Understanding the severity of keratitis and its impact in severe dry eye disease
The target mechanisms that drive the pathophysiology of dry eye disease (DED) are principally tear film instability, tear hyperosmolarity, apoptosis and inflammation. These factors, respectively, lead to cell damage, nerve stimulation, cytokine release, matrix metalo-proteinase activation and goblet cell loss and form a ‘vicious circle’ that can repeat multiple times.1,2 DED is rarely just a tear problem; it is usually related to disruption of the ocular surface and altered activity in the corneal epithelium. In DED, there is usually increased production of pro-inflammatory cytokines, release of prostaglandins and leukotrienes that activate leukocytes and enzymes. In addition, there is expression of adhesion molecules responsible for leukocyte tissue infiltration and expression of major histocompatibility complex (MHC) class II peptides that are responsible for antigen presentation and T lymphocyte activation.3 In addition, natural or artificial tears will not form the normal film over the epithelium, resulting in dry eyes, vulnerability to infection and corneal damage.4 It is, therefore, essential to address corneal problems in patients with DED. Inflammation of the cornea (keratitis) is itself a source of further inflammation and thus the process is self-sustaining.
The air–tear interface is the most important refractive interface in the eye and the cornea is responsible for two-thirds of optical power. DED and keratitis, however, can degrade the quality of vision, increase secondary infection risk and reduce quality of life in DED patients.5,6 This was emphasised in a study of 52 patients with DED, in which reading speeds were significantly reduced compared with normal control subjects, and this effect worsened with severity of disease.7 This study also showed that central corneal punctate keratitis significantly affected reading speed compared with peripheral staining or normal corneas.
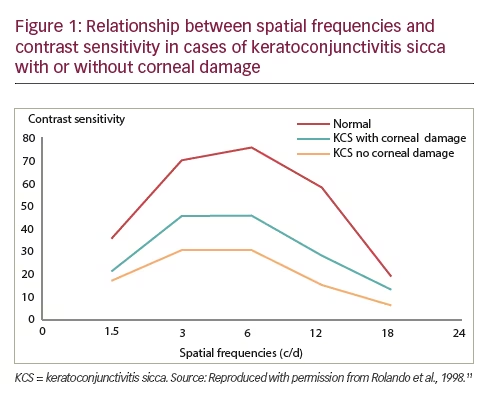
Several studies that included a combined total of approximately 90 patients with epithelial disease showed that the increasing ocular surface damage is associated with decreasing level of contrast sensitivity and this has proven to be a useful measure of DED severity (see Figure 1).8–11 In another study, 20 patients with DED were found to have visual impairment that diminished their capability to drive compared with 20 age- and sex-matched controls (p<0.01).12
Corneal epithelium changes in dry eye disease
The corneal epithelium is highly impermeable, largely due to the tight junctions between cells at the surface. This creates a potential difference across the epithelium. The surface is charged negatively compared with the interior, which has a more positive charge.13 This is advantageous because most environmental pathogens are negatively charged at the surface and so are not attracted to the corneal surface. If the tight junction is interrupted, as it is in DED, this repulsion is lost, allowing ingress of bacteria. Analysis of corneal surfaces in patients with DED reveals epithelial punctate keratopathy, sub-epithelial nerve plexus damage and stromal keratocyte activation. Such corneas also show sub-epithelial invasion with mature dendritic cells.14,15 This indicates a profound change in the immune condition of the cornea. In addition, in inflamed corneas in DED there is a change in the appearance of corneal nerves which show increased tortuosity, reduced length, decreased thickness and increased beading.
Markers and grading scales to address challenges in severe dry eye disease diagnosis
For assessment of DED, epithelial punctate keratopathy is an important feature for grading the level of chronic corneal damage and the severity of the disease.16,17 In addition, the epithelial cells of the cornea express inflammatory markers including matrix metallopeptidase 9 (MMP9), interleukin-1 (IL-1), IL-6, IL-8, IL-16, IL-33, human leukocyte antigen-antigen D related (HLA-DR), CD69, CD154 and transforming growth factor-alpha (TGF-α) and these factors all have potential as laboratory indicators of disease.18–24 In addition, lissamine green and fluorescein stains (used with a Wratten 12 filter) enable the visualisation of keratitis. The diagnosis and severity of disease using these stains is assisted using one or more grading scales including the Oxford (most commonly used), National Eye Institute (NEI), SICCA Ocular Staining Score, Efron etc.25–28 These use different criteria to produce a severity score within a defined scale. The Oxford scale gives some indication of the position of staining on the ocular surface.25 The NEI-Industry Workshop in 1995 produced a more precise scale to more accurately record the position and extent of staining.27 There is, however, no universal consensus among investigators and clinicians on the best single grading scale to use in DED.
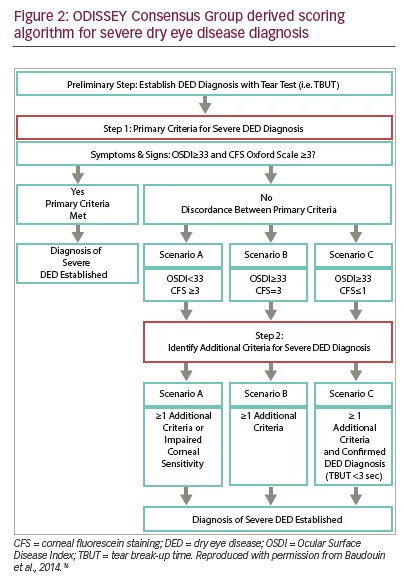
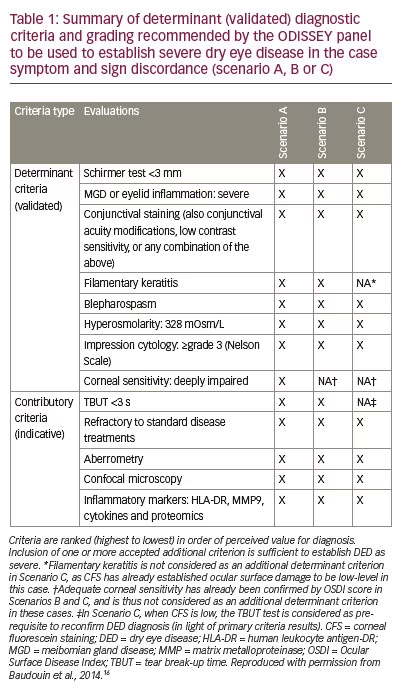
In many patients there is discrepancy between signs and symptoms in DED making diagnosis and the assessment of treatment efficacy difficult. To address this issue the ODISSEY Consensus Group proposed a new approach for severe DED diagnosis.16 This group of experts in the field of DED from across Europe devised an algorithm that could be used when there is discordance between primary assessment criteria (symptom-based assessment and corneal fluorescence staining [CFS]). This algorithm consists of three scenarios that would determine additional criteria recommended to further evaluate DED severity. These are: scenario A (Ocular Surface Disease Index [OSDI] <33 CFS ≥3), scenario B (OSDI ≥33, CFS=2) and scenario C (OSDI ≥33, CFS ≤1). In each of these scenarios additional criteria are required to establish a diagnosis of severe DED. A schematic of this algorithm is given in Figure 2 and the additionally required criteria are listed in Table 1. Use of this scheme may help in the diagnosis of DED.

Inflammation – the new target to treat dry eye disease
DED is not a syndrome but a disease of the tear film that involves inflammation, which is a ubiquitous mechanism in ocular surface diseases (OSDs). The International Dry Eye Workshop (DEWS I) provided a consensus definition of DED in 2007.29 DEWS II met in September 2016 and provided an updated definition: ‘Dry eye is a multifactorial disease of the ocular surface characterised by a loss of homeostasis of the tear film, and accompanied by ocular symptoms, in which tear film instability and hyperosmolarity, ocular surface inflammation and damage, and neurosensory abnormalities play etiological roles.’
The variable signs of dry eye disease
In DED, signs of inflammation can be difficult to identify even though it plays a key role; there is often a lack of redness, swelling or pain (which often feels ‘uncomfortable’ rather than ‘painful’) that are the classical signs of inflammation. There are two main classes of dry eye, which are aqueous deficient (ADDE) and evaporative (EDE) types. In ADDE, there is a reduction in tear flow and volume, whereas in EDE tear production is normal but there is increased evaporation.30 In a recent retrospective study of 224 patients with DED conducted in the US, 79 (35%) were found to have EDE (caused by meibomian gland dysfunction [MGD]), 23 (10%) had ADDE and the remaining 55% had a mixed or unknown type.31 MGD is the leading cause of EDE.32 Among ADDEs, Sjogren’s syndrome (SS) is a frequent cause in addition to lacrimal deficiency, lacrimal duct obstruction, reflex block and systemic drugs.
The pathophysiology of dry eye disease
In the proposed ‘vicious circle’ mechanism of DED, tear film instability or deficiency leads to hyperosmolarity, cell damage, apoptosis, nerve
stimulation, inflammation, cytokine release and goblet cell loss which in turn, leads to greater tear film instability.1 These factors are affected by a range of disease, lifestyle, drug and environmental factors. When such influences are chronic, or multiple factors are present, the patient can enter a self-stimulated loop that maintains or worsens the disease.33 MGD is affected by these same factors but is also affected by an additional vicious circle involving meibomian gland blockage/inflammation, leading to bacterial flora changes, esterase and lipase changes and lipid alterations, which result in further meibomian gland blockage.1,34
The mechanisms of DED are increasingly recognised as more wideranging than was previously understood. A new concept is that DED is a mucosal autoimmune disease that is not restricted to the ocular surface but involves the lacrimal glands, corneal and conjunctival epithelia and meibomian glands that form the ocular surface system. In addition, the disease involves the innate and adaptive immune systems. Stress to the ocular surface results in a pro-inflammatory cytokine response and upregulation or exposure of autoantigens (e.g. M3R and KLK13).35,36 This results in self-antigen recognition through B-cell receptor antigen presentation, leading to autoantibody presentation with consequent tissue damage. In addition, in DED, mechanisms that maintain homeostasis of the ocular surface, which are mediated by nerve connections and systemic hormones, are disrupted.14
Immune aspects of dry eye disease
The importance of the immune component in the DED mechanism has been demonstrated in various in vitro, in vivo and clinical studies and these increase understanding of the mode of action of a newer treatment approach. In one study, human conjunctival cells were exposed
to hyperosmolar conditions which induced an increase in secretion of the chemokine C-C motif ligand 2 (CCL2).37 This increase, however, was substantially reduced with the addition of either ciclosporin A (CsA) or dexamethasone. Further evidence of the mechanism of CsA action in DED was provided by a study of clinical conjunctival biopsies.38 Biopsies from patients with DED treated with topical CsA showed decreases in cells positive for cluster of differentiation 3 (CD3), CD4, CD8 antigens compared with increases in control-treated patients. In addition, CsA treatment also significantly reduced lymphocyte activation markers CD11a and HLA-DR compared with controls (p<0.05).38 Biopsies from patients with the autoimmune disease, SS showed significant decreases in CD11a (p<0.001) and CD3 (p<0.03) after CsA treatment.
In a large study that included 311 patients, flow cytometry on cells from corneal imprints showed that HLA-DR was significantly increased in patients with SS (p<0.0001) or MGD (p=0.0223).39 Levels of HLA-DR were found to be positively correlated with corneal fluorescence staining (r=0.30, p<0.0001), OSDI (r=0.12, p=0.0426 and Visual Assessment Scale (r=0.14, p=0.0176). HLA-DR was negative with the Schirmer test (r=-0.20, p=0.0003) and TBUT (r=-0.13, p<0.0226). The authors proposed that HLA-DR could be used as a surrogate biomarker for DED investigation especially when there is a discrepancy between clinical signs and symptoms. Further evidence came from an in vivo study in which conjunctival cells obtained from DED patients, using conjunctival imprint cytology, showed a 46.16% rise in HLA-DR expression when exposed to hyperosmolar conditions.40 This was compared with a 7.48% increase in cells from normal individuals (p<0.0001) and this response was significantly correlated with tear osmolarity (r=0.614, p<0.0001). These findings were supported further by the phase III SANSIKA trial (n=246) in which patients with DED who received 0.1% CsA showed significant reductions in HLA-DR after 1 month and 6 months of treatment compared with placebo-treated patients (p=0.019 and p=0.021, respectively).41
Other studies have provided evidence that DED is an inflammatory disease. In one example, impression cytology methods and flow cytometry were used to collect and analyse cells from the corneas of 243 patients with DED with or without SS and 50 from control individuals.20 Cells from DED patients with or without SS were shown to significantly overexpress inflammatory and apoptosis-related markers (e.g. HLADR, CD40, transmembrane protein [Fas] and mitochondrial membrane antigen [APO2.7]) compared with cells from normal eyes. These data supported the use of immunomodulatory and or anti-inflammatory drug in DED treatment.
Neuropathic pain – a frequent lasting effect of dry eye disease
Neuropathic pain is a significant issue in DED. Many patients continue to have inflammation and experience severe pain despite showing corneal improvements. Studies using confocal microscopic analysis of ocular tissues have shown abnormal nerve appearance in DED that are associated with lower sensitivity to mechanical, thermal and chemical stimuli.42 It is speculated that in chronic cases these alterations may ‘imprint’ on the trigeminal pathway, creating long-term pain. This raises a series of unanswered questions such as why some patients with DED evolve toward hyposensitivity of the cornea mostly when it is extensively damaged and why sensitivity is retained in others. Other questions include: why continuous corneal nerve stimulation would induce neurogenic inflammation? and whether second order neuron stimulation could play a role even when the cornea recovers or improves?
Recent work in mice has shown that induction of inflammation in the eye using benzalkonium chloride instillation reduced tear production and increased inflammatory cell infiltration.43 This inflammation induced activation of neurons and microglial p38 mitogen-activated protein kinase (MAPK) pathway in the sensory trigeminal complex. This suggested that inflammation could have a priming role in the sensitisation of ocularrelated brainstem nerves, which is a key factor in pain development.
Treatments that target inflammation in dry eye disease
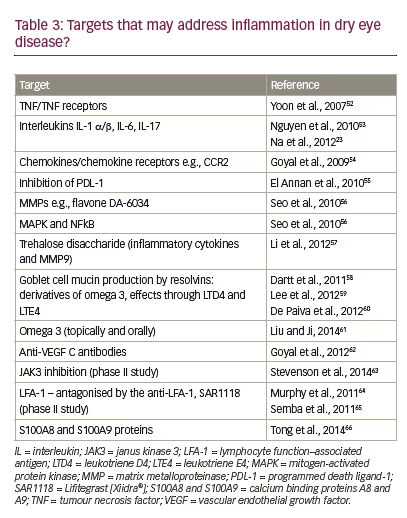
A limited number of treatments that address inflammation in DED currently exist although few are approved for this indication. Of interest are lifitegrast (an integrin lymphocyte function associated antigen binding inhibitor – approved in the US only) and CsA (an inhibitor of T-cell function and IL-2 release). Topical CsA (CsA 0.05%) treatment was approved to increase tear production in patients whose tear production is presumed to be suppressed due to ocular inflammation associated with DED by the US FDA in 2003 and more recently for severe keratitis in DED in Europe in 2015 (Ikervis®, 0.1% CsA, Santen, Evry, France) following successful phase III clinical trials (Table 2).1,41 In addition to these, there is a large series of targets that could be exploited to better address inflammation in DED (Table 3) and drugs that act on these targets may improve future management of DED.
Ciclosporin in the treatment of severe keratitis in dry eye disease – today and tomorrow
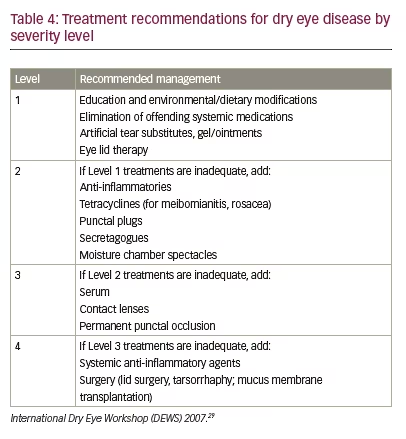
Currently, there is substantial disagreement on the definition and diagnosis of DED. This is due to DED being viewed either as a disease or as a range of different symptoms or ocular surface condition or as a condition that is associated with other conditions.44 This situation may be improved with the forthcoming publication of the revised DEWS definitions and classifications. These guidelines indicate escalating treatments according to DED severity level (Table 4). Differentiating between sequential severity levels, however, can be challenging especially when there is a discrepancy between signs and symptoms as discussed previously.
Ciclosporin A – a key treatment in dry eye disease
A key treatment for severe (level 4) DED is topical CsA. This is a cyclic undecapeptide of fungal origin. It is widely used as immunomodulation to inhibit rejection of organs after transplant surgery and to control some autoimmune disorders. CsA inhibits activation of T lymphocytes and apoptosis by binding to cyclophilin A and D. Topical CsA is a valuable treatment in various ocular conditions including DED, types of keratoconjunctivitis, and types of keratitis.45,46 CsA has notable safety advantages over corticosteroids but has a low solubility in water and is formulated as a topical emulsion.
In the multicentre, randomised controlled, phase III SANSIKA trial, 246 patients with severe DED were randomised to receive daily 0.1% CsA
eye drops lipid emulsion (Ikervis, 0.1% CsA) or vehicle (the same lipid emulsion found in Ikervis but without CsA) for 6 months followed by a 6-month open-label safety follow-up period.41 The primary endpoint was the proportion of patients who achieved ≥2 grades of improvement in CFS and ≥30% improvement in OSDI by month 6. The initial analysis showed a numerical advantage for CsA that was not statistically significant (28.6% for 0.1% CsA versus 23.1% for vehicle treatment [p=0.326]). In addition, the mean OSDI changes from baseline in these groups were similar (-13.6% for CsA and -14.1% for vehicle at month 6 [p=0.858]). However, there was a significant difference in reduction in ocular surface inflammation (assessed by HLA-DR expression) at month 6 (p=0.021) in favour of CsA. The most frequent adverse event was instillation site pain (29.2% for CsA versus 8.9% for vehicle), which was mostly mild.
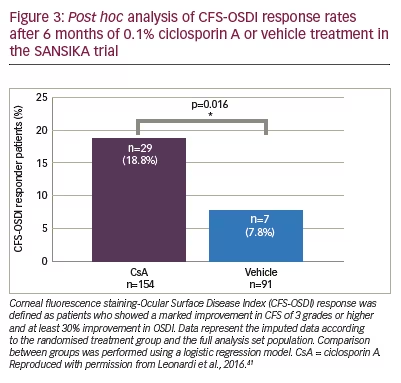
A post hoc analysis of the primary efficacy endpoint (CFS-OSDI responder rates) was conducted to explore a potential treatment effect on patients who had a marked improvement in CFS over 6 months.41 In this analysis, the threshold for improvement of CFS was increased from ≥2 grades to ≥3 grades. Using this more stringent criterion, there were 29 (18.8%) responders receiving CsA versus seven (7.8%) receiving vehicle (p=0.016) (Figure 3), which showed a significant treatment effect.41
The SANSIKA trial, therefore, showed that a topical formulation of 0.1% CsA was well tolerated and effective in improving corneal surface damage and ocular surface inflammation among responders. These benefits were achieved with a single daily instillation that contrast with other DED treatments that require multiple instillations for optimal efficacy.
The benefits of 0.1% CsA in patients with severe DED who have not responded to previous treatments are illustrated in the two typical case
examples below. In both cases symptoms were resolved within a few months after commencing 0.1% CsA topical treatment.


Conclusion
DED continues to be a serious medical burden, causing visual disturbance and discomfort, especially in the elderly. It affects up to 100 million individuals globally, but prevalence varies depending on the populations studied.47–49 The prevalence of the condition is likely to increase as worldwide populations age. Reliable diagnostic methods, clear guidelines for best practice and effective, tolerable treatments are therefore critical in the present and future management of DED. It is increasingly understood that DED is not simply a disease confined to the ocular surface but affects structures of the entire ocular surface system and the trigeminal pathway. As such, DED involves both inflammatory and immune processes operating in a ‘vicious circle’ which serves to maintain the disease after it is initiated by a variety of different factors. Diagnostic and treatment approaches to the disease should take account of these factors to control it effectively.
The lack of consensus on a definition and diagnostic process in DED remains a problem that hampers good management. In addition, clinical experience, as discussed in the sections above, highlights the serious discrepancy between signs and symptoms in DED. Some patients may not present with all the expected symptoms of DED and consequently may not be correctly diagnosed leading to appropriate treatment not being received or being much delayed. It is important that patients with suspected or possible DED are investigated thoroughly using appropriate staining, and graded correctly using a scheme such as that of the ODISSEY Consensus Group, which has attempted to address diagnostic difficulties.16 In DED there is currently a lack of good biomarkers in clinical use. HLA-DR, MMP-9, cytokine profiles, lactoferin/lysozyme and tear osmolarity have potential in the clinical diagnosis of DED51 as shown by clinical studies and case examples. Other potential biomarkers for this disease such as antibodies are now emerging using proteomic approaches.50 Another evolving diagnostic approach in ocular surface disease is confocal microscopy. This technique is largely confined to in vivo research studies but work is in progress to move it from the bench to the bedside.51
0.1% CsA is currently the only approved topical medication for severe keratitis for patients with DED in the EU. Findings from clinical trials support its use in this indication. The efficacy was particularly apparent when a higher threshold of improvement of 3 CFS grades was used in a post-hoc analysis. The treatment was well tolerated with pain at instillation the only frequently reported adverse event. In many patients with DED, however, neuropathic pain can persist for a long time after other symptoms have resolved. This pain may be primed by inflammation but the relationship between nerves and DED needs much further investigation and managing pain continues to be a challenge in DED.
The two case reports show that the treatment is also effective in real-world clinical use. Both patients had severe DED and keratitis and had not responded to previous treatments but improved after 0.1% CsA treatment. The cases also emphasised that corneal disease is reversible but takes time. It is essential to inform patients not to expect rapid resolution of their symptoms.
In future, better consensus approaches, greater adherence to current guidelines, improved, more rapid diagnosis, correct grading and willingness to use appropriate treatments are likely to reduce the burden and enhance outcomes in DED. The use of stem cells also has potential as a treatment approach in DED. Increased knowledge of inflammatory and immune processes has identified a range of potential drug targets. The exploitation of these approaches may eventually lead to a broader range of effective treatments for this increasingly common and debilitating ocular disease.







