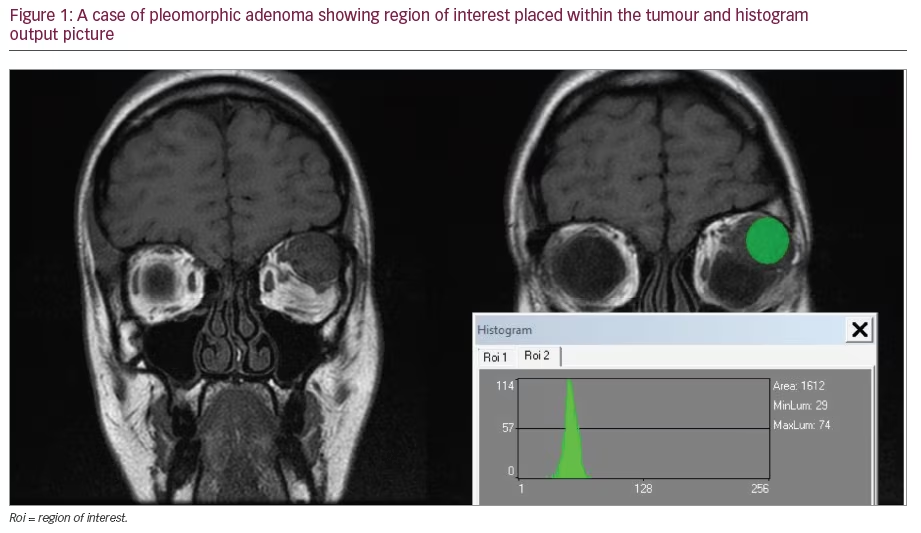
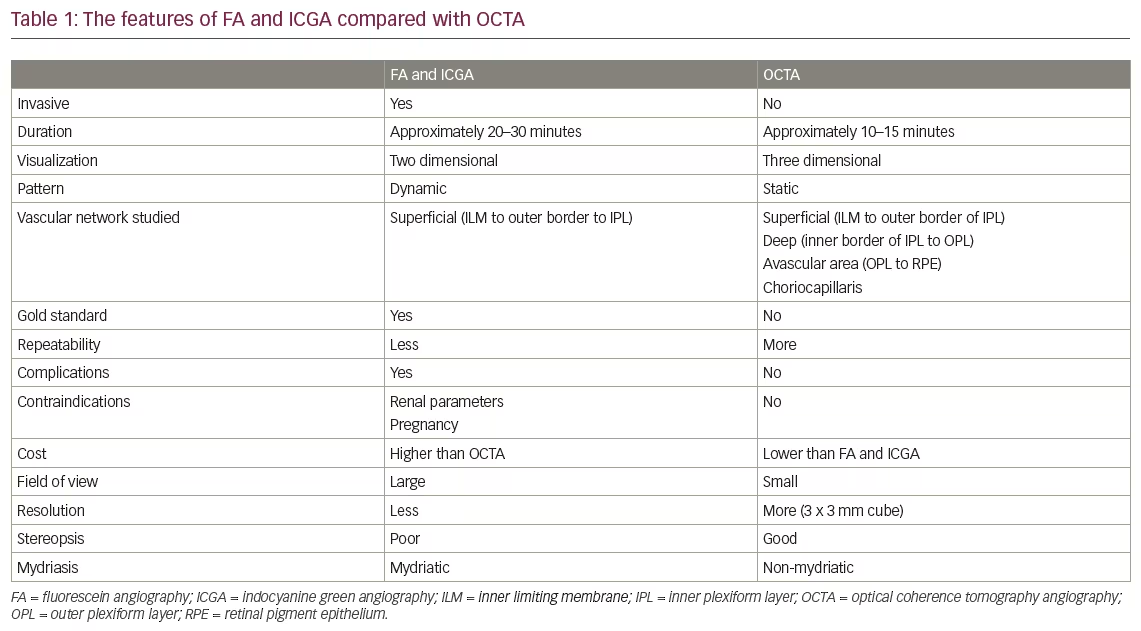
A Clear Vision for Life
Santen, one of the oldest ophthalmic pharmaceutical companies in the world, is Japan’s pioneer and leader in ophthalmic pharmaceuticals. Today, we at Santen are focusing our research and development (R&D) efforts on glaucoma and retinal and corneal disorders. We are also accelerating our efforts towards globalisation to deliver our products and our message of ‘A clear vision for life’ to people around the world.
The Pioneer
A Clear Vision for Life
Santen, one of the oldest ophthalmic pharmaceutical companies in the world, is Japan’s pioneer and leader in ophthalmic pharmaceuticals. Today, we at Santen are focusing our research and development (R&D) efforts on glaucoma and retinal and corneal disorders. We are also accelerating our efforts towards globalisation to deliver our products and our message of ‘A clear vision for life’ to people around the world.
The Pioneer
Founded in Osaka, Japan, in 1890, Santen has a long and distinguished history. The highlights include the launch of Japan’s longest selling eye drops in 1899. In the late 1980s, we achieved significant growth in our prescription ophthalmics business, including the successful launch of Tarivid® (ofloxacin ophthalmic solution) 0.3% and the world’s first ophthalmic fluoroquinolone anti-infective, which became a blockbuster prescription drug. In the 1990s we completed our current business platform, which has enabled our rapid growth by launching innovative prescription drugs, strengthened our R&D capabilities and facilitated our presence outside Japan.
The Leader in Japan
Santen leads Japan’s ophthalmic pharmaceuticals market with around 40% share of the total market. Eighty per cent of total sales come from prescription ophthalmics, including Japan’s first corneal disorder treatment Hyalein® (sodium hyaluronate ophthalmic solution) 0.1% and 0.3%, launched in 1995 and establishing the market for dry eye treatments, Japan’s market-leading anti-infective Cravit® (levofloxacin ophthalmic solution) 0.5% and three glaucoma ophthalmic solutions.
Evolving with a Global Vision
As our R&D goal, we strive to continuously discover and develop innovative pharmaceuticals and services that address unmet medical needs and thus improve quality of life of patients around the world. At present, we are focusing our resources on these selected therapeutic areas, namely glaucoma and retinal and corneal disorder treatments, where we can maximise the strength of our expertise.
As part of our global R&D strategy, we have established strong clinical development capabilities in Japan, the US and Europe. We have harmonised clinical protocols in the three regions whenever possible, thereby allowing us to leverage data obtained in one region to support regulatory approval in another region. As a result of these efforts we expect the launch of Santen’s own prostaglandin, tafluprost, for the treatment of ocular hypertension and glaucoma in Japan and Europe in 2008.
Nara Research and Development Centre – A Climate of Innovations
Built in 1996 as the R&D centre of the Santen Group, our Nara R&D Centre transcends national, organisational and team boundaries and offers opportunities to some 450 researchers and staff to explore their scientific interests in synthesis, pharmacology, pharmacokinetics and drug safety. In addition to cutting-edge laboratory equipment, the centre is equipped with special spaces, where researchers from different disciplines exchange information and share ideas to improve patients’ quality of life.
We also make dedicated efforts for environmental protection of our surrounding environs. In the central surveillance room, for example, we monitor operations and energy consumption of all facilities and equipment around the clock. To minimise air pollution and prevent water contamination, we have installed special equipment to collect all organic solvents.
Growing with a Global Vision
With innovative ophthalmics, we deliver our prescription pharmaceuticals to patients outside Japan by taking the best possible course of action based upon our assessment of local market needs and conditions. In Europe, we acquired Oy Star AB in 1997, and now market a broad range of ophthalmic pharmaceuticals, mainly in Northern and Eastern Europe, Germany and Russia. Prescription ophthalmics include:
• Timosan® – a gel formulation of 0.1% timolol for once-daily treatment of glaucoma;
• Fotil® – a timolol/pilocarpine combination;
• Oftaquix® – levofloxacin 0.5% eye drops; and
• Oftan® Catacrom – for the prevention of cataract formation
The product portfolio is completed by several non-prescription dry eye products:
• Oxyal®;
• Oftagel® ;
• Ocutears®;
• nutritional products for general eye health; and
• advanced micro devices (AMD) e.g. Lutax®, Lutax® AMD, and Optilutin® Omega3.
Between 2003 and 2006, Santen Europe has supported the European Society of Cateract and Refractive Surgeons (ESCRS) Study on Prevention of Postoperative Endophthalmitis, with more than 16,000 patients, supposedly the largest randomised controlled clinical trial ever undertaken with antibiotics. The objective of the study was to prove the effect of intra-cameral cefuroxime and topical levofloxacin given perioperatively.
Our subsidiary in the US is focused on R&D activities, not only for product development in the US, but also for the development and clinical support for European and Japanese product. We distribute three products in the US through a sales and distribution partnership with Vistakon Pharmaceuticals LLC, a division of Johnson & Johnson Vision Care, Inc.
Levofloxacin 0.5% ophthalmic solution is marketed in 5ml bottles under the trade name Oftaquix in Europe and Quixin® in the US. Oftaquix is also available as unpreserved formulation in unit-dose containers in selected European markets.
Santen Corporate Philosophy
Deeply aware of the sanctity of human life, we apply our unique capabilities and technologies in our areas of expertise to contribute to the health and quality of life of patients and their loved ones, and society as a whole.