Age-related macular degeneration (AMD) is the third leading cause of blindness worldwide and the primary leading cause of vision loss in the Western world. Its prevalence is expected to increase as a consequence of an aging population, such that it is estimated that close to 288 million people will be affected by AMD by 2040.1 AMD presents in two major forms: the non-neovascular, non-exudative “dry” form affecting 85–90% of patients and the neovascular, exudative “wet” form affecting 10–15% of patients. Up until the late 1990s, treatment for AMD was limited to destructive thermal laser therapy for the neovascular form. In the last two decades, we have experienced a renaissance with more targeted approaches for the treatment of neovascular AMD. Liposomal verteporfinbased photodynamic therapy (Visudyne®) was used to selectively close choroidal neovascularizations (CNV)—it is the first pharmacotherapy for AMD that is able to reduce and slow vision loss.2 Further work to understand the biological process of new vessel development, and demonstration of the key role of vascular endothelial growth factor (VEGF) led to extremely effective therapies, revolutionizing the treatment of neovascular AMD and preserving sight for millions of people.3 Subsequently, anti-VEGF therapy was applied to other diseases with abnormal angiogenesis and vascular leakage, including diabetic retinopathy, retinal vein occlusions, and pathologic myopic neovascularization, among others.
However, the pathologic processes underlying dry AMD remain elusive to date, with a lack of effective therapies. Non-exudative AMD is characterized by accumulation of deposits under the retinal pigment epithelium (RPE) and neurosensory retina, as well as degeneration of the RPE, photoreceptors, and even the choroidal vasculature. All of these ultimately lead to photoreceptor dysfunction, death, and vision loss. Although epidemiological and genetic studies have identified several candidates for the formation and progression of dry AMD, they also point to involvement of multiple biological pathways, including: lipid metabolism and transport regulation, inflammation (especially the complement system), extracellular matrix remodeling, cell adhesion, cellular toxicity, cell death, and angiogenesis. However, there is a lack of a unifying hypothesis that can explain how the disease starts and progresses—the causes of RPE and photoreceptor degeneration and loss remain obscure. The failure in truly understanding the pathogenesis of the disease, the lack of effective therapies, and the increasing prevalence all underscore a significant unmet clinical need. There is an overwhelming importance to address this issue by improving our classification system, identifying better therapeutic targets—especially early in the disease process—and developing better biomarkers and clinical endpoints.
Current classification and characterization of age-related macular degeneration
Several classification schemes of AMD have been developed, mostly based on color fundus photos. The most well known is the system used in the Age-Related Eye Disease Study (AREDS) study that classified AMD into early, intermediate, and late stage, accordingly.4 Early-stage AMD is defined by the presence of a few medium-size drusen and pigmentary abnormalities such as hyperpigmentation or hypopigmentation; intermediate-stage AMD is defined by the presence of at least one large druse, numerous mediumsize drusen, or geographic atrophy (GA) that does not extend to the center of the macula.5 Currently, early and intermediate AMD are only treated with AREDS-based vitamin supplementation. Late-stage AMD can be divided into advanced non-neovascular AMD and neovascular AMD. Advanced non-neovascular AMD is marked by drusen and GA extending to the center of the macula, while neovascular AMD is characterized by CNV and any of its potential sequelae—subretinal fluid, lipid deposition, hemorrhage, RPE detachment, and/or fibrotic scarring.
Despite the impact of the AREDS study on both the classification of the disease and treatment with vitamin supplementation for the disease, consensus is still lacking among physicians regarding terminology for the staging and progression of the disease. To tackle this, a new proposed scheme of clinical classification was put forward in 2013 by the Beckman Initiative for Macular Research Classification Committee, proposing 3 stages of the disease and one for a normal, aging phenotype (only small drusen <63 μm without pigment changes).6 They defined early AMD by the presence of medium drusen (>63 μm; ≤125 μm) and no AMD pigmentary abnormalities. The presence of large drusen and/ or any pigmentary changes was considered intermediate stage, and advanced stage disease was characterized by presence of any CNV or GA (see Table 1). It is important to note that this classification is also based on fundus photography and does not include information from other imaging modalities—optical coherence tomography (OCT), autofluorescence, or wide field imaging—and does not account for the presence of subretinal drusenoid deposits. Race, genetic, or environmental information is not included in the classification scheme, nor is it necessarily based on biological pathogenic processes.
While the late stages of AMD seem to converge into common pathogenesis pathways such as cell senescence and death in non-neovascular AMD, and angiogenesis in neovascular AMD, it appears that different biological pathways (lipids, autophagy, inflammation) may predominate in early- and intermediate-stage AMD. Thus, it is interesting that some clinical trials have aimed to target specific early biological pathways (such as inflammation and complement) to halt late stages of the disease such as progression of GA. It is possible that interventions of this type are ineffective this late in the disease course. By this time, we may need to consider approaches to inhibit cell death. Targeting underlying disease biological processes should occur in the early/intermediate stages and should include approaches involving lipid metabolism and transport, inflammation and complement, and cellular aging and senescence. Advanced stage (CNV, GA) targets should include anti-angiogenesis (anti-VEGF and neovascularization maturation) and anti-cell death (neuroprotection), respectively. Data also show that while re-classification of AMD based on biological processes is necessary, development of biomarkers to identify therapeutic targets for different subtypes of early and intermediate AMD is also critical.
Targeting lipids in age-related macular degeneration
One of the sine qua non of AMD is the accumulation of lipid-rich basal laminar deposits and drusen; it is thought that at least 40% of drusen volume is comprised of lipids.7 Unlike atherosclerosis, serum low-density lipoprotein (LDL) levels in AMD do not have a strong association.8 However, there are certainly similarities between these two diseases. A number of studies have shown a link between cardiovascular risk factors and AMD,9–13 as well as several shared susceptibility genes. In addition, genome-wide association studies (GWAS) of AMD patients have identified several lipid metabolism-related genes (Table 2).14–16
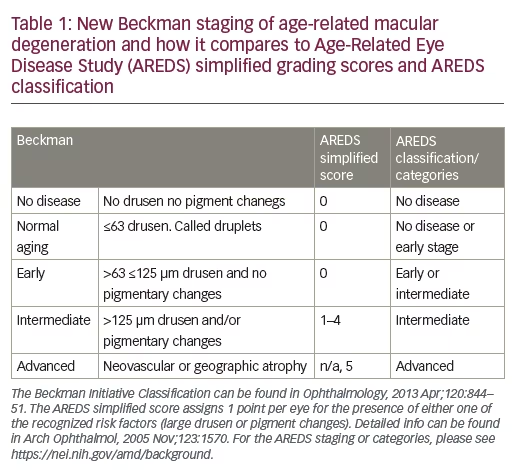
Even before we had the genetic and epidemiological evidence for shared pathogenic mechanisms in cardiovascular disease and AMD, histopathologic data pointed to similarities between these two diseases.17 Bruch’s membrane forms the inner margin of choriocapillaris and is considered an analog of the vascular intima sharing similar changes with aging. Similarities in molecular composition between drusen and atherosclerotic deposits lend further support to this concept. In both conditions, there is cholesterol and Apolipoprotein B (ApoB) accumulation with subsequent oxidation and modification. Drusen components are derived from local sources (retina/ RPE secretion of ApoB and ApoE lipoproteins) and, to a lesser extent, from the circulation. The retention of lipids leads to the formation of a lipid wall, basal linear deposits (BlinD), and drusen (Figure 1).
Given the many similarities between AMD and atherosclerosis, there have been several epidemiological studies investigating the role of statins in AMD. Previous studies examining the effect of statins on AMD status or the ability of statins to alter disease progression showed mixed results. Van der Beek et al. showed that increased serum LDL and triglycerides, plus more than 1 year of statin use, led to increased risk of neovascular AMD while a meta-analysis by Klein et al. of three cohorts showed no association of AMD incidence or progression with serum lipids, statin use, or lipid pathway genes.18,19 A small, proof-of-concept, randomized, placebo-controlled study suggested that daily simvastatin at 40 mg (equivalent to 20 mg atorvastatin) may slow progression of early/intermediate AMD in patients with complement factor H (CFH) genotype CC (Y402H).20 A 2016 Cochrane review concluded that there is insufficient evidence to conclude that statins play a role in preventing or delaying the onset or progression of AMD.21 All of these studies are limited by AMD disease heterogeneity and a lack of standardization of statin dosages or lipophilicity. A review of cardiovascular literature suggested that statin dosage affects outcome—low/moderate doses showed decreases in disease progression,22–27 whereas high-dose (80 mg) atorvastatin led to regression of atheromas.28–30
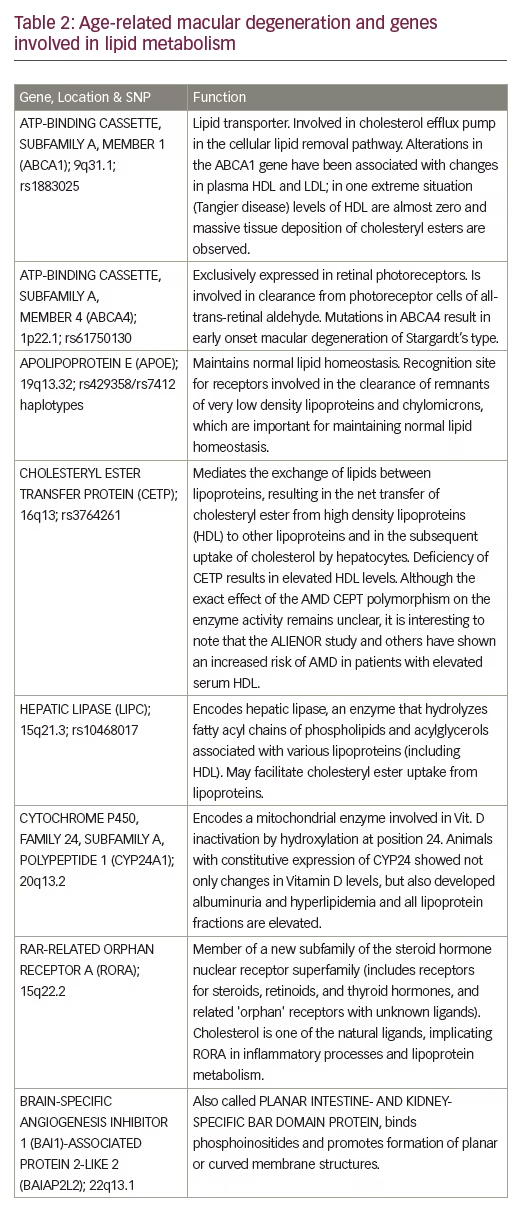
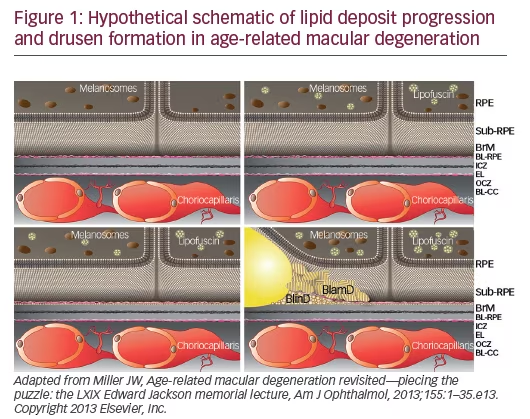
A small pilot phase I/II study of high-dose atorvastatin (80 mg) in selected patients with large soft drusenoid deposits/pigment epithelial detachments (PEDs) showed regression of drusenoid deposits in ten out of 23 patients with an average follow-up time of about 1.5 years.31 Responders had stable or slightly improved vision. None of the study patients developed atrophy or progressed to neovascular AMD. Possible mechanisms of statin therapy could include changes in RPE lipoprotein metabolism, creation of a systemic permissive state for lipid efflux, improvement of macrophage lipid clearing status, as well as anti-inflammatory and protective effects on RPE and antiangiogenic effects. The results of this pilot study are consistent with the “oil spill” hypothesis proposed by Curcio and colleagues,17 and suggest for the first time that this disease can be reversed anatomically and functionally (Figure 2). These results need to be confirmed in larger randomized studies that will include genetic analysis, lipid sub-species measurements, and functional studies, such as dark adaptation.
Inflammation and Immunity
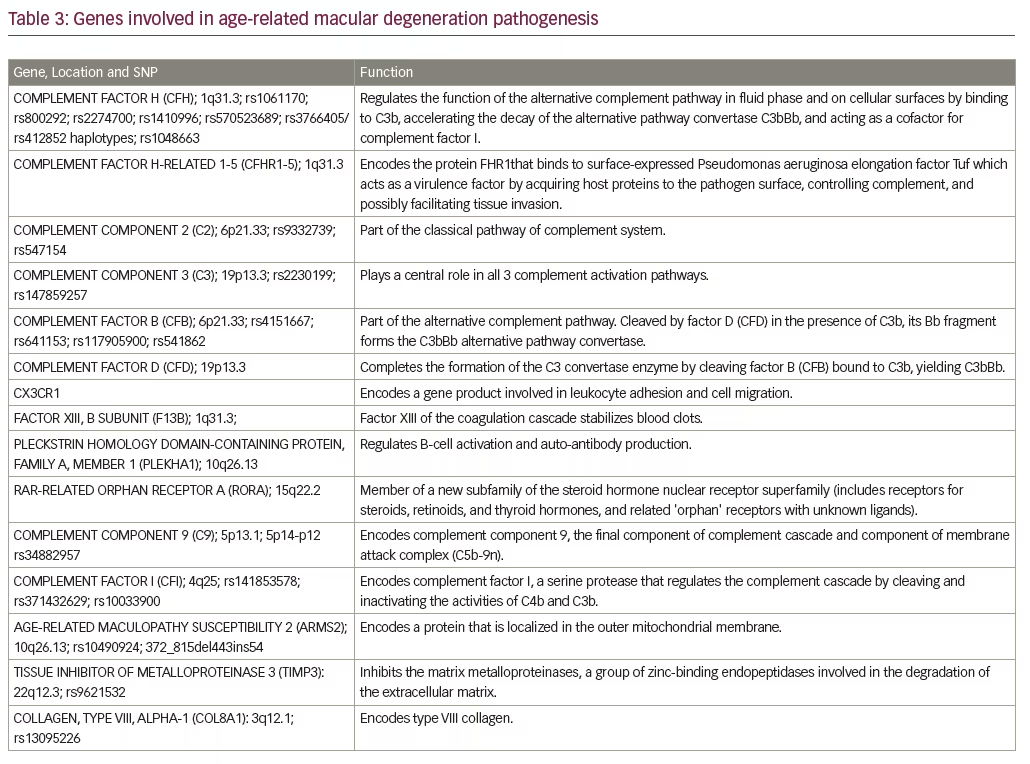
Aging and lipids are required for AMD but are probably not sufficient to cause AMD. Inflammation and immune dysfunction appear to be required as well, and multiple genes involved in inflammation have been associated with AMD (Table 3).
Inflammation appears to be central to all stages of AMD. However, it is likely not classic inflammation, but rather a “para-inflammation”—a low-grade inflammation responding to aging and other insults.32 While it is thought that some level of para-inflammation may be helpful, at a given point it becomes pathogenic, leading to disease development. It is important to note that human histological/biochemical data on pathological inflammation or para-inflammation in AMD remain sparse. A study in 2015 showed involvement of CD163+ cells in the eyes of patients with AMD;33 another study found elevated vitreal granulocytemacrophage colony-stimulating factor (GM-CSF) and increased CD68+ choroidal macrophages.34 In a more recent study from 2017, complement factor 3-positive immune cells were observed in AMD specimens.35 With imperfect animal models, sparse human data, and the potential for parainflammation to also be protective in aging, it remains unclear how we should modulate the inflammatory response to obtain a therapeutic effect in patients with AMD. There is clearly a need for further investigation into the role of inflammation in the pathogenesis of AMD.
Complement
The discovery of the association with AMD of gene polymorphisms in the complement regulatory component, factor H (CFH) that regulates the alternative complement pathway was a seminal finding. Additional studies have implicated single-nucleotide polymorphisms (SNPs) in other complement genes including CFI, CFB, and CFD as risk alleles as well.36–42
Klein et al., Haines et al., and Hageman et al. in 2005 identified CFH polymorphisms in AMD. All groups identified a tyrosine to histidine polymorphism in the region of CFH that binds heparin and C-reactive protein. The odds ratio (OR) varied according to homozygosity and among the different studies, ranging from 2.46–7.4. Smoking was found to increase AMD risk related to CHF as smokers homozygous for the CFH Y402H variant had an OR of 34 for late-stage AMD compared with non-smokers without the risk allele.43 It is important to note that these results suggest that up to a third of the US population 65 years or older without AMD (12.7 million people)44 may have the most frequent at-risk haplotype for AMD without developing the disease.
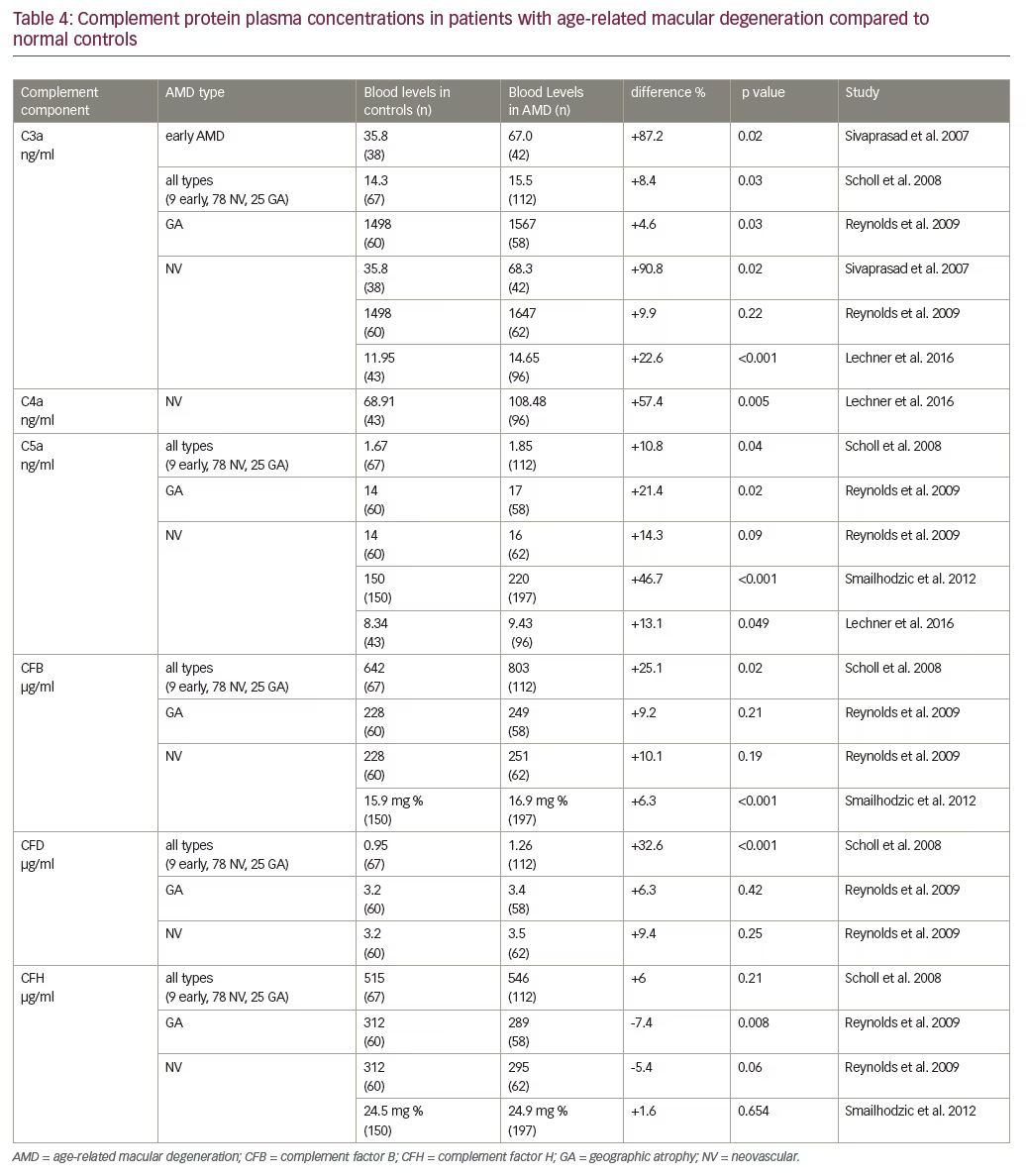
Plasma levels of complement proteins have been shown to increase with age,45 but, it is postulated that patients with AMD have a stronger imbalance of complement activation and regulation, possibly leading to complement over-activity. Greater increases in complement protein plasma levels have been associated with advanced stages of AMD, leading to the hypothesis that complement activation may be correlated with disease progression as well.45,46 There is weak evidence that genetic polymorphisms in CHF associated with AMD may lead to over-activity of complement. Scholl et al. found that median plasma concentrations of CHF were not increased in patients with AMD,47 whereas complement factor D (CFD) levels did, indicating that CFD may be a more promising target for AMD therapy than CHF. In these studies, the increased plasma concentrations of complement protein observed were wide-ranging and the ranges for different components of the complement system also varied greatly (Table 4). A significant increase of C3a was found in AMD patients, ranging from 4.6–87.2%; C5a levels were found to range from 10.8–46.7%.47–51 Scholl et al. found a significant increase in CFD levels by 32.6% compared to normal controls and no significant difference in CFH levels.47 However, Reynolds et al. did not find a significant difference of CFD levels between AMD patients and controls, but identified a significant decrease (7.4%) in CFH plasma concentrations in patients with GA.48 Table 4 summarizes the data on systemic complement factor levels in AMD and controls. Please note the wide variability in ranges of these studies.
Histology of AMD eyes has demonstrated expression of many complement components in drusen.52,53 It should be noted, however, that antibody studies are tricky, and it is difficult to draw definitive conclusions from them because many antibodies are notoriously “dirty”, as many bind non-specifically—especially in “sticky” drusenoid deposits. Mullins et al. demonstrated labeling of membrane attack complex (MAC) in Bruch’s membrane and choriocapillaris with age and in AMD, concluding that choroidal endothelial cells are targeted by MAC leading to choroidal thinning in AMD.54 The immunofluorescence performed by this group shows extensive MAC-labeling throughout the choriocapillaris, targeting almost every choroidal endothelial cell.54 The widespread labeling of choriocapillaris (CC) endothelial cells with MAC without apparent massive and rapid loss of CC in AMD suggest that this observed MAC labeling maybe an artifact or, as the authors speculate, the MAC labeling seen may result in as yet unproven sub-lytic deleterious effect and slow long-term deterioration of endothelial cell function.
Despite the evidence suggesting a role of the complement system in AMD, trials targeting complement proteins in AMD have so far failed to demonstrate efficacy. The anti-C5 antibody eculizumab (Soliris®; Alexon Pharmaceuticals, Cheshire, CT, US) investigated in the COMPLETE study (NCT00935883), as well as by Filho et al., failed to show reduction in GA growth rate at 6 months, or reduction in drusen volume at 26 weeks of treatment.55,56 A phase II/III trial of ARC1905 (Zimura; Ophthotech Corp., Princeton, NJ, US), an intravitreous anti-C5 aptamer is currently recruiting participants with GA (NCT02686658).57 More recently, the MAHALO phase II clinical trial (NCT01229215) investigated the safety and efficacy of lampalizumab—a complement factor D antibody—for the treatment of GA in monthly injected subjects and controls over 18 months. The study showed a trend to reduction in GA progression of 20%, but was not significant.58 Two phase III trials to investigate the safety and efficacy of monthly or 6-weekly injections of lampalizumab have completed recruiting participants (CHROMA, NCT02247479 and SPECTRI, NCT02247531).59,60 Results of the phase III study SPECTRI were recently announced and showed no efficacy. Similar negative results of CFD inhibition in AMD from the CHROMA study are expected in a few months.
A phase I trial for the intravitreal complement C3 inhibitor POT-4 in patients with neovascular AMD showed no safety concerns, however, a phase II trial has not been initiated as of yet (NCT00473928).61,62 Finally, two phase II clinical trials have investigated amyloid-beta antibodies for the treatment of GA (NCT01577381, NCT01342926). Amyloid-beta, a component of drusen, is believed to be an activator of complement, and is thought to play a role in AMD progression. Outcomes for these studies are not yet available.63–66
NLRP3 inflammasome
Another component of inflammation that has been proposed to play a role in AMD is the NACHT, LRR, and PYD domains-containing protein 3 (NLRP3) inflammasome. The NLRP3 inflammasome is a protein complex within immune cells and is part of innate immunity leading to activation and release of interleukin (IL)-1β and IL-18. In 2012, Tarallo et al. published that Alu RNA transcripts accumulated in RPE following loss of DICER1 expression primed and activated the NLRP3 inflammasome in RPE, leading to IL-1β and IL-18-mediated degeneration of RPE.67 However, in the same year Doyle et al. suggested that NLRP3 in infiltrating macrophages and microglia was activated by drusen and drusen components such as C1Q. This NLRP3 activation led to increased IL-18 levels, ultimately providing protection from neovascular AMD in rodent and primate models.68–70 More recent studies by our group suggest that inhibition of RPE NLRP3 is unlikely to be an effective approach in AMD (unpublished data).
Aging and senescence
Aging remains one of the biggest risks in AMD. Cell senescence is a biological change linked to aging and many age-related diseases. There are multiple alterations that happen in the senescent cell, including shortening of telomeres, activation of the DNA damage response (DDR) through the ataxia telangiectasia mutated (ATM)-p53-p21 axis, and the p16Ink4a protein (p16) leading to an activation state of retinoblastoma (Rb) protein. Senescent cells are also known to be marked by an elevation of lysosomal-β-galactosidase (SA-β-GAL) that has served as a rather easy and specific marker of senescent cells.71–75
Cellular senescence is thought to cause tissue repair impairment through the production of inflammatory senescence-associated secretory phenotype (SASP)—pro-inflammatory and matrix degrading molecules that are mediated largely by NF-κB and p38 MAPK signaling.76,77 Although senescence is associated with some harmful effects, not all senescent cells are thought to be detrimental. Short-lived cellular senescence may help in morphogenesis, wound repair, and tumor suppression.78–81 Senescent cells may also be effectively cleared by the immune cells that are called in by the SASP components.82,83 However, chronic senescent cells that are not cleared are thought to be harmful and contribute to tissue dysfunction.
It is rather surprising that cellular senescence has not been systematically or extensively studied in AMD.84 RPE cells show senescence in vitro,85–89 and senescence-prone mouse strain 8 has shown photoreceptor loss and increased p16 expression in RPE cells.90 To date, there appears to be a lack of cell senescence data in humans, and there is only one non-human primate study which detected senescence markers in the RPE of aged monkeys.75
In aging and senescence, not only do lysosomal hydrolases like SA-β-GAL change, but the lysosomes themselves, as well as many of their functions, are altered.91 Lysosome-associated membrane protein-2 (LAMP-2) is a lysosomal protein essential for many functions including autophagy, and its expression is known to decline with age in the body.92 Systemic mutation of this protein leads to Danon’s disease, characterized by the classic triad of cardiomyopathy, skeletal myopathy, and mental retardation.93 Importantly, Danon’s disease also includes progressive retinal degeneration.94–96 In experimental systems, impeded phagocytic degradation of photoreceptor outer segments, compromised lysosomal degradation, and increased lysosomal exocytosis all contributed to the formation of sub-RPE deposits in Lamp2-deficient RPE cells.97 Notably, Lamp-2-deficient mice recapitulate several classical features observed in AMD such as extensive sub-RPE drusenoid deposits, and progressive RPE cell loss followed by photoreceptor cell loss and atrophy.97
Another feature of aging is impairment of clearing damaged DNA. It has been shown both in human AMD specimens and in vitro experiments that there is an increase in damaged mitochondrial DNA (mtDNA) in the RPE of patients with AMD, leading to para-inflammation.98–103 Similar findings have been observed with damaged genomic DNA.104 What contributes to the damaged DNA and whether it is causative or a feed-forward epiphenomenon need further investigation.
Studies on longevity regulator proteins have focused primarily on silent information regulator T1 (SIRT1),105,106 and, to a lesser degree, AMPdependent kinase (AMPK).107 SIRT1 is a member of NAD+-dependent protein deacetylases responsible for controlling a wide variety of signaling and transcription factors.105,106 AMPK is the energy sensor of the cell— responding to the AMP/ATP ratio, suppressing anabolic pathways, and stimulating energy producing processes.107 Caloric restriction is one of the most potent longevity stimuli and is known to increase SIRT1 expression, as well as AMPK activity.107–114 Relatively little is known about the role of SIRT1 and AMPK in AMD. However, one study suggested that genetic variations of SIRT1 could be implicated in the pathophysiology of AMD in the Chinese Han population.115 Another study indicated that lower expressions of SIRT1 and PGC1α were observed in iPSC-derived RPE cells from patients with AMD.116 Even less data exist for AMPK and AMD—a few reports suggested AMPK and mechanistic target of rapamycin (mTOR) as potential therapeutic targets in AMD,117,118 while in vitro experiments suggested a protective effect for AMPK activation on RPE cells and downregulation of complement factor B (unpublished data, Eun Jee Chung et al.).119 Work from our group has shown that aging changes in photoreceptor connectivity are associated with reduction of AMPK in mice. Pharmacologic activation through Metformin or caloric restriction can reverse these aging changes.120 Further investigation is needed to identify therapeutic targets for AMD that will have the ability to reverse senescence and stimulate longevity.
Neovascular age-related macular degeneration
Anti-vascular endothelial growth factor therapy and long-term results
Vascular endothelial growth factor (VEGF)—first identified as a vasopermeability factor and initiator of angiogenesis due to hypoxia—is the key angiogenic factor in neovascular AMD. Anti-VEGF treatment for eye diseases has been one of the greatest success stories in modern medicine, and has resulted in preserving and/or improving vision for millions of people. Although more than 80% of patients have “dry” retinas with anti- VEGF monotherapy, there is an incomplete gain in visual function.121–125 Over longer periods of time, there is loss of the vision gains occurring within the first 2 years of anti-VEGF treatment.122,126–128 Part of the reason for the loss of visual gains in the long-term is likely due to under-treatment in the “real” world.129,130 However, as these retinas are virtually “dry” with little to no intraretinal fluid present, this suggests that something else is responsible for the decline in visual function observed in these patients. Even when the neovascular process is controlled, the underlying degenerative process continues with a progression of GA in patients with neovascular AMD. This suggests an important role for neuroprotection, to be discussed later. The progression of atrophic changes may be furthered by decreased perfusion and resulting ischemia, as the regression of CNV with anti-VEGF may eliminate the only remaining blood supply for the outer retina.131 Another explanation for this phenomenon could be the that the neurotrophic effect of VEGF is blocked by anti-VEGF treatment; however, there is little clinical evidence for this.132
It is also important to remember the considerable burden of monthly anti-VEGF injections on patients, their support network, and providers.133 Improving treatment for neovascular AMD should therefore both: (1) include a better outcome in terms of improved visual acuity, and (2) reduce the number of injections needed for effective treatment. Some believe that targeting another angiogenic factor could be helpful. To date, these approaches have been unsuccessful. To decrease the number of injections, investigators have studied ways to extend the duration of anti-VEGF effect through long-term sustained release of macromolecules, with no success. The lack of success in finding methods for long-term sustained release may be due in part to the large size of the molecules (50,000–150,000 Da), limiting the number of molecules that can effectively be packed within a usable volume. With a smaller fragment (25,000 Da) of anti-VEGF antibody called RTH258 (brolucizumab), Novartis has been able to increase the injection amount to 6 mg (equivalent in molar dose to 12 mg of ranibizumab), successfully extending the dosing frequency to 12-week intervals.134–137
Another approach to circumvent these physical limitations is to use a refillable reservoir that can contain material for 6 months. This approach is championed by Genentech after it acquired ForSight Vision. The two companies have been collaborating for several years to develop the refillable rigid port delivery system (RPDS). The RPDS is an intravitreal implant that is placed surgically through a scleral incision that, in theory, can be refilled by a physician using proprietary refill needle in the office. Although it appears that it can contain enough material for slow release over 4–6 months, it has yet to show that it can be refilled successfully several times. The clinical trial investigating this refillable device (LADDER; NCT02510794) is still active.138 Additionally, hydrogels as sustained-release deposits for both small and large molecules are being investigated by Regeneron with Ocular Therapeutix, but these studies are still in preclinical stage.139
Platelet-derived growth factor
Platelet-derived growth factor (PDGF), a dimeric glycoprotein, is critical for pericyte survival, recruitment, and maturation. PDGF-receptor-beta (PDGFB) deficiency has been shown to result in microvascular pericyte loss, the development of capillary microaneurysms leading to proliferative retinopathy, and the inability of sprouting capillary endothelial cells to attract PDGFB-positive pericyte progenitor cells.140,141 Pericytes protect endothelial cells from VEGF inhibition; therefore, pericyte loss in the neovascular complex was believed to act synergistically with anti-VEGF therapy, leading to an increased endothelial cell response to anti-VEGF treatment.
Despite the early excitement about the potential of anti-VEGF and anti- PDGF combination therapy for neovascular AMD, two large clinical trials failed to meet the optimism created by successful preliminary studies. The failure of the PDGF trials was not entirely surprising as there were several hints suggesting that they may be less than successful. First, anti- VEGF monotherapy is sufficient to “dry” the macula in the clear majority of patients. Thus, it is difficult to conceive additive effects with an adjuvant therapy that also targets angiogenesis. Second, targeting of pericytes in neovascularization may not be desirable, since pericytes are needed for vessel maturation and therefore decreased vascular leakage.142 Third, evidence suggested that PDGF-BB and VEGF do not synergize in all models of ischemia-related angiogenesis; PDGF may synergize with FGF instead.140 Finally, the role of PDGF expression in the outer nuclear layer of the macula of patients with AMD is still undetermined and other off-target effects of PDGF inhibition remain to be elucidated.140
Angiopoietin-TIE Pathway
Another major player in the process of angiogenesis, is angiopoietin (ANG)-1, a glycoprotein that binds to tyrosine kinase receptor TIE2. TIE2 is expressed on endothelial cells as well as early hematopoietic cells.140 The ANG/TIE2 pathway is involved in maintaining vascular integrity and stability.143 Knockout of ANG-1 or TIE2 leads to embryonic lethality, with failure of smooth-muscle and pericyte precursor recruitment. ANG-2 is a competitive antagonist of ANG-1 for the TIE2 receptor. Binding of ANG-2 to TIE2 does not lead to phosphorylation of the receptor.144 Overexpression of ANG-2 in mice leads to disruption of blood vessel formation and gives a phenotype similar to that of ANG-1 deficient mice.144 It is thought that ANG-2 mediates endothelial cell survival, increasing their responsiveness to VEGFenhancing neovascularization.144 It was also noted by the authors of this study that ANG-2 in fibroblasts could activate TIE2 receptors when VEGF is absent, and this ANG-2 stimulation may lead to vessel regression.144 In the corneal micropocket assay, neither ANG-1 or ANG-2 alone could lead to neovascularization, however, they were able to augment VEGF effects, with ANG-2 being more potent than ANG-1. In cases of VEGF-inhibition, ANG-2 binding resulted in apoptosis.140
There has been a lot of interest in exploring angiopoetin as a therapeutic target for neovascular AMD and other retinal vascular diseases. Elevated levels of ANG-2 (43 versus 9 pg/L) were found in the aqueous humor of patients with neovascular AMD,145 and a small study from Hong Kong has shown some suggested associations between ANG-2 SNPs and neovascular AMD, particularly polypoidal.146 Otani et al. showed that surgically excised CNV stained positive for ANG-1 and ANG-2 with increased ANG-2 immunoreactivity in the highly vascularized regions of CNV—similar to the staining pattern of VEGF.147 Heras et al. also detected variable amounts of VEGF, ANG-1, and ANG-2 in surgically excised CNV membranes; TIE-2 and VEGF receptor (VEGFR) were not detectable in their study.148
Several preclinical studies suggest that direct or indirect activation of the ANG-1 system diminishes CNV and VEGF-induced leakage.149–151,143 A 12-week phase IIa clinical trial of AKB-9778 (an inhibitor of vascular endothelial protein tyrosine phosphatase, and indirect activator of ANG- 1/TIE2) showed that patients with diabetic macular edema receiving a combination therapy of ranibizumab with subcutaneous AKB-9778 had significantly more reduction in mean macular thickness at 12 weeks compared with the ranibizumab monotherapy group. However, there was no better visual acuity than with monotherapy alone (NCT02050828).152 A phase IIb clinical trial of AKB-9778 is currently recruiting patients with non-proliferative diabetic retinopathy (TIME-2b, NCT03197870). In the field of oncology, a phase II study targeting angiosarcoma using antrebananib to block both ANG-1 and ANG-2 failed to show efficacy.153 A new bispecific crossed monoclonal antibody (crossMAb) has been developed by Roche targeting both VEGF and ANG-2, and may be more efficacious than anti- VEGF alone in the non-human primate model of CNV.154
In summary, ANG-2 is important for vascular physiology and there is a potential for synergistic effects in combination with anti-VEGF in retinal diseases. However, for the reasons alluded to above, combination antiangiogenic therapy in neovascular AMD may not be effective. Anti-VEGF treatment alone usually very effectively “dries” the retina in patients with neovascular AMD, leaving little room for improvement. In addition, while most studies suggest advantages of blocking ANG-2 in neovascular diseases, some basic science research suggests that ANG-2 may have opposing roles in neovascularization depending on the environmental context. It may be that ANG-2 blockade is more valuable in diseases with more typical inflammation and in true anti-VEGF non-responders, such as one finds in diabetic retinopathy.
As such, future treatments targeting the neovessels in neovascular AMD should be aimed at increasing the dosing interval and decreasing the need for frequent injections, as opposed to increasing the anti-angiogenic effect. It is also important to consider increased anti-angiogenesis therapy aimed at the regression of CNV may eliminate the only remaining blood supply to the outer retina, leading to progression of atrophy and worsening of the disease.
Future treatments—neuroprotection
Ongoing neurodegeneration leads to GA. Given that AMD is a multifactorial polygenetic disease, our group has used many animal models to investigate commonalities in cell death processes and the reasons why prior approaches on inhibiting apoptosis have failed. Using the separation of photoreceptors from the RPE as a model of cell death, we and others have found evidence of caspase-mediated apoptosis as well as elevation of upstream death signals such as TNF and FAS ligand (FasL).155–167 However, blocking caspases did not lead to prevention of cell death,167,168 leading us to investigate other forms of cell death. Through morphological studies, we have known since the 1970s that at least three different forms of cell death exist.169 The first one (Type I) was characterized by cell condensation and fragmentation and is now known as caspase-mediated apoptotic cell death. The second type (Type II) was characterized by the presence of numerous double membrane vacuoles/structures and is now known as autophagymediated cell death. The third form of cell death (Type III), necrosis, was characterized by cell swelling, membrane blebbing, vacuolization, and cell rupture—for years it was thought to be unregulated and haphazard. However, more recent studies have indicated that the Type III modality of cell death is also regulated by a set of protein kinases called receptorinteracting protein kinases or RIPK.170,171
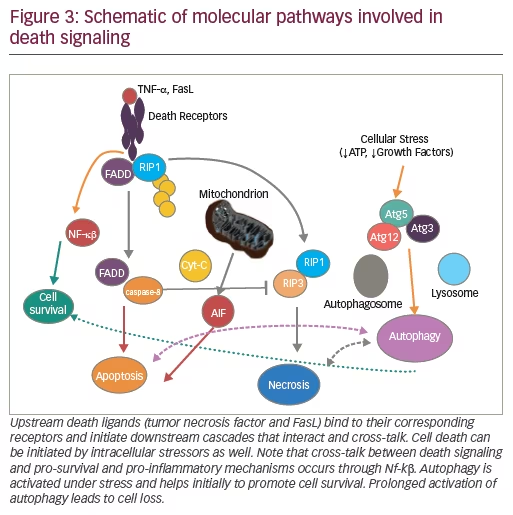
In addition to evidence for caspase-mediated cell death, we examined evidence of RIPK-mediated necrosis in photoreceptor cell death. Indeed, using the retinal detachment model, we found upregulation of expression and phosphorylation of RIPK along with activation of caspases (Figure 3).160,167,172,173 When we tried to inhibit RIPK or caspases in isolation, no major effect on photoreceptor loss was observed.167 In contrast, combined treatment led to significant rescue.167 In further investigations using morphologic assessment via electron microscopy, we observed that caspase inhibition alone led to decreased apoptotic cell death; however, it increased necrotic cell death, thus leaving the overall level of cell death unaltered. It was only when both cell death pathways were blocked that reduced overall cell death was observed (Figure 4).167 Thus, we can conclude that there are common cell death pathways that are redundant and complimentary to each other. Cells have alternative death pathways to caspase-mediated apoptosis that are mediated through RIPK activation. Blocking only one pathway is not sufficient and in order to prevent photoreceptor cell death, it likely is necessary to block both apoptotic and necrotic pathways.
Our findings from the photoreceptor/RPE separation model of cell death were then further explored in other models of AMD such as the dsRNA model of retinal degeneration. Using that model of photoreceptor and RPE cell death, we found that the predominant cell death modality may be different between photoreceptors and RPE. Photoreceptors appeared to die predominantly through apoptosis, whereas RPE cells exhibited mainly necrotic features.174 Similar to our prior work, inhibition of caspases or RIPK in isolation was less effective than combination in preventing overall photoreceptor and RPE cell death after dsRNA-induced injury. The significant role of RIPK in RPE cell death was also observed in an in vitro model of RPE toxicity induced by tamoxifen. Pan-caspase inhibition failed to protect RPE cells, whereas addition of RIPK inhibitors, alone or in combination, led to significant RPE survival.175
In contrast to apoptosis, regulated necrosis is more inflammatory. We recently found evidence of increased inflammasome activation in patients with photoreceptor injury due to retinal detachment.172 Using an animal model of retinal detachment, we showed that the primary source of inflammasome activation and production of IL-1β was a partially RIPKdependent pathway from the infiltrating macrophages rather than the injured retinal cells.172 Additionally, we found that infiltrating macrophages (and maybe resident microglia) expressed FasL that was responsible for increased neuronal cell death. In contrast, soluble FasL was found to be neuroprotective.160
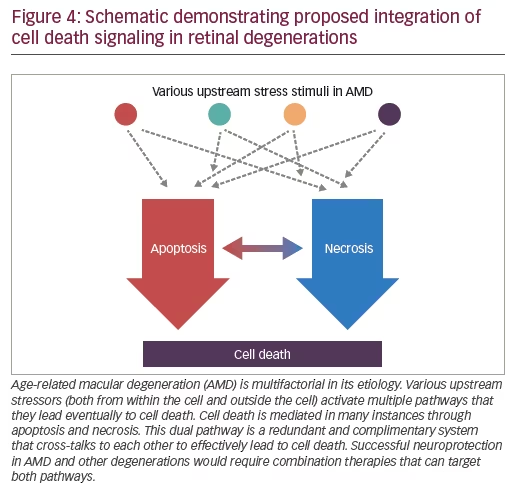
Thus, past neuroprotection strategies may have failed in part because of the focus on monotherapy. With the recognized redundancy of cell death pathways, combination therapy to block both pathways may be more effective (Figure 4). Neuroprotective strategies based on the above findings may apply to both dry AMD and wet AMD. Adjuvant neuroprotective therapy, along with anti-VEGF treatment, may prevent photoreceptor cell death in neovascular AMD, possibly leading to improvement in both short- and longterm vision outcomes. Furthermore, if successful, this type of combination therapy may also provide a broad-based, long-term treatment approach for a variety of retinal disorders.
It should be noted that evidence of specific end-stage cell death processes in human AMD specimens is sparse. This is inherently difficult since only a very limited number of cells are in the process of cell death at any given time point due to the slow process of the disease. As an example: assuming RPE cell death accounts for the observed GA growth rates of 1.85 mm2/year, and knowing that macular RPE cell density is about 5,000 cells/mm2,176,177 we can conclude that just over 9,200 cells are dying per year. Using these calculations, the rate of cell death would be only 25 cells dying at any given day or approximately one cell dying per hour. As such, detecting end-point death signals in autopsy specimens is a tall order in the analysis of human AMD specimens.
Biomarkers
Identification of patients with AMD in earlier stages of the disease and prediction of individual progression rates will be of paramount importance for successful management of the disease. Previous attempts to identify serum biomarkers (C-reactive protein, homocysteine, and lipids) to identify patients with AMD and that correlate with disease progression yielded inconsistent data.8,178–180 More recently, researchers have looked into more systematic and unbiased approaches of finding biomarkers through metabolomics. Metabolomics is the study of all the metabolites (metabolome), the small molecule “fingerprints” of cellular processes. While genomic analysis gives us a snapshot of DNA code variation, and proteomics the set of gene products being produced in the cell, metabolomics enables us to study the relationship between genotype and phenotype, as well as the environment including nutrition and commensal organisms. It has been used to determine biofluid (blood and urine) marker profiles for several diseases, including cancer and Alzheimer’s disease, and may provide an integrated biomarker signal for AMD.
In a recent metabolomics study at Massachusetts Eye and Ear, patients with AMD and without vitreoretinal disease (age >50 years) were recruited prospectively, examined, imaged, and a fasting blood sample was collected for metabolomics analysis.181,182 Study results revealed that, after controlling for age, gender, body mass index, and smoking status, 87 metabolites were significantly associated with AMD. Most of these metabolites (82.8%, n=72) belonged to the lipid super-pathway, particularly glycerophospholipids. Of the different metabolites between control and AMD patients, over half (48 metabolites) also differed significantly across AMD severity stages. Consistently, in patients with AMD versus control patients, and among the various stages of AMD, the vast majority of the identified metabolites were involved in lipid metabolism. These results led to further support for the importance of lipid metabolism, specifically glycerophospholipid metabolism, in AMD and suggest that metabolomic profiling is a potentially powerful tool to identify AMD, and to provide prognostic information and precise treatment.
Future advances in treatment
The lessons we learned from our successes in the development of therapies for neovascular AMD is that effective therapeutics arise either from understanding the pathogenesis of the disease or at least the key components of shared processes of complex multifactorial diseases. For example, despite the different causes of neovascularization, it is the same molecule—VEGF—that drives the process of new vessel formation. This latter understanding was crucial in leading not only to the success in treatment of neovascular AMD, but also to other retinal diseases. Understanding of common and shared pathogenetic processes in photoreceptor and RPE degeneration will be needed before we can be successful in generating the next generation of therapies in non-neovascular “dry” AMD. This can be achieved with better classification, better disease biomarkers, and improved basic science understanding of cell death machinery. There is no doubt that it is a matter of time before success arrives upon us.







