Proceedings of a Symposium Presented at the 14th EURETINA Congress, London, 12th September 2014
Diabetic macular oedema (DMO) is a serious threat to vision that is showing increasing prevalence as a consequence of the worldwide increase in both type I and type II diabetes.1 DMO affects approximately 7 % of people with diabetes and is responsible for 12 % of new cases of blindness each year.2,3 In many cases, diabetes leads to diabetic retinopathy (DR) approximately 10 years after disease onset and this can lead to DMO. In some cases, however, DMO can occur without DR. Effective means of controlling DMO to prevent widespread vision loss is a growing need. Until recently, the only treatment that was available for the management of DR and DMO was argon laser photocoagulation but now new pharmacological treatments are emerging that are improving the understanding of this disease and enabling better patient outcomes.4,5 Two classes of intravitreal injections include inhibitors of vascular endothelial growth factor (VEGF) and corticosteroids. These treatments provide a much improved choice for the management of DMO and, in the case of corticosteroid treatments, can sustain long-term improvements (in visual acuity and reduced macular oedema) for up to 3 years.6 In some patients, such medicines are used too late in the disease course and optimal outcomes in terms of vision and retinal thickness are consequently not achieved.7–9 This is one of the matters that was discussed at the symposium held by Alimera Sciences at the 2014 EURETINA congress in London. This article reports the proceedings from this symposium.
Objectives of the Symposium
The symposium aimed to consider the inflammatory processes involved in chronic DMO, to review the clinical trial data supporting the use of fluocinolone acetonide (FAc) implant therapy and to report important interim findings from a ‘real world’ study of this treatment in early regular clinical use (non-randomised, open label, single-centred, phase IV efficacy and safety study) that is in progress at a treatment centre in France.
The Role of Inflammation in Chronic Diabetic Macular Oedema
As diabetes progresses over time inflammatory complications, including DR and DMO, substantially worsen.10 In many patients DR appears first and this subsequently leads to DMO. In some cases DMO has been observed without DR suggesting alternative routes to this condition. When DR manifests, increased circulating levels of VEGF are initially observed but as the severity of the condition worsens (decreasing Early Treatment Diabetic Retinopathy Study [ETDRS] scores) these levels remain relatively stable.11,12 However, levels of other inflammatory markers, such as procoagulants, markers of endothelial dysfunction and angiogenesis increase relative to VEGF.13,14 As a consequence, the duration of diabetes has been correlated with the development and progression of DR. This was shown in the Wisconsin Epidemiology Study of Diabetic Retinopathy,15 which included 1,585 patients with diabetes, who were taking insulin and were diagnosed with DR before 30 years of age. This population-based study showed that progression to proliferative retinopathy and clinically significant macular oedema was significantly related to increasing severity of retinopathy at baseline (p<0.001).16 Further evidence of this was shown in the Diabetes Control and Complications Trial (DCCT correlated)17 that included 1,441 patients with insulin-dependent diabetes, type 1 diabetics, aged 13–39 years. The results indicated that during the 8.5 years of follow-up, DR increased with duration of diabetes but the cumulative incidence of DR was significantly decreased with intensive diabetes therapy (i.e. with an external insulin pump or by three or more daily insulin injections) compared with conventional therapy (one or two daily insulin injections).
In patients with diabetes there are multiple processes leading to DR and DMO among which inflammation is important but is not the only mechanism involved. Clinical studies have shown that the effects of diabetes are multifactorial: intraocular levels of various cytokines associated with inflammation and angiogenesis increase with greater severity of DR. The main interleukins that are associated with DR and DMO include: interleukin-1β (IL-1β) (lymphocyte activation and autoinflammation), IL-6 (increases vascular permeability) and IL-8 (increases vascular permeability and simulates inflammatory proteins).18,19 Other important cytokines in DR and DMO are monocyte chemo-attractant protein-1 (MCP-1, increases vascular permeability altering tight junctions increasing cellular recruitment), interferon gamma-induced protein-10 (IP-10, a chemo-attractant for monocytes, macrophages, T cells, natural killer cells and dendritic cells), platelet-derived growth factor (PDGF, promotes embryonic development, cell proliferation, cell migration, and angiogenesis) and VEGF (stimulates angiogenesis, vasodilation and is a chemo-attractant for macrophages and granulocytes).20–24 A study by Dong et al.11 showed that among consecutive patients with diabetes (n=136), levels of IL-1β, IL-6, IL-8, MCP-1 and IP-10 in the aqueous humor increased with the severity of DR. These levels were also significantly higher in patients with type II diabetes compared with patients without diabetes controls (p<0.001 for all these cytokines). With increasing severity of DR (10–80 on the ETDRS scale) there was little increase in VEGF levels (p=0.733), but there were a significant increases in levels of IL-1β (p=0.003), IL-6 (p<0.001), IL-8 (p=0.001), MCP-1 (p<0.001) and IP-10 (p<0.001). Levels of many of these cytokine factors can be reduced with intraocular steroid therapy. A
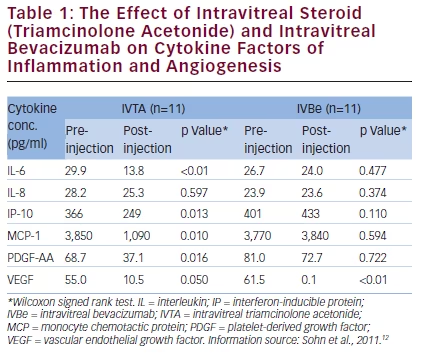
study by Sohn et al.12 showed that in aqueous humor samples, levels of cytokines were significantly raised in patients with DMO. Eleven patients (mean age 54.5 years, with type II diabetes, DR, DMO and no previous vitreous surgery or photocoagulation) were treated with intravitreal triamcinolone acetonide (TA) in one eye and intravitreal bevacizumab in the other eye. Aqueous humor samples taken 4 weeks later showed that in the eyes treated with TA, there were significantly reduced concentrations of IL-6, IL-10, IP-10, MCP-1, PDGF and VEGF. This demonstrated the multifactorial effect of corticosteroid treatment. The eyes treated with bevacizumab, however, showed only a specific reduction in VEGF (p≤0.05) but no reduction in the other cytokines monitored (see Table 1).
A main approach to treating DMO is with the use of intravitreal anti- VEGF therapy such as ranibizumab25 or the recently approved agent, aflibercept.26 Ranibizumab is a monoclonal antibody therapy that is approved for DMO and was evaluated in the RIDE (NCT00473382) and RISE (NCT0047330) studies (n=759). Patients with DMO were originally randomised to ranibizumab 0.3 mg/day, 0.5 mg/day or sham treatment.27 After 24 months, there were marked differences between ranibizumaband sham-treated groups in terms of visual acuity (VA) (improvements in mean best-corrected VA [BCVA] from baseline for ranibizumab 0.5 mg, 0.3 mg and sham/0.5 mg were 12.0, 10.9 and 2.3 for RIDE, and 12.5, 11.9 and 2.6 for RISE, respectively). At 24 months, the sham-treated patients were switched to ranibizumab 0.5 mg/day. Over the following 12 months, however, the patients who were switched failed to show the level of improvements in BCVA scores that were seen in the patients treated with ranibizumab from the start (improvements in BCVA scores from baseline for ranibizumab 0.5 mg, 0.3 mg and sham/0.5mg were 11.4, 10.6 and 4.6 for RIDE, and 14.2, 11.0 and 4.3 for RISE, respectively). This showed that delaying ranibizumab in DMO may have a negative effect on outcomes and that this treatment may be less effective in more advanced disease. These results are consistent with the principle discussed above that in chronic DMO, inflammation is up-regulated and to achieve a sufficient response a multifactorial approach, targeting multiple inflammatory cytokines, may be needed.
Further evaluation of ranibizumab was provided by results from the Ranibizumab (intravitreal injections) in RESTORE extension study, (NCT00687804), which in addition to primary visual acuity endpoints, enabled a comparison of early administration of ranibizumab versus 12-month delayed ranibizumab in the treatment of DMO.28 Patients were originally randomised to ranibizumab 0.5 mg/day, ranibizumab 0.5 mg/ day + laser or laser alone for 12 months. Patients (n=240) then entered the 24-month open-label extension study in which ranibizumab-treated patients continued treatment but laser-treated patients were switched to ranibizumab. At the end of the 12-month period, the mean change in BCVA from baseline in the three groups were 7.9, 7.1 and 2.3, respectively. At the end of the extension study (at 24 months), however, these scores were: 8.0, 6.7 and 6.0. This showed that patients who crossed over from laser to ranibizumab treatment at 12 months experienced the same benefit as those who were treated early. It should be noted, however, that patients recruited to this study were not required to have persistent DMO so the cohort included those with early-stage disease. In this study, therefore, anti- VEGF therapy may have been effective in early-stage DMO but for more progressive disease, this switching strategy was not effective. Therefore, in more chronic cases, the response to anti-VEGF may be blunted and a second-line therapy such as a corticosteroid should be considered.
The above responses have been observed in the Fluocinolone Acetonide for Diabetic Macular Edema (FAME) studies.29 Indeed, in the sham control group, rescue laser (61.6 and 62.5 %) and off-protocol treatments (34.8 and 30.6 %) were given in similar proportions to chronic and non-chronic patients, respectively. However, improvements were quite different with only 13.4 % of chronic DMO patients achieving ≥15-letter in BCVA from baseline compared with 27.8 % of non-chronic DMO patients. Thus, intermittent therapies, such as rescue laser and off-protocol treatments, were effective in patients in whom DMO had been diagnosed more recently but less effective in patients with longer standing DMO. This indicates that different treatment approaches are needed to successfully manage chronic DMO and achieve optimal outcomes for the patient.
Albert Augustin: “As DR worsens with time, the incidence of DMO increases which, in chronic cases, is no longer mediated by VEGF but by pro-inflammatory cytokines. In these patients, steroids provide an effective therapeutic option”

Overview of ILUVIEN® (0.2 μg/Day Fluocinolone Acetonide Implant) in Chronic Diabetic Macular Oedema Yit Yang, Wolverhampton Eye Infirmary, New Cross Hospital, Wolverhampton, UK
ILUVIEN® (FAc implant) is a very long-acting, second-line therapy indicated for the treatment of chronic DMO. The product consists of a non-bioerodable micro implant (dimensions: 3.5 mm × 0.37 mm) that contains 190 μg of FAc and delivers 0.2 μg FAc/day for up to 36 months. The implant is administered intravitreally using a 25-gauge injector, which creates a self-sealing wound. After a brief burst, levels of FAc in the aqueous humor have been shown to stabilise by 6 months (1.18 ng/ml) and are sustained through to 36 months (0.55 ng/ml).30,31 The FAME studies recruited patients with DMO that was unresponsive to previous macular laser treatment. Patients were also required to have a foveal thickness ≥250 μm despite at least one prior focal/grid macular laser photocoagulation treatment and BCVA in ETDRS letter score between 19 and 68 (Snellen equivalent range, 20/50–20/400).32
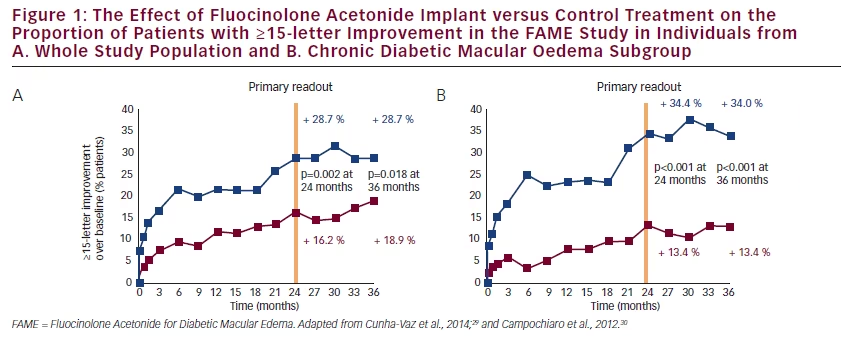
Treatment with 0.2 μg/day FAc implant (n=375) led to rapid improvements in retinal thickness compared with sham (control) treatment (n=185). At 6 weeks, foveal thickness had rapidly decreased from 461.1 to 345.7 μm (–115.4 μm) in the 0.2 μg/day FAc implant-treated group compared with a change from 451.3 μm to 450.4 μm (–0.9 μm) in the sham control group. VA was also significantly improved by the FAc implant. The proportions of patients with a ≥15-letter improvement after 24 months was 28.7 % and 16.2 % (Δ, 12.5 %; p=0.002) and after 36 months was 28.7 % and 18.9 (Δ, 9.8 %; p=0.018) in FAc implant- and sham-treated patients, respectively.30 The FAME studies also show the efficacy of the FAc implant is greatest in patients with chronic DMO. This is the population in which the FAc implant is indicated. In these patients the proportions with a ≥15-letter improvement at 36 months were 34.0 % and 13.4 %, respectively (Δ, 20.6 %; p<0.001) (see Figure 1).29,30 These results indicate that the FAc implant is an effective treatment in a group of patients who have not responded to prior treatment with first-line therapies such as laser.
Common side effects of corticosteroids are the potential for cataract and raised intraocular pressure (IOP). These were both managed effectively in the FAME studies.33 Indeed, 38.4 % of FAc implant-treated patients needed IOP-lowering drops compared with 14.1 % of controls. Therefore 62 % of patients treated with 0.2 μg/day did not require IOP therapy and when IOP-lowering treatment was given, it did not affect vision. This was demonstrated in the FAME studies, where the mean change in BCVA was similar when patients receiving chronic IOPlowering therapy (taken for >28 days; n=144) were compared with those that were not (n=230).34 It should be noted that the UK National Institute for Health and Clinical Excellence (NICE) recommends the FAc implant for use in pseudophakic patients due to the associated cost of performing cataract surgery.35,36 In other countries in Europe, such as Germany, there is no restriction on lens status and the FAc implant can be used as indicated in the ILUVIEN Summary of Product Characteristics.
Yit Yang: “IOP rise during steroid treatment is not a disaster – this and cataract removal can be managed with good visual outcomes”

Understanding and Managing Steroid-induced Elevation of Intraocular Pressure in the Context of the FAME Studies
Michael Diestelhorst, University of Cologne, Cologne, Germany
One side effect of chronic topical or systemic treatment with steroids is reduced outflow facility of the trabecular meshwork. As a consequence, the IOP may rise significantly within weeks. If not treated appropriately these eyes may show significant glaucomatous optic nerve head (ONH) cupping. Thus steroid glaucoma could be termed ‘high-speed open angle glaucoma’. It may cause blindness within months. Such patients have no pain even when the IOP is 50 mmHg. When possible, the steroid application should be stopped. If the ONH and visual field (VF) are normal and the IOP is up to 25 mmHg, eyes should be controlled without antiglaucomatous medication. At higher IOP levels, topical or systemic IOP-lowering therapy is needed. If this does not reduce the IOP level to normal, ocular surgery should be considered.
Elevated IOP is often associated with intravitreal corticosteroid treatment. In the FAME studies, steroid-induced ocular hypertension was limited and controlled among patients who received an FAc implant. Among the FAc implant-treated eyes, 43.1 % had a maximum IOP <21 mmHg (it should be noted that patients with IOP >21 mmHg or those receiving IOP-lowering drops at baseline were excluded).34 Patients receiving this treatment with an IOP up to 25 mmHg are considered to be ‘low responder’ and could be compared with ‘ocular hypertensives’ when the VF and the ONH are normal (see European Glaucoma Society Guidelines).37 In the FAME studies, 67.3 % of patients had an IOP <25 mmHg. The proportion of patients with an IOP above 30 mmHg (‘high-responder’) in the FActreated group was 18.4 % versus 4.3 % in the sham-control group.
Analysis of the time to the first treatment with IOP-lowering drops shows that roughly 25 % occurred within the first 3 months and 70 % within the first 12 months.30,34 However, frequent IOP readings are mandatory within the first 6–12 weeks after treatment is started. If the IOP increases >25 mmHg, treatment should be initiated. Among FAc implant-treated patients, 62 % did not require IOP-lowering eye drops. A summary of the topical anti-glaucomatous medication prescribed in the FAME studies is shown in Table 2. The most frequent of these were prostaglandins, beta blockers and topical alpha 2 agonists. Combination therapies were also prescribed. The usage of IOP-lowering therapies
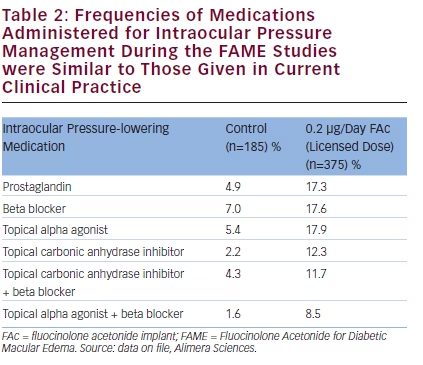
indicates that steroid-induced IOP elevation occurs in a minority of patients who receive the FAc implant. Appropriate monitoring and treatment is important to manage this side effect without detrimental effect on the ONH.
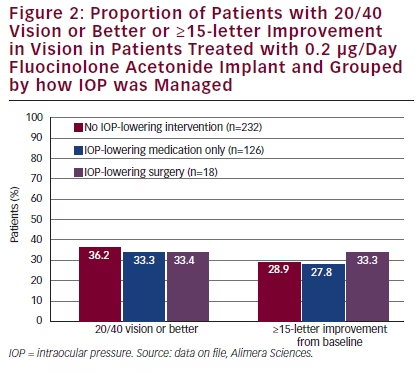
In the FAME studies, 95 % of patients treated with an FAc implant did not require incisional IOP-lowering surgery.30 Trabeculoplasty was performed in 1.3 % of FAc implant-treated and in 0 % of control patients. Four patients had laser trabeculoplasty 574–776 days after the study start, which successfully reduced IOP. Prior to trabeculoplasty, the IOP was 32.8 mmHg and decreased to 17.5 mmHg after the procedure and was 18.5 mmHg after 360 days. This emphasises that topical anti-glaucomatous therapy is not sufficient in ‘high-responders’. Of the four laser patients, three required no incisional treatment. One patient who had been treated with an FAc implant required additional IOP-lowering surgery. Elevated IOP had little effect on VA. The proportions of patients with 20/40 vision or better or had a ≥15-letter improvement in BCVA score from baseline were similar for those who received IOP-lowering drops or surgery to those who received no IOP-lowering treatment (see Figure 2).
Michael Diestelhorst: “In case of raised IOP: after removal of the FAc implant there is a good chance that IOP will return to normal within weeks”
Real-world Experience with the 0.2 μg/Day Fluocinolone Acetonide Implant
Pascale Massin, Hôpital Lariboisière, Paris, France
At the time of writing, the FAc implant has been launched in the UK and Germany and licensed in the UK, Germany, Austria, France, Portugal, Spain, Italy, Norway, Sweden and Denmark.38 Licences are still pending in the Netherlands, Belgium, Luxembourg, Finland, Ireland, Poland and Czech Republic.39 Furthermore, as of 26 September 2014, the FAc implant has been approved for use in DMO by the US Food and Drug Administration (FDA). Current experience of regular clinical use of the FAc implant is limited and this study was conducted to gain real usage experience in France. This study was designed to assess the efficacy and safety of the FAc implant in DMO patients considered insufficiently responsive to available therapies (laser, anti-VEGF) with or without prior history of corticosteroid therapy. Intermediate results after month 1 are reported here.
This phase IV study is an ongoing non-randomised, open-label, singlecentre, investigation in a planned population of 20 eyes. It started in January 2014 and is a 1-year study with assessments at 1 week, and 1, 3, 6, 9 and 12 months. At entry, patients were divided into two groups: Group 1, patients with prior laser treatment only (i.e., no anti-VEGF); and, Group 2, patients with prior laser and three or more monthly anti-VEGF treatments. Inclusion criteria included: VA between 20/60 and 20/400, central foveal thickness ≥350 μm and DMO that was unresponsive to laser photocoagulation (≥3 months prior to screening) or unresponsive to anti-VEGF therapy. Exclusion criteria included: IOP ≥21 mmHg at screening, a history of IOP increasing to ≥25 mmHg following prior intravitreal steroid therapy or the use of ≥2 IOP-lowering drops.
At baseline, patient characteristics in the two groups were similar; however, Group 1 (n=7) had a longer duration of DMO than Group 2 (n=10) (7.6 versus 3.6 years, respectively) and more than 80 % of patients were pseudophakic. Interim analysis showed a greater gain of VA in Group 2 compared with Group 1, but the duration of chronic DMO was shorter in Group 2 than in Group 1. In terms of anatomical response, decreases in macular thickness were observed in all the eyes in Group 1 as compared with Group 2 where responses were heterogeneous. In Group 2, the average decrease of the macular thickness was less marked versus Group 1. After 1 month, these changes in foveal thickness and BCVA were similar to those seen in the FAME studies.
Overall, these findings show that the FAc implant was beneficial to patients in this ‘real world’ study whether or not they had received prior anti-VEGF therapy and laser photocoagulation. The better VA outcomes in Group 2 suggest that it may be beneficial to administer the FAc implant earlier in the disease course. Interim analyses at subsequent time points and at the end of the study will be reported at a future date. Pascale Massin: “In the two [FAc-treated] groups we observed a reduction in macular thickening.” “We now know that we should probably have treated these chronic DMO patients [with FAc] at an earlier stage than we did in this ‘real world’ study” Conclusion The symposium objectives, see above, were met and valuable insights into chronic DMO pathophysiology and effective treatment with corticosteroids were provided. The FAc implant produces rapid and marked improvements in VA and retinal thickness. The interim ‘real world’ study results seem to support those found in the FAME studies, which showed marked gains in both VA and retinal pathology in patients with chronic DMO after only 1 month. In particular, these ‘real world’ data emphasise that the FAc implant is effective after prior treatment with laser and anti-VEGF. Clinical evidence indicates that several therapies provide benefits in the treatment of DMO but these treatments may be appropriate at different stages of the disease. As DMO progresses and becomes more chronic, the pathophysiology of the disease changes and different inflammatory and angiogenic pathways become activated. While some patients respond well to anti-VEGF therapies, in chronic disease they may become insufficiently responsive meaning a suboptimal response is being achieved. The interim study defined this as being an insufficient response to three or more monthly anti-VEGF treatments. In the interests of the patient, it is appropriate that this insufficient response is detected and a second-line therapy considered. The FAc implant is a second-line therapy and has been shown, in the FAME studies and now in a real-life setting, to produce rapid and marked improvements in vision and foveal thickness in chronic DMO patients that are not responding optimally to prior therapies. In Europe the FAc implant is approved for chronic DMO patients who are insufficiently responsive to available therapies, while NICE recommends that it should only be used in pseudophakic patients based on costeffectiveness grounds rather than benefit-to-risk. The administration of intravitreal corticosteroids is associated with cataract, elevated IOP and floaters. While these effects can be adequately managed, the more advantageous safety profiles of the anti-VEGF medications make them more suitable for first-line use. Nevertheless, the administration of FAc implants is an effective therapy for patients who do not respond to initial anti-VEGF treatment. The FAc implant is a second-line therapy. Experience suggests that it is effective in chronic DMO patients but optimal effects may be gained if the treatment is initiated once a patient has been identified as being insufficiently responsive to prior therapies such as laser and anti-VEGF. Such treatment could dramatically improve outcomes such as gaining 20/40 vision, which is the minimum UK eyesight standard for driving. It could also allow individuals such as bus and lorry drivers to remain in employment (needing VA of at least 0.8 [6/7.5] Snellen scale in the best eye and at least 0.1 [6/60] on the in the other eye40). DMO is an increasingly serious threat to vision worldwide. One FAc implant provides beneficial improvements in VA and foveal thickness that last for up to 36 months with one injection. FAc implants are indicated for use in chronic DMO patients. DMO is a complex disease involving multiple pathways and the FAc implant addresses the multifactorial nature of DMO as it affects multiple inflammatory cytokines that are upregulated as DMO progresses. Early identification of non-responsiveness to prior therapies is particularly helpful for the physician as it helps guide subsequent and appropriate treatment options that meet their patient’s needs. It also potentially provides insight into the underlying state of the condition. This helps improve the patient outcomes and potentially helps preserve longer-term vision.








