in a rat angiogenesis model.40 They were able to identify differences between lymph and blood vessels. Lymphatics were characterised by a darker lumen and smaller diameters and lower velocity of flow of leucocytes.
Lymphatic Imaging
The above imaging techniques involve evaluation of vessels with or without lymphatics. The role of lymphatics in corneal angiogenesis has been well recognised.7 Therefore, corneal vessel evaluation strategy should ideally involve lymphatic vessel estimation as well. Yuen et al. used subconjunctival injection of large molecular weight fluorescein isothiocyanate labelled dextran that is preferentially taken up by lymphatics.41 The dye uptake was continuously monitored under a custom-built live imaging system. This technique needs further evaluation for clinical application. Steven et al. used two photon microscopy to assess fluorescence of lymphatics after intrastromal injection of fluorescently labelled LYVE-1 antibody.42 In addition to being tenuous, none of these methods utilised quantification methods.
Corneal blood vessel imaging has evolved significantly from descriptive analysis of colour photographs to computer-aided quantification methods. No single method has emerged as a useful clinical or research tool so far. The 3D shape of cornea and multilevel distribution of blood vessels in the stroma in a random manner makes evaluation of vasculature difficult. To overcome these limitations, we are developing use of stereophotographs allowing measurement of vessels in different planes.