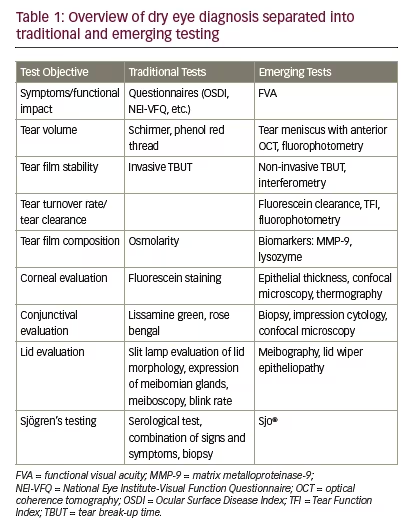
The International Dry Eye Workshop Dry Eye Workshop (DEWS) defined dry eye as “a multifactorial disease of the tears and ocular surface that results in symptoms of discomfort, visual disturbance and tear film instability with potential damage of the ocular surface. It is accompanied by increased osmolarity of the tear film and inflammation of the ocular surface”.1 This condition is divided into two general types: deficient aqueous production by the lacrimal gland and increased evaporation of the tear film, with the latter being more prevalent. It is also common for patients to exhibit a combination of both types of dry eye disease. Despite the seemingly clear definition of the disease, the diagnosis presents many challenges to the practitioner. First, no gold standard protocol for diagnosis exists2 and no one test is sufficient for diagnosis due to poor reliability for many common tests, multiple causative components of the disease and lack of well-defined cut-off values to distinguish disease from normal.2 To further complicate the diagnosis, the signs and symptoms do not always correlate and both can vary based on influences, such as diurnal or seasonal fluctuations.3 In addition, many of the tests are invasive in nature and this may influence the outcome. Lastly, other conditions can mimic dry eye such as ocular allergy. Due to these challenges, alternatives to traditional dry eye testing have emerged. The purpose of this article is to describe the traditional and emerging tests for diagnosis that are clinically useful, including the benefits and limitations and practical pearls. Table 1 lists both the traditional and emerging tests categorised by test objective.
Traditional tests
Symptom questionnaires
Subjective symptoms and their quality of life impact are a critical component of dry eye evaluation. The clinical signs and symptoms do not always correlate and the patients’ experience of their condition is ultimately the most important measure and will cause them to seek treatment. Symptom questionnaires are one of the most repeatable of the dry eye diagnostic tests4 and allow for diagnosis screening, assessment of treatment efficacy and grading of disease severity. Symptom questionnaires are also a critical part of dry eye clinical research trials. The Ocular Surface Disease Index (OSDI) is commonly used, validated and includes 12 questions related to experience during the previous week regarding ocular symptoms, the severity, how these affect visual function and the ocular response to environmental triggers. The score can range from 0–100 with a higher score being worse. A score of 15 has moderate sensitivity and specificity, 60% and 83%, respectively, for the diagnosis of dry eye disease.5 The Standard Patient Evaluation of Eye Dryness (SPEED) questionnaire helps identify symptoms and focuses on their severity and frequency.6 Table 2 provides a summary of popular questionnaires including links to obtain the questionnaires for use.
Practical pearls
• Symptom questionnaires should be used in combination with objective findings to aid in diagnosis.
• These questionnaires can easily be given by a trained technician.
• The The International Dry Eye Workshop Dry Eye Workshop (DEWS) report recommends adopting one of the questionnaires to be used routinely in the clinic setting for screening purposes.4
Schirmer test
The Schirmer test is a common, cheap and easy clinical test to perform to indirectly measure tear production. There are several versions of this test but the most common are the Schirmer 1 without topical anaesthetic and a variation with anaesthetic. Schirmer with anaesthetic measures the basal aqueous tear production and without anaesthetic additionally measures reflex tearing. The test begins with the application of a filter paper test strip in the inferior temporal conjunctival sac of both eyes. If anaesthetic is applied, the inferior fornix should be blotted to remove residual fluid. The strip is removed after 5 minutes and the length of the tear wetting is measured in millimeters. A reading of 10 mm or greater is generally considered the cut-off for a normal value for both tests. An abnormal finding is highly suggestive of aqueous deficient dry eye. This test suffers from variable repeatability and a wide range of sensitivity and specificity values but the accuracy seems to increase as the severity of the disease increases.2
Practical pearls
• No consensus has been established as to whether the eyes should be open or closed during testing.2
• This test should be performed after conjunctival and corneal stain tests because the testing strip may disrupt these surfaces and alter their accuracy.
• A 1-minute Schirmer test has been proposed to reduce patient discomfort and speed up the test. In this variant, severe dry eye is considered 2 mm and mild to moderate is 3–6 mm.2
• Avoid punctal plugs as a treatment for dry eye in patients with test results above 10. They have a higher risk of excessive tearing as a result.
Phenol red thread test
The phenol red thread test is another, less-common, measure of tear production. This variation is more appealing compared with the Schirmer
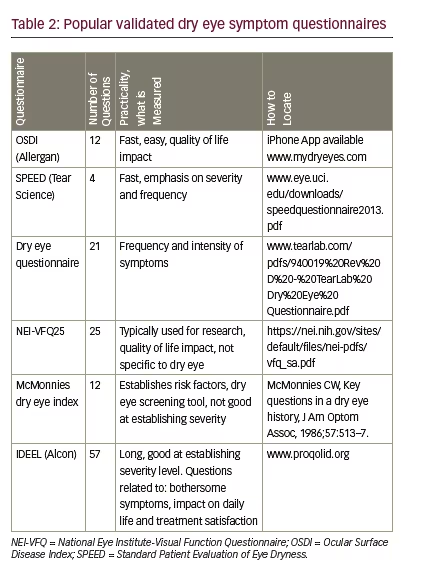
test because it is less irritating and has only a 15-second test duration, but the downside is that it is technically harder to perform. A cotton thread that contains phenol red dye is placed in the same location as the Schirmer test and no anaesthetic is used (see Figure 1). The alkaline pH of the tears causes the dye to turn from yellow to red and then the length of the tear wetting is measured in millimeters. The cut-off value for normal is 10 mm or greater. There is controversy about the accuracy of this test compared with the Schirmer test.2
The International Workshop on Meibomian Gland Dysfunction notes that “if testing suggests the diagnosis of a generic dry eye and tests of tear flow and volume are normal, then evaporative dry eye is implied, and quantification of MGD will indicate the meibomian gland’s contribution”.7
Tear break-up time
Tear break-up time (TBUT) is common, cheap and easy to perform. This test is performed to determine tear film stability, specifically local evaporation from the tear film surface.3 A fluorescein strip is moistened with saline and placed in the inferior cul-de-sac. For best accuracy, this test should occur before any eye drops have been used or the lids manipulated. The patient is observed with a cobalt blue filter and diffuse illumination at the slit lamp. The time (in seconds) between a blink and the appearance of a dark spot in the fluorescein is the TBUT. The patient should be allowed to blink freely prior to the test because forced blinking can cause reflex tearing and affect the accuracy. The test should be repeated three times and the average used to obtain the most reliable result. Ten seconds or greater is considered normal. Abnormal results can occur in evaporative and aqueous deficient forms of dry eye. This test has been criticised because the fluorescein itself can cause tear film instability and
give a falsely low result. The non-invasive TBUT (NI-TBUT) test will be discussed later in this article.
Practical pearls
• The results are dose dependent with higher volume of fluorescein causing a falsely high result. To minimise variability, consider wetting the strip with a single drop of saline and then shake off excess fluid with a quick flick. A micropipette can be used but this is generally not practical clinically.
• If a patient has immediate dark spot(s) with TBUT testing in the same location, consider anterior basement membrane dystrophy as the cause.8
• An additional measure of tear film instability is the Ocular Protection Index that can be calculated as the ratio of TBUT/BI (blink interval, the time between blinks). The lower the value, the more unstable. A value of <1 is pathological and implies that tear break-up is occurs more quickly than the blink.9
Osmolarity
Osmolarity testing measures the concentration of solutes in the tear film where higher levels indicate a reduced aqueous component, either by increased evaporation or reduced aqueous secretion. Unfortunately, this objective test does not distinguish the type of dry eye. Increased osmolarity is a critical component of dry eye disease pathology. The TearLab test (TearLab Inc., San Diego, CA, US) is easy and readily available for use in clinic. It is performed by obtaining 50 nanolitres of tears, applied to an assay, and the results are available immediately in clinic (see Figure 2). The cut-off for normal is 308 mOsm/L or less.10 This test is excellent for severe dry eye disease diagnosis but poor for mild disease. In addition to diagnosis, osmolarity testing can serve as a longitudinal measure to follow the effectiveness of treatment. Normal tears will lack variability with repeated measures,11 but dry eye can be inconsistent, which is likely reflective of the nature of the disease. The test results can also be affected by non-ocular factors such as systemic medications and environmental conditions such as humidity, caffeine intake, etc.12 For these reasons, the US Food and Drug Administration (FDA) recommends two measurements per eye.13 The literature suggests this test may be the more accurate for dry eye diagnosis than Schirmers, TBUT and corneal/ conjunctival staining,14 although there is a cost for osmolarity testing, which is negligible for these other common tests.
Practical pearls
• No drops should be used prior to this test.
• Test both eyes and use the higher number to determine the severity of the dry eye. If there is more than 8 mOsm/L difference between

the eyes, there is a high correlation with dry eye disease even if the number is less than diagnostic cut-off value. The bigger the intereye difference, the more suggestive of dry eye disease.13
Ocular surface dye staining
The use of dyes can help assess the superficial ocular surface to detect damage that can be present in dry eye disease. These dyes include fluorescein, lissamine green and rose bengal. These evaluations are cheap and easy to perform clinically. All of the dyes are applied with the same technique as previously described for the TBUT test.
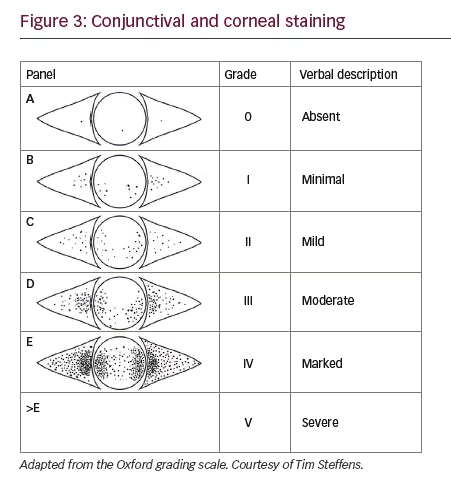
Fluorescein dye will be taken up by corneal and conjunctival tissue where there is disruption in the intercellular junctions. The staining of the cornea is easily seen with use of the cobalt blue filter on the slit lamp whereas the conjunctival staining is best seen with a yellow (blue-free) filter, such as a Wratten filter. The classical staining of the cornea in dry eye includes superficial punctate keratitis (spk) concentrated in the intrapalpebral or inferior area initially. Unfortunately, several other conditions can cause spk therefore this test is not very specific2 and approximately only 10% of dry eye patients have spk therefore it is also not very sensitive.5
Lissamine green and rose bengal evaluate the conjunctival surface by staining areas not properly covered by mucin. Rose bengal requires the use of anaesthetic when applied due to ocular irritation and epithelial toxicity. Lissamine green does not have this limitation and can additionally be helpful as it stains lid abnormalities such as Marx’s line and lid wiper epitheliopathy.
There are three common grading systems that are used largely in clinical trials, including Collaborative Longitudinal Evaluation of Keratoconus,15 Oxford16 (see Figure 3) and van Bijsterveld.17 These grading systems can also be used clinically to aid in consistency.
Practical pearls
• All of the dyes are dose dependent.
• The rose bengal dye staining is best observed with a red-free filter.
• The fluorescein staining evaluation should be carried immediately after TBUT because the dye absorbs into the tissue and can make staining blur out.4
Corneal sensitivity
Dry eye patients potentially have decreased corneal and conjunctival sensitivity due to the deficiency/instability of the tear film that may make the superficial nerve endings susceptible to damage. Corneal sensitivity testing alone is not a good diagnostic test for dry eye but it may be an adjunctive test that can help grade the severity and monitor for therapeutic improvements. There are presently three ways to perform this test: cotton wisp, Cochet-Bonnet esthesiometer and the air jet esthesiometer esthesiometer. All three tests must be carried out before any anaesthetic is placed in the eye. The cotton wisp test is an easy clinic test and is performed by pulling the cotton into a wisp on a cotton swab, touching the cornea from the side while the patient looks away and subjectively evaluating the blink reaction. The Cochet-Bonnet esthesiometer uses a fine nylon filament for evaluation but disrupts the corneal epithelium, thus, making further dry eye testing less accurate. The CRCERT-Belmonte esthesiometer uses a non-contact air jet and applies mechanical, chemical and thermal stimuli. A study by Bourcier et al. found that dry eye patients showed increased stimulus thresholds compared with controls using the CRCERT-Belmonte.18
Practical pearls
• The cotton wisp test is easy and the cheapest method of corneal sensitivity. The CRCERT-Belmonte may be ideal due to its non-invasive and objective methodology but may not be clinically practical.
• Consider this test when the signs and symptoms do not correlate.
Lid evaluation
Two important aspects of the lids can be evaluated at the slit lamp, including the lid anatomy/positioning and the meibomian glands. The lid positioning evaluation includes screening for conditions such as lagophthalmos (the inability to fully close the eyelids), entropion,

ectropion, blink evaluation and tear duct positioning and patency. All of these can have an effect on the tear dynamics and corneal wetting. The meibomian glands secrete a lipid called meibum that comprises the outer layer of the tear film. Dysfunction of these glands is the leading cause of evaporative dry eye, the most common type of dry eye. This dysfunction is most commonly caused by inflammation that can affect both their morphology and expression quality and quantity, all of which can be grossly examined at the slit lamp. Expression can be performed with the fingers, a cotton swab and custom-made devices. In 2011, the International Workshop on Meibomian Gland Dysfunction published a grading scale from 0 to 3 (see Table 3). Meibomian gland evaluation is easy to perform but has poor repeatability and is variable between practitioners.19 Other important lid margin characteristics to note are the presence of collarettes, lid notching and ridging, keratinisation, telangiectasias and chalazia.
Practical pearl
• Meiboscopy can be easily performed in clinic by applying a transilluminator to the cutaneous side of the inverted eyelid while observing the silhouette of the gland. This can give an indication of meibomian gland atrophy/dropout, which is present in severe disease and dysfunction,3 and is associated with dry eye symptoms.20
Sjögren’s
Sjögren’s syndrome (SS) is an autoimmune disease with classical symptoms of dry eye and a dry mouth due to damage to the lacrimal and salivary glands. This condition causes aqueous deficient dry eye due to lacrimal secretion impairment, therefore, tear volume tests should be abnormally low. When this finding is present, eye care practitioners should ask about dry eye mouth symptoms and co-manage with a rheumatologist, if indicated. In those that have signs and symptoms suggestive of Sjögren’s, the proposed classification criteria of Sjögren’s diagnosis were outlined in 2012 by the American College of Rheumatology. They must meet two of the following three features:21
• Positive serum testing (anti-SS-A/Ro and/or anti-SS-B/La or rheumatoid factor and anti-nuclear antibody >1:320).
• Salivary gland biopsy meeting specific diagnostic characteristics.
• Keratoconjunctivitis sicca with ocular staining ≥3. One downside of the serum biomarkers is that they are often not positive in SS, especially in the early stages of the disease. About half of patients with SS are negative for the SS-A/Ro and SS-B/La testing. Another downside is that these biomarkers are not specific to SS and can be positive in other conditions, such as lupus.21
Emerging technologies
Functional visual acuity
Dry eye often negatively impacts visual acuity because of the tear film instability. Patient will note intermittent blur during daily tasks depending on the environmental conditions, time of day, visual demands and/or blink. Functional visual acuity (FVA) was developed to more accurately assess everyday vision. This measure specifically quantifies the decay in vision between blinks.3 A device was developed (FVAM, SSC-350®, NIDEK, Gamagori, Japan) to measure FVA in patients while they refrain from blinking for 30 seconds after the instillation of topical anaesthetic. FVA is significantly worse in those with dry eye compared with controls and can improve with treatment.2
Practical pearl
• Blink rates have been shown to decrease during computer use and can result in a worsening of FVA and other dry eye symptoms during this activity.22
Tear volume
The tear meniscus occurs at the intersection of the eyelid (typically evaluated inferior) and the bulbar conjunctiva. The most commonly measured parameter is the tear meniscus height (TMH). Abnormal values would be expected in aqueous deficient dry eye syndrome. Traditionally this measure was performed with a slit lamp, photography, video recording, a Tearscope or reflective meniscometry. More recently, the evaluation is carried out with anterior segment optical coherence tomography (OCT). With this device, if the height was less than 0.3 mm, the sensitivity was 67% and specificity was 81% for the diagnosis of dry eye according to a study conducted in Japan.23 No consensus has been made for the height cut-off level. Advantages of OCT to assess tear volume include its non-invasive nature, objective and quantitative measurements and additional measurements of meniscus curvature and cross-sectional area. The downside is that the OCT is an expensive instrument and measurements can be time consuming.
Practical pearls
• Serial measurements of TMH should be considered due to variations depending on time of day.19
• TMH should be performed before any drops are applied.
Tear clearance Reduced tear clearance, or tear turnover, can cause inflammation of the ocular surface and is correlated with dry eye symptoms.3 Tear Function Index (TFI) and fluorescein clearance test (FCT) measure tear clearance and are performed by instilling fluorescein and a testing strip in the lower cul-de-sac. Serial measurements are taken and the amount of residual dye present on a strip is compared with a standard colour scale. The FCT is considered normal if all of the dye is gone after 20 minutes.19 Fluorophotometry is the gold standard for estimating tear turnover and volume; however, it is expensive, requires a lab and special expertise, therefore, it is not clinically practical.3
Tear film stability
Non-invasive methods of evaluating the TBUT have an advantage over the traditional TBUT because the tear film is undisturbed by fluorescein. The methods include the xeroscope, tear film stability analysis system

(TSAS), wavefront aberrometer, TearScope (Keeler, Windsor, UK), Oculus Keratograph 5M (Oculus, Arlington, WA, US)24 and LipiView II (TearScience, Morrisville, NC, US). All of these devices, except the aberrometer, use images of rings captured as they are reflected off the precorneal tear film.11 The time between the blink and the first sign of ring distortion is the NI-TBUT. The Oculus and LipiView are superior for clinical use because they give objective data and have other dry eye evaluation features such as meibomian gland evaluation (which will be discussed later in this article). A study by Hong et al. found that the NI-TBUT measured with the Oculus Keratograph had good correlation with traditional TBUT, Schirmer 1 and meniscus height. In addition, for the diagnosis of dry eye, this device had a sensitivity of 84.1% and specificity of 75.6% when the NI- TBUT was set at 2.65 seconds.24 The LipiView can also measure the lipid layer thickness and when this number is low, it is correlated with dry eye symptoms.19
Practical pearls
• Tear film analysis can be altered by eye rubbing, oil-based ointment, artificial tears, make-up and swimming in a chlorinated pool.25
Tear film components/composition
Specific biomarkers for dry eye may be present in the tear film to aid in diagnosis. First, matrix metalloproteinases (MMPs) are enzymes that are found in high levels with ocular inflammation, which is a large contributor to the cause of dry eye disease. MMP-9, specifically, can be measured with InflammaDry (Rapid Pathogen Screening Inc., Sarasota, FL, US). A tear sample is retrieved from the lower cul-de-sac and results are given in 10 minutes (see Figure 4). For dry eye diagnosis, it has an 85% sensitivity and a 94% specificity.26 It does not distinguish the type of dry eye. This test is approved for use in Europe, Canada and the US and is easy to use.

Practical pearl
• Avoid punctal plugs in patients with high levels of ocular inflammation; consider first treating with topical anti-inflammatory agents.
• No drops prior to performing test.
• False positives can result from any cause of inflammation including infection, blepharitis and ocular allergies.
Lactoferrin is a biomarker for allergy that is produced by the acinar cells of the lacrimal glands and would be abnormally high in those with aqueous deficient dry eye. The TearScan System, which measures lactoferrin, as well as immunoglobulin E (an allergy marker), can be performed in the clinic and has a sensitivity of 83% and specificity of 98%.11
Lysozyme is another protein that is reduced in dry eye disease but cannot be easily tested clinically. There are approximately 1,543 proteins in the normal tears but their relationship to dry eye, if present, is largely unknown.11 The clinically practical biomarkers to test at the current time are MMP-9 and lactoferrin, but this may change in the future.
Corneal evaluation Confocal microscopy is a non-traditional method of examining the corneal cells in vivo. Studies have shown dry eye disease is correlated with changes in the epithelial and stromal layers, as well as the corneal nerves.27 This method is not commonly performed clinically to aid in diagnosis due to the availability of the machine.
Another potential diagnostic corneal test includes infrared thermography. This test when applied to the eye offers an objective, non-invasive, quick measure of the temperature of the cornea. Compared with normal eyes, this temperature has been shown to be higher in dry eye patients. In addition, after sustained opening for 10 seconds, dry eye patients’ central corneal temperature becomes significantly lower where controls remain the same. Therefore, thermography can be used as a diagnostic tool. The Ocular Surface Thermographer (TOMEY Corporation, Nagoya, Japan) has recently been developed and takes measurements in a similar fashion to an autorefractor. A study by Kamao et al. showed that the decrease in the temperature after sustained opening was significantly related to TBUT (p<0.001) and the sensitivity and specificity for dry eye diagnosis were 0.83 and 0.80, respectively.28 There is limited research in the literature on this diagnostic method and the expense may be inhibitory.19 This testing is sensitive to the room temperature and humidity levels.
An additional corneal indicator of dry eye is measurement of epithelial thickness using anterior segment OCT. This layer is thicker in dry eye presumably due to desquamation. A study by Kanellopoulos et al. found a statistical significantly epithelial thickness difference of 6.5 μm between women with dry eye compared with controls.29
Practical pearls
• Kanellopoulos et al. recommend this test for early stage disease or subclinical disease and not for severe dry eye.29
• Confocal microscopy measures show dry eye patients with decreased corneal nerve density compared with controls.27
Conjunctival evaluation The conjunctiva can be evaluated by surface biopsy, confocal microscopy and impression cytology to detect signs due to dry eye disease. None of these methods may be clinically practical.
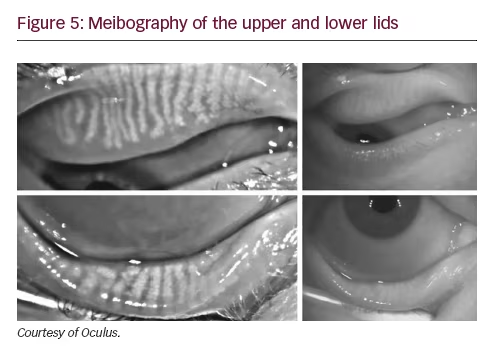
Lid evaluation
Meibography, the imaging of the meibomian glands, is now possible with the Oculus Keratograph 5M (Oculus, Arlington, WA) (see Figure 5) and LipiView II (TearScience). These technologies use infrared light to quantify the extent of meibomian gland dropout. These tools could help with diagnosis of dry eye, aid in treatment strategies and monitor for changes over time. A recent study by Finis et al. showed that meibomian gland atrophy and TBUT were significantly inversely correlated.30
Practical pearl
• Both of these devices are expensive but they also include features such as topography, tear meniscus and tear film evaluation.
Sjögren’s The Sjo® test (Bausch & Lomb, Rochester, NY, US) is a commercially available panel, which includes the traditional serum biomarkers for SS and additionally includes other early biomarkers that have shown better sensitivity and specificity than traditional testing (previously mentioned in this article). The sensitivity from a collective group of studies is around 90% and specificity of approximately 80%. This new test may allow for earlier diagnosis in those patients with suggestive SS who have tested negative on traditional testing. This allows for earlier treatment interventions and possible better long-term outcomes.21 Early diagnosis is important for the eye but for other possible systemic conditions associated with SS that include: lymphoma, arthritis, thyroid dysfunction, fibromyalgia, interstitial nephritis, vasculitis/purpura, fatigue, peripheral neuropathy, pulmonary issues and dry skin and mouth.31
Practical pearl
• Beckman et al.21 recommend all patients with aqueous deficient dry eye be considered for serological testing given the advances in testing and potential harm in a missed diagnosis.
Conclusion Despite the challenges of dry eye diagnosis, numerous tests, some traditional and some emerging, are available and can produce useful data. There is a logical sequence of tests for patients suspected of dry eye disease (see Table 4). Combining tests results, including signs and symptoms, will yield in the most accurate diagnosis. This article focused on the application of clinically useful tests, the benefits and limitations, as well as practical pearls to implement use.







