Age-related macular degeneration (AMD) is a chronic, progressive degenerative disease with subsequent reduction in visual acuity. The disease is the foremost cause of visual impairment in the elderly.1,2 The most recent data from Europe indicate that the prevalence of AMD may be decreasing, despite the increase in life expectancy and projected increases, which may be in part due to adoption of healthier lifestyles.3 Moreover, recent studies indicate that AMD-associated blindness and visual impairment appears to be decreasing, which is probably due to improvements in early diagnosis and the widespread use of anti-vascular endothelial growth factor (VEGF) therapies.4,5
AMD is normally divided into early-stage disease, characterised by mild to moderate loss of visual acuity, and late-stage disease, which is associated by severe loss of vision. Late-stage AMD is further subdivided into a dry form, consisting of central geographic atrophy, and a wet form in which choroidal neovascularisation is present. Current therapeutic options for wet AMD include laser therapy and anti-VEGF agents, while treatment options for dry AMD are notably lacking. As such, preventive and supportive strategies to slow the progression of the disease are of primary importance. In this regard, smoking is undoubtedly the most important modifiable risk factor, even though lifestyle, diet and nutritional aspects are believed to play key roles in the disease.6 In the broadest terms, even though there is lack of undisputable evidence for their use in the prevention of AMD in the general population,7 oral vitamin and mineral supplementation may however help to slow disease progression in selected subgroups of patients.8 Other compounds, such omega-3 fatty acids, may also have additional protective effects.9 Herein, the current data on nutritional supplementation are overviewed with focus on the utility of micronutrients in preventing progression of AMD. This will allow for a more in-depth discussion of the role that dietary supplements can play in managing the pathology in daily practice.
Pathogenesis of age-related macular degeneration
The exact pathogenesis of AMD is not completely understood, despite progress in knowledge of the disease and its underlying mechanisms. Current hypotheses regarding its pathogenesis involve an interplay of complex multifactorial interactions of several factors, including metabolic, genetic and environmental factors.6 As for many other diseases, oxidation, inflammation and angiogenesis in the retinal pigment epithelium (RPE) are held to have crucial roles in the pathogenesis of AMD.10 Impaired function of the RPE is involved in the majority of theories about the pathogenesis of AMD. The RPE is composed of a layer of highly specialised cells that have a number of functions aimed to support and sustain the survival of photoreceptor cells as well as to allow the exchange of nourished and metabolic wastes.11 Indeed, the photoreceptor layer has high metabolic activity, thus requiring a large amount of nutritional needs and biological clearance of photoreceptor debris, functions that are aided by the RPE.11 Therefore, even if a specific insult leading to damage of the RPE has not been found, it is clear that it can be affected by many factors such as the levels of certain nutrients and immune damage.12 It is for this reason that damage to the RPE may be worsened by extrinsic factors such as smoking.11

Complement is an aspect of the innate immune system that is also clearly involved in the pathology of AMD. In this regard, defects in complement system genes have been shown to predispose individuals to AMD, and many common (e.g., CFH, C2/CFB, C3, and CFI) and less common (e.g., C2/CFB, C3, C9, CFH, CFI, and VTN) genetic variants have been modified.13 It is thought that complement produces autoimmune-mediated retinal damage, which can help to establish a sustained inflammatory response that leads to retinal damage.14
Considering the pathophysiology of AMD, the early changes of AMD often start with drusen formation in the macular area; drusen are tiny yellowish accumulations of extracellular material, lipids, amyloid, complement factors and cellular debris that build up between Bruch’s membrane and the RPE.15–17 These deposits may be related to failure of the RPE to process cellular debris associated with outer segment turnover. AMD is believed to progress along a continuum, starting from drusen formation up to the atrophic changes typical of dry AMD, the choroidal neovascularisation characterising wet AMD, being considered a step toward a more global atrophic degeneration, especially after anti-VEGF treatment. The manifestation of drusen is associated with several processes which include thickening of the collagenous layers of Bruch’s membrane, deterioration of elastin and collagen within Bruch’s membrane, augmented advanced glycation end products, and accumulation of both lipids and exogenous proteins.17 Such alterations may act as a physical barrier to passage of fluid and nutrients between the choroid and outer retina resulting in relative ischemia, leading to further deterioration. Early AMD is usually asymptomatic and is clinically defined by accumulation of medium-sized drusen (63–125 µm), and accompanied by pigmentary abnormalities (Table 1).18
In roughly 10–15% of patients with dry AMD, the deterioration will become more extensive and accompanied by significant vision loss due to geographic atrophy, progressing to the wet form.17 As AMD progresses, there is a gradually increased loss of RPE and photoreceptor cells. Progression to wet AMD implies that fluid, exudates and/or blood are present between the neural retina and the RPE and/or, as in the case of detachment of the RPE, between the RPE and Bruch’s membrane. Patients will experience gradual loss of visual function over a time span of many years. Larger drusen (>125 µm) are present in intermediate and advanced forms of AMD (Table 1). If the condition is not treated, AMD advances further to a scarring stage over the course of over several months and years.
Retinal pigment epithelium and oxidative stress
Without a doubt, oxidative stress is a substantial contributor to macular degeneration, and is related to cellular damage caused by reactive oxygen species (ROS). ROS include a range of compounds that comprise free radicals and many by-products of oxygen metabolism. During aging, the production of free radicals increases, an event that is associated with progressive damage to cellular structures.19 The RPE, as with other structures, are susceptible to damage by oxidative stress, and is highly influenced by an inadequate dietary intake of antioxidants.20–22 Damage to the RPE leads to accumulation of proinflammatory substances in addition to molecules that generate additional ROS and, consequently, additional oxidative stress, further damaging the RPE and establishing a sort of vicious circle.23 Drusen, in fact, contain several of these products, such as lipofuscin, which is accumulated in RPE.8 Lipofuscin, a mixture rich in lipid-protein complexes, contains by-products of vitamin A metabolism and lipid peroxidation.8
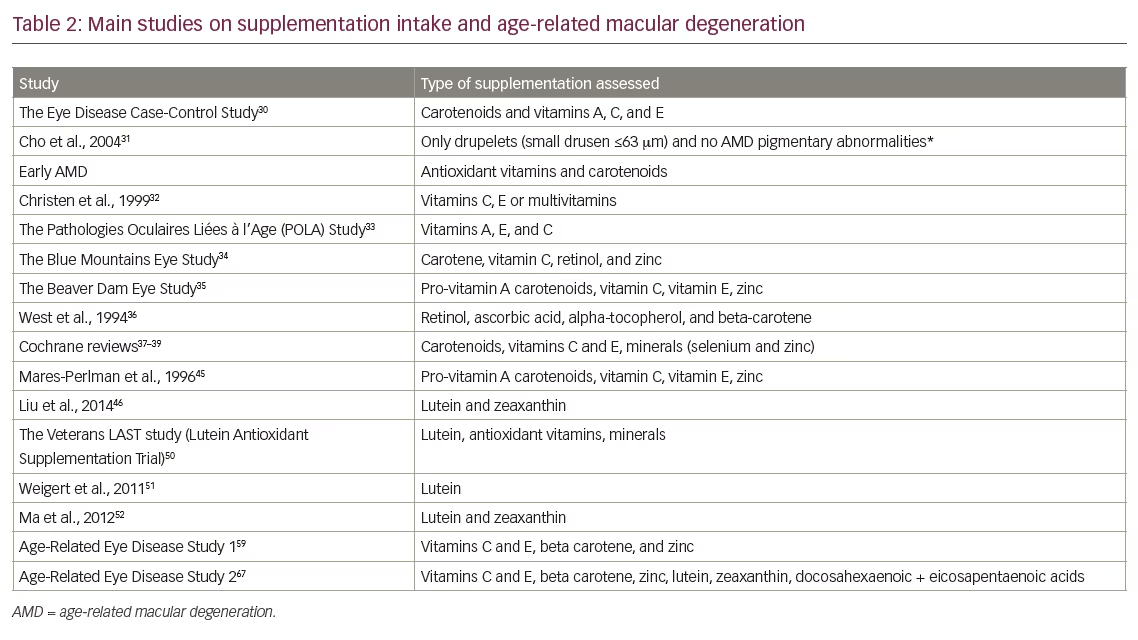
Of interest, the existence of endogenous defence mechanisms can help to counteract the negative effects of ROS. The endogenous antioxidant network includes several vitamins (C and E), carotenoids (carotenes, lycopene, lutein and zeaxanthin) and several other compounds with antioxidant activity such as flavonoids, uric acid, and coenzyme Q10.24,25 Such antioxidant molecules can have protective effects against damage caused by free radicals.19 In the eye and retina, vitamins C and E along with lutein and zeaxanthin appear to be the most relevant.26 This highlights the importance of adequate dietary intake of these micronutrients. While in most settings, normal dietary intake is adequate, this however varies among countries and individual dietary consumption. Moreover, lutein and zeaxanthin are not synthesised by humans and are found in some fruits and vegetables like broccoli, lettuce and spinach.27 This then raises the possibility that dietary supplementations may be of benefit in preventing or slowing progression of AMD. Indeed, several types of compounds have been studied in terms of a potential role in protection of AMD: antioxidant vitamins (mainly C and E), carotenoids and omega-3 fatty acids. The most relevant results on these factors are summarized below, whereas the main studies assessing supplementation and/or dietary intake of vitamins and antioxidants are listed in Table 2.
Micronutrients and age-related macular degeneration
Vitamins C and E
As humans do not have the ability to synthesise vitamin C, it can be considered as an essential nutrient;28 it is perhaps the most important soluble antioxidant given its effects on the reduction of ROS. Vitamin C has long been held to be protective in age-related retinal disease, even if epidemiological data on the relationship between dietary intake of vitamin C and the risk of AMD are not entirely consistent.29 Earlier studies, such as The Eye Disease Control Study, reported that a lower risk for AMD was suggested among subjects with higher intake of vitamin C.30 However, these results were not entirely confirmed in later studies where no unambiguous association was found between intake of vitamin C and risk of developing AMD.30–36 Three Cochrane analyses have failed to find clear clinical evidence for vitamin C in prevention of AMD when administered alone.37–39 Notwithstanding its role as an important antioxidant, additional studies are needed to fully understand the potential role of vitamin C in AMD. In fact, a potential role for vitamin C in AMD may be difficult to demonstrate when administered alone, and it is entirely possible that this antioxidant may also act in concert with other antioxidants.
Vitamin E is a fat-soluble vitamin and an effective scavenger of free radicals; vitamin E supplements usually contain α-tocopherol or a mixture of other forms of vitamin E. In contrast with vitamin C, the evidence for the benefits of vitamin E supplementation in AMD is based on several lines of well-established evidence. This is due firstly to the fact that photoreceptors and the RPE likely have the highest concentrations of vitamin E among all tissues.40 Moreover, retinal levels of vitamin E levels in the retina appear to be dictated by the level of oxidative stress.41 Dietary insufficiency of vitamin E is also associated with accumulation of lipofuscin accumulation in the RPE42 and is further accompanied by loss of photoreceptors.43 It is also well-known that patients with disorders that alter absorption of fat-soluble vitamins have retinal degradations including lipofuscin deposits, the progression of which can be circumvented by exogenous vitamin E supplementation.44
The role of vitamin E in AMD is further supported by a wealth of epidemiological data,33,36,45 including the landmark Beaver Dam Eye Study which documented a higher risk of developing large drusen in subjects with low vitamin E intake.35 The Pathologies Oculaires Liées à l’Age (POLA) Study on AMD and antioxidant status reported that the risk of late AMD was reduced by 82% in the highest quintile of plasma tocopherol levels compared with the lowest; lipid-standardised plasma tocopherol levels were inversely associated with early signs of AMD (odds ratio 0.72; p=0.04).33 There are, however, some data that do not fully support its role in retinal disease. For example, the Eye Disease Case-Control Study Group found no significant interaction between the risk of AMD and vitamin E.30 However, such differences are likely to lie within the different types of vitamin E sources studied, which probably have different biological antioxidant activities in vivo. While there are limited data on the use of vitamin E in limiting progression of AMD, the most recent Cochrane analysis reported that vitamin E supplements alone will not prevent or delay the onset of AMD.39
Carotenoids (lutein and zeaxanthin)
There is strong reason to believe that the carotenoids may have a significant biological role in prevention and slow the progression of retinal disease. By scavenging ROS, the carotenoids lutein and zeaxanthin can protect the macular region from photooxidative injury.27,46 Lutein and zeaxanthin supplements may also protect against the increase in lipofuscin granules.46 In addition, the status of the macular pigment has also been shown to improve following a healthier diet.27,47 Several studies have indicated that, indeed, there is an association between reduced dietary intake of lutein/zeaxanthin and the development of AMD.48,49 Data from the The Age-Related Eye Disease Study (AREDS)2 study (discussed below) appear to confirm that such a relation is possible, even if the exact degree to which and at which stage the greatest benefits are seen is not clear at present.29 Prior to the AREDS study results, several interventional studies provided data for a beneficial effect of lutein and zeaxanthin in retinal disease. Most investigations have indicated that macular pigment optical density can be increased with lutein supplementation.50,51 In particular, a recent study by Ma et al. reported that supplementation with lutein and zeaxanthin in patients with early AMD improved macular pigment optical density as well as visual function.52 In addition, the PIMAVOSA trial reported that there was significant correlation between macular pigment optical density and plasma levels of both lutein and zeaxanthin.53 Lastly, in a meta-analysis by Ma et al., high dietary intake of lutein and zeaxanthin was found to be protective against late AMD, although there were no apparent effects on early AMD.54
Some of the apparently conflicting results in the literature may be explained if such supplements are effective only when dietary intake is below a sufficient threshold.55 A Cochrane review from 2017 concluded that there is ‘no evidence’ for supplements such as lutein and zeaxanthin.39 As such, larger trials are warranted to further define its precise role in AMD and identify those individuals who will benefit most from supplementation with lutein and zeaxanthin.
Zinc
Zinc ions have been implicated in the regeneration of rhodopsin and in the phototransduction cascade in photoreceptors;56 zinc is also a cofactor for several ocular enzymes involved in oxidative processes such as superoxide dismutase and catalase.57 Only a very limited number of trials have attempted to study the potential role of orally supplemented zinc on AMD in the absence of other supplements. In 2013, a systematic review of trials with zinc was published, which included 10 studies, with the main conclusion that the effect of zinc intake was inconclusive in prevention of AMD.58 However, the AREDS1 trial demonstrated that 80 mg zinc oxide, alone or combined with antioxidants, showed a significant reduction in the risk of progression to advanced AMD in patients with moderate AMD, even if the difference in loss of visual acuity was not statistically significant.59 Another randomised trial reported a significant increase in visual acuity in early AMD patients, although a second study documented no significant effects of zinc on visual acuity in advanced AMD.58 Based primarily on the above studies, it is current opinion that zinc treatment may be effective in preventing progression to advanced AMD, even if it is likely that zinc supplementation by itself is may not be adequate to produce clinically meaningful differences in visual acuity.58
Omega-3 fatty acids
Both eicosapentoic acid (EPA) and docosahexaenoic acid (DHA) are considered essential dietary compounds. The omega-3 fatty acids EPA and DHA are clearly implicated in AMD, and the clinical data establishing a link between insufficient intake of omega-3 fatty acids and AMD are relatively strong. The Blue Mountains Eye Study reported a relation between higher frequency of fish intake and a decreased lower risk of late AMD.60 Individuals with the highest intake of omega-3 fatty acids showed a reduced risk of early AMD versus those with the lowest intake (odds ratio 0.41). In particular, consumption of fish at least once a week was associated with 40% reduction of early age-related maculopathy, and consumption at least three times per week was furthermore associated with a reduced incidence of late age-related maculopathy (odds ratio 0.25).61 In a 10-year extension of the same study, fish consumption once a week, total omega-3 fatty acid intake, and consumption of 1–2 servings of nuts per week were all associated a reduced incidence of early AMD.62
The Nurses’ Health Study and the Health Professionals Follow-up Study similarly showed that there was an inverted correlation between intake of DHA and incidence of AMD.63 This was a large single-cohort study that investigated fat intake and development of AMD that followed 72,489 patients for 10–12 years; in patients with AMD, an inverse association was found between intake of long-chain DHA and AMD (odds ratio 0.70; p=0.05). In addition, consumption of more than four servings of fish per week was associated with a reduction in risk of AMD by 35% versus fewer than three servings per month (odds ratio 0.65; p=0.009).
Other studies have also found a relation between consumption of omega-3 fatty acid and a reduced risk of neovascular AMD. The European Eye Study (EUREYE) was a cross-sectional population-based study that investigated dietary intake of DHA and EPA in 105 participants with AMD and 2,170 controls. This study found that consumption of DHA or EPA led to a reduced risk of neovascular AMD (odds ratio 0.32; p=0.03 and odds ratio 0.29, p=0.02, respectively). In addition, consumption of oily fish at least once per week reduced the risk of neovascular AMD by about one-half versus consumption less than once per week (odds ratio 0.47, p=0.002).64
In AREDS2, the addition of DHA + EPA to the prior AREDS formulation did not further reduce the calculated risk of AMD, an unexpected finding considering previous epidemiological studies.65 However, differences among trials may be due to a number of factors, including differences in formulations used or variations in consumption of fish.65 Considering the available evidence for omega-3 fatty acids, it is nonetheless reasonable to promote the consumption of dietary fatty fish in patients with AMD.
Nutritional supplements in therapeutic approach to dry age-related macular degeneration
The Age-Related Eye Disease Study
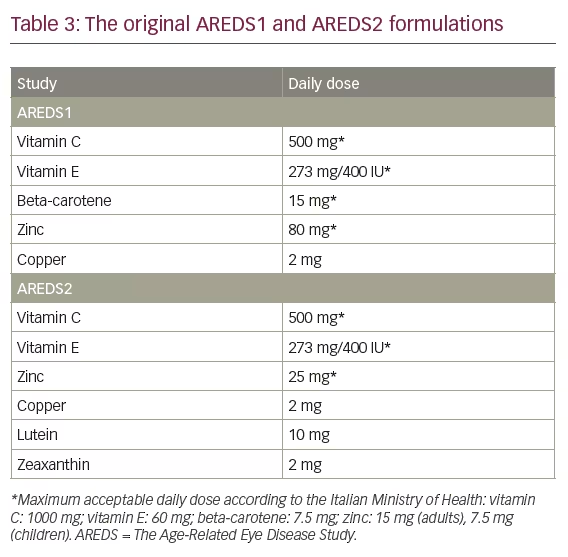
AREDS was a landmark clinical trial sponsored by the US National Eye Institute to evaluate the effect of high doses of vitamin C, vitamin E, beta-carotene, and zinc on the progression of AMD and cataract. From 1992–1998, AREDS1 enrolled 3,640 patients aged 55–80 years with a diagnosis of AMD. The supplementation given in AREDS1 contained vitamin C (500 mg), vitamin E (273 mg/473 IU), beta-carotene (15 mg), zinc (80 mg), and copper (2 mg) (Table 3).59 AREDS1 showed that there was a significant benefit of antioxidants in combination with zinc, which was associated with a reduced risk of progression to AMD by 28% (odds ratio 0.72; 99% confidence interval = 0.52–0.98) after a mean follow-up of 6.3 years in individuals >55 years of age. Shortly thereafter, the AREDS formulation became part of standard of care. Due to serious concerns about the possibility that beta-carotene might increase the risk of lung cancer in current or former smokers, this compound was replaced by lutein/zeaxanthin in AREDS2.
The Age-Related Eye Disease Study 2
The primary aim of AREDS2 was to evaluate the efficacy of lutein/zeaxanthin and/or omega-3 long-chain polyunsaturated fatty acid supplementation in reducing the risk of developing advanced AMD.66 In AREDS2, the amount of zinc was reduced and beta-carotene was omitted from the formulation. Differences in the composition of the AREDS1 and AREDS2 formulations are shown in Table 3. Primary randomisation was to one of four groups: placebo, lutein/zeaxanthin, DHA/EPA, or a combination of the latter two. Secondary randomisation included the original AREDS1 medication, the AREDS1 formulation without beta-carotene, the AREDS1 formulation with reduced zinc, or the AREDS1 formulation without beta-carotene and low zinc. Median follow-up was 5 years, and the study included 4,203 participants aged 50–85 years who were at risk for progression to advanced AMD.67 Comparison with placebo in the primary analyses showed no significant reduction in progression to advanced AMD, with no apparent effect of beta-carotene elimination or lower-dose zinc on progression to advanced AMD. However, an excess of lung cancers was observed in the beta-carotene versus no beta-carotene group (p=0.04), which were mostly in former smokers. Secondary analyses confirmed that lutein/zeaxanthin was more appropriate than beta-carotene in the AREDS-type supplements.55 Nonetheless, evaluation of the main effects of lutein/zeaxanthin in the entire study cohort that received the lutein/zeaxanthin supplement compared with the cohort that did not receive lutein/zeaxanthin showed a significant reduction of progression to advanced AMD (p<0.05), confirming the overall benefit and rationale for an AREDS-type formulation in AMD. The AREDS2 study, however, has a number of limitations including its complex randomisation scheme with numerous subgroups, which could have resulted in the lack of statistical power needed to confirm its primary endpoint.8
The role of dietary supplements in age-related macular degeneration
There is general agreement that both the AREDS1 and the AREDS2 formulations exert their positive effects via antioxidant properties.59,67 The available evidence shows that all AMD patients should be given advice to increase the consumption of green leafy vegetables and to eat fatty fish, at least twice a week.26 A Mediterranean type diet may also provide additional benefits in other age-related diseases that go well beyond AMD.26
Given its potential benefits, it is unsurprising that a wide number of commercially-available dietary supplements are based on, or make reference, to the AREDS studies. These include those which include, but are not limited to, the compounds present in the original or second AREDS2 formulation (e.g., Centrum Silver®, Pfizer, New York, NY, US) as well as those specifically dedicated to age-related eye diseases with additional supplements (e.g., PreserVision®, Bausch & Lomb Incorporated, Rochester, NY, US). Such formulations may be particularly beneficial for individuals, such as the elderly, who cannot achieve the necessary levels of these micronutrients with diet alone. The AREDS2 formulation with 10 mg lutein/day and 2 mg zeaxanthin/day can now be considered as the standard of care for reducing the probability of advanced AMD in patients with substantial risk factors for progression to severe visual loss, and there is evidence that subjects receiving AREDS2-type supplements might have stabilisation and improvement of best-corrected visual acuity.8
It is current consensus that patients with moderate or advanced AMD should be advised to use AREDS-based supplements, while current or ex-smokers are advised to avoid formulations with beta-carotene.26 This is also considered sound advice given the overall favourable safety profile and lack of side effects observed with the AREDS formulations. These dietary modifications may provide the opportunity to delay the onset of AMD, and perhaps progression of the disease. However, there is less consensus on the effects that AREDS-based supplementation may have on prevention of AMD.
The oral supplements proposed have several advantages. In particular, supplements are readily-available and do not require a prescription. Moreover, a nutritional supplement based on the AREDS2 formulation is not known to have any adverse effects. Taken together, if recommended by experts in AMD, patients are likely to show high levels of compliance with the formulation. This latter point is especially important, and ophthalmologists and other healthcare providers caring for patients with AMD should be aware of the importance that their advice is likely to have regarding the benefits of oral for AMD.68
Adherence to supplementation in patients with age-related macular degeneration
In spite of the potential positive effects on public health, a variable adherence to the use of the supplementation in patients with AMD has been repeatedly described. More specifically the available studies have reported an adherence to AREDS recommendations ranging from 36–88%.69–74 Failure to comply with AREDS recommendations may be ascribed to a number of reasons including the lack of awareness of the importance of supplementation in prevention of AMD, the poor collaboration with ophthalmologists regarding the nutritional support, the fear of potential interactions with other medicine, and the additional cost of antioxidant supplementation. Interestingly the rate of adherence tends to reduce in proportion to the increase of patients’ age and duration of the disease, since just 38% of patients over 80 were taking supplementation compared with 45% of patients aged 50–65.69 Moreover, the rate of adherence was reported to be 41% during the first year of therapy, reducing to 36% after the third year.69 Patient education seems essential in the attempt to reduce all the modifiable AMD risk factors, especially regarding supplementation. Intensive patient education including a policy of providing verbal and written instructions along with verbal repetition of these instructions from each staff member on each patient visit could double the adherence to supplementation.75 To increase the patient adherence, it may be hypothesised that the use of a nutrition oral supplement in orodispersible microgranules, instead of tablets, would improve the assumption. Indeed, the size of tablets, even when small, are not so easy to swallow, which is relevant is certain patient populations, especially the elderly. Overall, ophthalmologists should stress the importance of nutritional supplementation in AMD patients in order to implement the awareness of the role of nutrition in AMD.







