Lamellar procedures have their origins in the 1960s, when they were introduced by Barraquer in Columbia.1 In the 1990s, Pallikaris and Buratto introduced the concept of combining a lamellar procedure with surface ablation excimer lasers,2,3 giving rise to laser-assisted in situkeratomileusis (LASIK) surgery. Several iterations and techniques have been developed since then, and over the last three decades LASIK has become one of the most common and successful elective refractive procedures performed.4,5 Since its inception, more than 15 million patients have undergone the surgery.6 Overall, the complication rate from LASIK is low, with reports ranging from <1–1.8%.7–9
The first critical step in LASIK is creation of the corneal flap. The LASIK flap is paramount to the procedure and allows for quick visual recovery with little pain,10 but is also a major source of complications. There are two main methods for creating corneal flaps. The first involves using a mechanical microkeratome with an oscillating blade.11 The second utilizes a femtosecond laser with a focusable, infrared-spectrum photodisruptive laser, which forms cavitation bubbles that spread to produce a dissected corneal flap.12
Advantages of femtosecond laser flap creation include reduced variation in flap thickness and increased repeatability.13 Some studies have suggested better final visual acuity,14 lower intraocular pressure (IOP) during the flap creation,15 and lower incidence of dry eye.16 Additionally, some prominent microkeratome complications including free/incomplete flaps, buttonholes, and epithelial erosions are less common due to the femtosecond’s precision.14 However, disadvantages of the femtosecond include transient light sensitivity,17 diffuse lamellar keratitis,18 rainbow glare,19 opaque bubble layer,20 and cost.21 Both device classes are still used, but femtosecond has grown in popularity over time. Several femtosecond laser systems are approved by The Food and Drug Administration (FDA) for LASIK use. However, discussion on each individual system is beyond the scope of this review.
Understanding, avoiding and managing potential femtosecond flap complications is essential for anyone performing femtosecond-assisted LASIK. This review will discuss intraoperative and postoperative flap complications associated with femtosecond-assisted LASIK using PubMed-identified relevant published literature. The PubMed search was conducted using “LASIK flap complications” as a primary search term, as well as “LASIK” alongside the various associated complications that will be discussed later.
Intraoperative complications
Suction loss
While not unique to femtosecond flap creation, issues with suction are one of the most commonly encountered problems. Reported rates of suction loss range from 0.06–0.8%.22 Given the unique lamellar cutting ability of femtosecond lasers, suction issues can often be overcome without compromising patient outcomes.23 Unlike keratome flaps, procedures are not automatically aborted when suction loss occurs.4
Predisposing factors for issues with suction include flat corneas (mean curvature of <42 diopters), deep-set eyes, narrow palpebral fissures, patient positioning, lid squeezing, and patient movement of head or eyes.4,5 In these cases, it is essential to ensure adequate initial suction and careful observation during docking. Once central suction is visualized, attention should turn to the periphery to evaluate for a peripheral asymmetric meniscus, which is one of the first signs of suction loss.5 If it occurs early in treatment, immediate cessation is required. Later in the treatment, there should still be very low threshold to stop and redock. A benefit of the femtosecond laser is that the laser cut can be repeated (at the same depth _with the same suction cone), given the reliability of the cutting depth, if no manufacture warnings are present. When completing a partially cut flap, it is essential to ensure that each aspect of the cut (vertical pocket, side cut, lamellar cut) is complete and contiguous. If the suction loss occurred during the raster stage, most surgeons use the “pocket off” setting since the vertical limbal pocket, which absorbs cavitation bubbles, is already complete for the second cut. If the suction loss occurs during the side-cut stage (after completion of raster cut), flap diameter can be reduced by 0.2–0.5 mm depending on the manufacturer.23 An initial inferior flap dissection and lift away from the hinge after a recut is preferred since the cut terminates inferiorly. This is thought to decrease risk of flap tear.23 One study reported that multiple cuts with the femtosecond laser do not cause an irregular stromal bed.24 Tomita et al. found that in cases with suction loss, 97.2% achieved an uncorrected distance visual acuity of 20/20 or better and 100% achieved a corrected distance visual acuity of 20/20 or better.23
If suction cannot be re-established after a partial cut, experts vary on their recommendation for timing of subsequent advanced surface ablation. Some recommend waiting as little as one week with use of mitomycin C to decrease stromal haze,4 while others recommend waiting two months prior to attempting surface ablation, to decrease the healing response and reduce the risk of stromal haze.5 It is difficult to know the exact frequency of suction loss resulting in abandonment of the procedure; however, Brenner et al. reported a rate of 0.003% for aborted femtosecond LASIK flaps in over 7,000 LASIK cases and showed good visual outcomes after subsequent advanced surface ablation.25
Epithelial defects
Epithelial defects are defined in most studies as an area 2.0 mm x 2.0 mm with a break in the epithelium or loose epithelial cells. Defect frequencies have ranged from 0–0.6% in femtosecond studies.26,27 While less common with femtosecond technology, the pocket and shock waves can predispose to epithelial defects. Additionally, inserting the dissecting spatula at the flap edge has the potential to cause a defect.5 Risk factors include anterior basement membrane dystrophy, older age, large flap diameter, recurrent erosion syndrome, or excessive use of topical anesthetic.
Intraoperative management is unchanged but postoperative management may include a bandage contact lens and antibiotics. Additionally, topical corticosteroids should be used more frequently (up to every 1–2 hours) to help prevent development of diffuse lamellar keratitis (DLK).28,29 Finally, patients should be followed more closely until the defect heals, and bandage contact lens use can be discontinued.
Opaque bubble layer
Opaque bubble layer (OBL) occurs when cavitation gas bubbles formed by the femtosecond laser expand in the cleavage plane and become trapped in the anterior stroma.30 Visualizing this layer is a well-known finding intraoperatively with all femtosecond platforms.4 The incidence of OBL when using the laser for flap creation ranges from 5–48%.31
Excessive OBL can lead to interference with many parts of the procedure including flap creation, residual stromal bed measurement, and excimer laser tracking systems.5,30 Risk factors include patients with thicker corneas30 and older patients with denser peripheral collagen that prevents the escape of the bubbles.32 Preventative measures include decreasing flap size if excessive scleral show is noted at the time of suction ring application and adjusting laser settings. Several laser settings can be altered including utilizing less line/spot spacing, larger pocket size, higher energy levels, and using a lighter applanation technique, whereby a complete meniscus is formed but not fully extended to the suction ring edge. One challenge of OBL is a tight flap adhesion which puts the patient at risk of flap tears during dissection.33 Management includes using a flap-lifting spatula with downward pressure to sweep and spread the bubbles along the cleavage plain.4 When present, some surgeons prefer to let the bubbles clear before proceeding which may take up to 30 minutes or more. No significant impact on visual acuity outcomes has been found.5,30
Vertical gas breakthrough
Vertical gas breakthrough can occur when cavitation bubbles dissect superiorly toward Bowman’s layer and through the epithelium. A buttonhole is created with a deep black appearance and the procedure should be terminated to prevent epithelial ingrowth and scarring.34 Subsequent surface ablative procedures after healing can be performed to remove the spot and treat the refractive error.25 If the bubbles stop beneath Bowman’s membrane, a whitish appearance may present but the procedure can typically be continued once a complete buttonhole is excluded.4 Frequency in large studies have shown a 0.3% risk of split flap and up to a 1.3% risk of a pseudo-buttonhole.35 Risk factors include a thin flap, corneal scar, previous radial keratotomy surgery, and microscopic breaks in Bowman’s membrane.4,5
Anterior chamber gas bubble
Evidence suggests that bubbles can enter the anterior chamber via pathways such as Schlemm’s canal, the trabecular meshwork, or pulses misdirected to the aqueous humor.4,36 There is correlation between this finding and the femtosecond dissection being too close to the limbus. This type of dissection can also occur with large flaps, small corneas, or high suction pressure.32 A smaller diameter flap should be considered if scleral show is excessive after applying suction.4 The main concern and complication from anterior chamber gas bubble is the ability to use the pupil-tracking function of excimer lasers. Most surgeons will test the pupil-tracking function and proceed if there is no issue or elect to observe until clearing if tracking is affected.5 In our experience, bubbles can take anywhere from 30 minutes to several hours to dissipate sufficiently for pupil tracking.
Flap tears
Flap tears during femtosecond LASIK typically occur during flap dissection. Femtosecond laser-created flaps can be more difficult to dissect and lift compared to microkeratome-created flaps.37 Thin flaps are at the highest risk of flap tears. One study showed flap tear incidence of 0.5%35 while a similar study showed a 0.4% rate of tears at the hinge.27
For small peripheral flap tears, complete dissection of the flap followed by stromal ablation is acceptable. When a larger flap tear involving the pupillary axis occurs, most surgeons recommend repositioning the flap and aborting the procedure, and considering future surface ablation as a safe way to proceed. For those that proceed with surface ablation and a free flap, a loose anchoring suture to secure the flap after stromal ablation can be used.38
Bleeding
There are two main bleeding complications that can occur with femtosecond LASIK. The first results in a subconjunctival hemorrhage (SCH) and is due to suction. This occurs more frequently (up to 69% in one study)39 with systems that dock on the conjunctiva/sclera such as the IntraLase® (Abbott Medical Optics, CA, USA) rather than platforms that dock on the cornea such as the VisuMax® (Carl Zeiss Meditec, Germany).39 Subconjunctival hemorrhage is not visually significant and clears over 1–2 weeks. SCH can be decreased by using slow, controlled use of suction and ensuring centration. The second type of bleeding occurs due to limbal vessel rupture at the edge of the flap. Risk factors include corneal neovascularization from contact lens use, rosacea, atopy, and any other predisposing factors.4 Bleeding vessels can interfere with laser ablation and cause irregular astigmatism due to uneven ablation if blood pools within the ablation area. During excimer ablation, sponges can be used to remove blood from the stromal bed. Subsequently replacing the flap after excimer ablation helps to control further bleeding. Copious irrigation of residual interface blood during flap repositioning is essential to prevent DLK.5 Flap centration and smaller flaps can help prevent bleeding in those with corneal neovascularization.
Interface debris
Debris is frequently present in the flap interface following LASIK and is typically due to Meibomian gland secretions, eyelash hairs, fibers from sponges, or talc from gloves. Methods to prevent interface debris include powder-free gloves, moistened gauze, clothes covers for the patient, and scrubs for the surgeon.4 During the procedure, adequate irrigation is essential. Prior to repositioning the flap, the surface should be irrigated to remove debris. If noted in the postoperative period and determined not to be infectious or inflammatory, it can be observed. If large amounts of debris are present and visually significant, flap lifting and copious irrigation may be necessary. In our experience, a slit lamp exam shortly after the treatment to look for significant debris with gentle irrigation and flap repositioning at the slit lamp can successfully manage this complication. Larger amounts may require flap lift with a lid speculum in the supine position under the microscope. Mimouni et al. reported rates of flap lift for interface debris to be 0.06%.40
Postoperative complications
Striae and folds
After LASIK, striae and folds are a relatively common flap complication and can be characterized as macro or microstriae. Macrostriae are due to misaligned flaps and are often visually significant, whereas microstriae are not typically visually significant. Several causes have been theorized, including dryness that leads to shrinkage, misalignment, and changes in the corneal contour.41Microstriae are typically observed if best corrected visual acuity is not affected. Management of visually significant macrostriae can range from use of a moist microsponge to gently stroke the flap, to lifting and repositioning the flap. In two very large retrospective studies, macrostriae requiring surgical intervention occurred at rates of 0.79–1.17%.40,42 Further techniques include the use of hypotonic solutions to swell the flap,24 removal of central epithelium, or suturing the flap.4 To prevent striae, marking the flap to ensure correct final positioning is useful. Early intervention leads to best outcomes and we recommend evaluation at the slit lamp immediately following the procedure to ensure appropriate approximation.
Keratitis
There are several types of keratitis that can affect patients after LASIK including infectious, diffuse lamellar keratitis, pressure-induced stromal keratitis (PISK), and central toxic keratopathy.
Diffuse lamellar keratitis
DLK, also known as diffuse interstitial keratitis or “sands of the Sahara,” is an uncommon, nonspecific sterile inflammatory response that occurs within one week of LASIK.43 It presents as inflammatory sterile infiltrate at the interface of the flap and stroma without an anterior chamber reaction. Its incidence in one large study was 0.3%.8 There are four stages of DLK that are based on a system developed by Linebarger et al.44 Stage 1 usually presents on day 1 as white granular cells in the periphery with no involvement of the visual axis. Stage 2 (see Figure 1) often presents during days 1–3 with white granular cells in the visual axis. Stage 3 includes clumping of granular cells, haze, and reduced vision. Stage 4 results in stromal necrosis and melt leading to irregular astigmatism and induced hyperopia (see Figure 2).4
Figure 1: Grade 2 diffuse lamellar keratitis
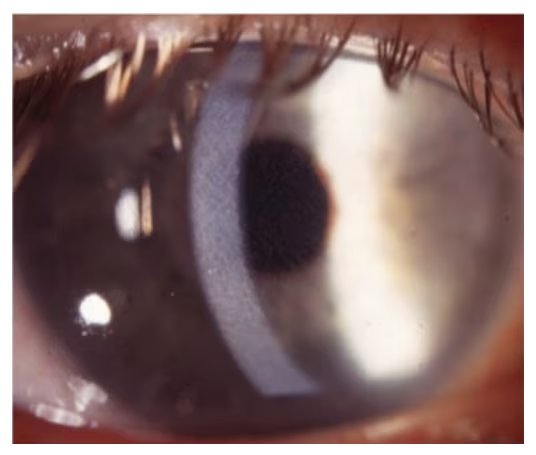
Image courtesy of William Trattler, MD.
Figure 2: Grade 3 diffuse lamellar keratitis

Image courtesy of William Trattler, MD.
Some studies have indicated a higher frequency of DLK in femtosecond LASIK cases compared to microkeratome8,27,45 with an incidence of Stage 1 or 2 DLK as high as 10.6% of patients, though all responded to steroids without visual consequence.27 New studies using updated femtosecond laser models suggest that the incidence is similar to LASIK performed with the microkeratome.46Patients are often asymptomatic but can present with pain or decreased vision. Factors that increase the risk of DLK are blood in the interface and flap epithelial defects.5 If either of these two findings are found, topical corticosteroid use should be increased. Early treatment is paramount with a focus on topical corticosteroids and occasionally oral steroids. In early stages, many surgeons increase topical prednisolone 1% to every hour and consider oral prednisone. If Stage 3 DLK is present, the flap is lifted, scraped, irrigated, and cultured, with possible application of steroids to the stromal bed. When this condition is recognized, it is important to follow closely to avoid stromal melt.4 Late-onset DLK can also happen and has been reported to occur as late as 17 years after LASIK.47
Infectious keratitis
Infectious keratitis is a rare complication following LASIK but one of the most dreaded (see Figure 3). It is not specific to femtosecond laser. Studies have reported an incidence of 0–1.5%48 in LASIK patients and one retrospective case-control study looking at over 500,000 post LASIK patients found an incidence of 0.0046%.49 Presenting symptoms can occur acutely or over days to weeks and include decreased vision, erythema, photophobia, and pain. Bacterial keratitis occurs earlier, typically within 3–5 days, while atypical infections such as mycobacteria or fungal present after a few weeks. Treatment is typically with flap lift and irrigation, culture, broad-spectrum antibiotics, and possible flap amputation if necessary.50 Prevention includes aseptic technique, good lid hygiene, broad spectrum antibiotics in the early postoperative period, and continuous evaluation of sterile technique and instruments.
Figure 3: Infectious keratitis
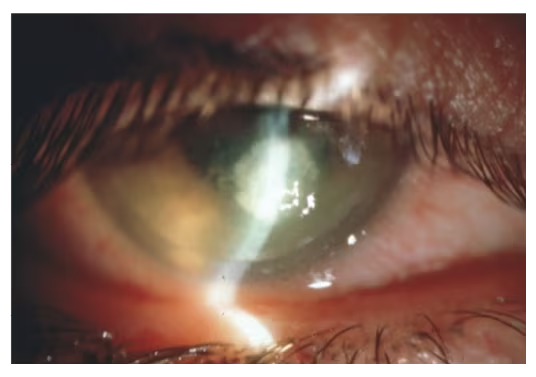
Image courtesy of William Trattler, MD.
Pressure-induced stromal keratitis
PISK occurs as a result of increased intraocular pressure, typically with prolonged steroid use. In this condition, fluid can accumulate in the interface, leading to falsely low readings which can delay the diagnosis. Since the fluid amount is often small, it results in diffuse haziness in the interface and stroma, with no obvious fluid layer, though it can occasionally result in a visible fluid cleft separating the stromal cleavage plain.51,52 It has been reported to occur acutely in the postoperative setting and as a delayed complication, with some cases occurring years after the original LASIK surgery.53,54 As PISK is frequently misdiagnosed as DLK, further use of steroids can exacerbate the clinical condition. Routine IOP checks for patients on prolonged postoperative steroids is essential to diagnose this condition. However, interface fluid can lead to falsely low IOP, necessitating peripheral corneal pressure measurements and maintaining suspicion for the presence this condition. Treatment is typically to remove the steroid and consider a pressure-lowering drop as the IOP will follow suit, resolving the underlying problem.50
Central toxic keratopathy
Central toxic keratopathy is a rare non-inflammatory central corneal opacification that is acute and non-inflammatory in nature (see Figure 4). It occurs within days of stromal ablation procedures. The etiology is unknown but it presents acutely and does not worsen, unlike many of the other interface processes. It presents without pain, which can help distinguish it from DLK. Some surgeons attempt aggressive topical steroid use or flap irrigation, though interventions have not been shown to improve the final outcomes.50,55,56,46 The central opacity often spontaneously resolves in 2–18 months without intervention.
Figure 4: Central toxic keratopathy

Discussion
The introduction of femtosecond laser technology has created a more reproducible and safe LASIK procedure and its use has increased over time. Microkeratomes have also improved with the rates of various complications, decreasing in both femtosecond and microkeratome-assisted LASIK cases. Over time, techniques, treatments and technologies have continued to improve, resulting in fewer complications.
The next wave of refractive treatment, ReLEx/SMILE (small incision lenticule extraction; Carl Zeiss Meditec, Germany) has arrived and can only be accomplished with femtosecond technology. Due to this technology and past femtosecond trends, it is our belief that femtosecond laser use will continue to increase.
Conclusions
Femtosecond laser-assisted flap creation has improved the predictability and thickness of LASIK flaps making LASIK safer. Compared to microkeratome-created flaps, there are fewer complications overall, though femtosecond laser has resulted in a few new complications. It is important for any surgeon performing femtosecond laser-assisted LASIK to be aware of all possible intraoperative and postoperative complications. While the complications are applicable to all femtosecond lasers, it is essential that the surgeon also review the mechanism of suction and flap creation for the specific unit being used to carry out the procedure, as each model has different features that may help reduce certain complications but could predispose the patient to others. In addition to gaining a clear understanding of the mechanisms specific to the laser being used, by reviewing all potential procedural complications and ensuring they have the knowledge to manage any complications that may arise, surgeons can utilize femtosecond laser technology in a way that enables them to perform safe, reliable LASIK. ⬛







