Age-related macular degeneration (AMD) is one of the main causes of irreversible vision loss in ageing populations worldwide.1 In 2019, the US recorded 1.49 million people aged 40 years or older with late-stage AMD, reflecting a crude prevalence rate of 0.94%.2 Geographic atrophy (GA), the late stage of non-neovascular AMD, is characterized by gradual and progressive atrophy of photoreceptors, while most of the inner retinal cells remain intact.3 The central atrophic area expands over time, leading to increased vision loss and a reduced ability to read and recognize faces, severely impacting patients’ quality of life.4 The PRIMA bionic vision system (Pixium Vision SA, Paris, France) is a photovoltaic wireless subretinal implant designed to improve central vision in patients with atrophic AMD.5 Currently, PRIMA is the only experimental therapy capable of restoring vision in patients with degenerated photoreceptors, such as those affected by atrophic AMD. This editorial provides an overview of (1) current treatment strategies for AMD, (2) PRIMA’s mode of action, clinical trial data and future steps, and (3) its potential impact on clinical practice.
Treatment strategies for AMD should aim at restoring functional central vision without jeopardizing the surrounding retina while also allowing for their simultaneous use.4 In this context, electronic retinal prostheses enable the reintroduction of visual information into the degenerate retina by electrical stimulation of the remaining neurons. Electrode arrays can be placed either epiretinally to target the retinal ganglion cells or subretinally to stimulate bipolar cells, which comprise the first layer of neurons above the photoreceptors.5 Argus® II Retinal Prosthesis System (Second Sight Inc, Sylmar, CA, USA), an epiretinal implant approved for patients blinded by retinitis pigmentosa (RP), contains 60 electrodes positioned relatively distant from the target retinal ganglion cells.6,7 The subretinal implant Alpha IMS and Alpha AMS (Retina Implant AG, Reutlingen, Germany) contain up to 1,600 pixels and has been tested in patients with RP.8,9 Visual acuity measured using Landolt C-rings was measurable in 4 out of 29 patients with an Alpha IMS, ranging from 20/2000 to 20/546 and in 2 out of 15 patients with an Alpha AMS, ranging from 20/1111 to 20/546.8,9
The PRIMA bionic vision system is a wireless subretinal prosthesis in which photovoltaic pixels directly convert projected light patterns into local electric current. It aims to partially restore central vision in subjects affected by GA due to AMD. In addition to the subretinal implant, it includes a pair of glasses equipped with a camera and a digital projector (Figure 1) and a pocket computer for image processing.4 Images captured by the camera and simplified by the pocket processor are projected from the glasses onto an 18-degree-wide central field on the retina using pulsed near-infrared light (880 nm). The subretinal implant substitutes the photoreceptors lost in GA with photovoltaic pixels, which convert the pulsed near-infrared light into electric current to excite the second-order neurons (bipolar cells) in the retina, providing formed visual perception. The first generation of the PRIMA implant is 2 mm in width and 30 μm in thickness and contains 378 hexagonal pixels, each with a size of 100 μm.4
Figure 1: The PRIMA bionic vision system for providing central vision to subjects with geographic atrophy. (a) PRIMA glasses with the projection module including the camera; (b) artistic rendering of the augmented reality glasses capturing a video of a flower and projecting it onto the retina using near-infrared laser.
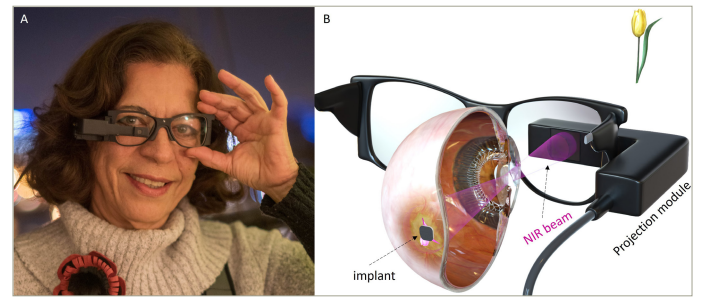
Reproduced and adapted (cropped) with permission from Palanker et al.4 under CC BY-NC 4.0 DEED (http://creativecommons.org/licenses/by-nc/4.0).
A two-phase study evaluating the PRIMA bionic vision system was conducted on five subjects with a GA exceeding three optic disc diameters (ClinicalTrials.gov identifier: NCT03333954). Implantation lasted approximately 2 h, and the subjects’ natural visual acuity in the operated eye did not decrease after the surgery.5 The first phase started after surgery and lasted up to 18 months. Initially, subjects used virtual reality glasses (PRIMA-1), blocking the natural residual vision. The primary endpoint, the perceptual threshold elicited by the implant, was measured in all five subjects.5 In three subjects with the implant centrally placed in the subretinal space, prosthetic visual acuity closely matched the pixel size, ranging from 20/460 to 20/550 (between 1.1 and 1.3 pixels).5 The second phase lasted from month 18 to month 36. Subjects used augmented reality glasses (PRIMA-2; Figure 1), allowing the simultaneous use of prosthetic vision and residual natural vision by the fellow eye and the operated eye.4 Using electronic magnification, the subjects’ prosthetic acuity was significantly higher than the residual natural acuity: they were able to recognize optotypes with acuity between 20/98 and 20/63 and to read words that they could not normally read without the prosthesis.4 The highest improvement in visual acuity was of logMAR 0.78 (20/71 with PRIMA-2 and electronic magnification versus 20/428 without the system) reported at 18–24 months. This feasibility study demonstrated promising results, showcasing the safety, stability and efficacy of the PRIMA subretinal implant over 36 months.4
Compared with previous trials using subretinal implants,9,10 PRIMA has shown better and more consistent performance, which might be partially attributable to patient selection. In patients with AMD, the inner retina in the central scotoma may be better preserved than at the end stage of RP, which is commonly associated with a nearly complete loss of photoreceptors in the entire retina and associated retinal rewiring.5 Patients using the Argus II system have been reported to have lower visual acuity, measuring at 20/1260 or worse.11 This implant not only activates proximal neurons but may also stimulate the axons of remote retinal ganglion cells passing underneath the electrodes, causing distorted visual percepts.12 With PRIMA, prosthetic visual acuity was found to closely match the pixel size, with an average value of 20/500 and 1.2 pixels, respectively), and the use of electronic magnification enabled optotype recognition, achieving an equivalent acuity superior to 20/100.4 Furthermore, the epiretinal Argus II and the subretinal Alpha IMS/AMS implants are powered via a transscleral cable, which requires complex surgical procedures and has been associated with postoperative complications.8,11 In patients with AMD, such subretinal cable may affect the residual photoreceptors and associated peripheral vision. The wireless PRIMA implant simplifies the surgical procedures, which can be performed within only 2 h instead of 4 h with the Argus II system9,11 and 6–8 h with the Alpha IMS/AMS implants.8
Recent studies on pharmacological,13 gene14 and stem cell15 therapies in AMD have focused on slowing or preventing cell loss in retinal degeneration, and there is no established therapy to restore vision in atrophic AMD to date. Compared with other retinal prostheses,16–18 PRIMA provides better performance and reading capability. Currently, PRIMA is the only therapy to restore vision in a degenerated area of the retina, and it has the potential to become a standard therapy for advanced atrophic AMD. Recently, the US Food and Drug Administration has granted the Breakthrough Device Designation to the PRIMA system.19 This designation will help Pixium Vision to expedite its development and work closely with the US Food and Drug Administration on refining its regulatory submission in the US. If the system is approved, this designation will also allow better accessibility to certain outpatient and inpatient reimbursement pathways. The PRIMA system is currently under clinical testing in the multicentre PRIMAvera study to demonstrate its performance and confirm its safety in 38 patients with atrophic AMD (ClinicaTrials.gov identifier: NCT04676854).20 Furthermore, second-generation PRIMA implants, which are now being optimized for future clinical trials, may enhance prosthetic visual acuity up to 20/100 and, with electronic magnification, potentially up to 20/20.21 Such prosthesis would provide very significant improvement of central vision for a large number of patients blinded by atrophic AMD.







