Currently four pharmacotherapies have been approved in the US for the treatment of diabetic macular edema (DME)—the antivascular endothelial growth factor (VEGF) therapies ranibizumab (Lucentis®), a binding fragment, and aflibercept (EYLEA®), a fusion protein, and the intraocular corticosteroids dexamethasone (Ozurdex®) and fluocinolone acetonide (ILUVIEN®). Although having four approved treatment options provides a choice for managing the disorder, it is important that retina specialists determine the optimal way to use them in clinical practice. This review examines these different therapies and their application in DME and highlights the advantages of administering a corticosteroid as a slowrelease implant providing therapeutic levels of drug for up to 36 months.
Primary Efficacy and Safety of these Pharmacotherapies from Phase III Clinical Trials Ranibizumab
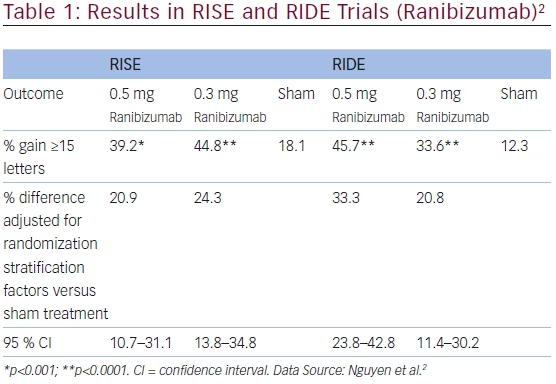
Ranibizumab was evaluated in the Diabetic Retinopathy Clinical Research Network (DRCR.net) study and the RISE and RIDE trials.1,2 The multicenter, randomized DRCR clinical trial enrolled 854 study eyes of 691 patients with visual acuity (VA) of 20/32 to 20/320 (approximate Snellen equivalent) and DME involving the fovea. Treatment groups were sham injection and prompt laser (n=293), 0.5 mg ranibizumab and prompt laser (n=187), 0.5 mg ranibizumab and deferred (≥24 weeks) laser (n=188), or 4 mg triamcinolone and prompt laser (n=186). The main outcome measure was best corrected VA (BCVA) and safety at 1 year. Both the ranibizumab and prompt laser group and the 0.5 mg ranibizumab and deferred laser group had a significantly higher VA letter score from baseline to 1 year compared with the sham and prompt laser group (+9 ±11; p<0.001, +9 ±12; p<0.001, respectively, versus +3 ±13) measured in Early Treatment Diabetic Retinopathy Study (ETDRS) letters. Furthermore, the change in the triamcinolone and prompt laser group was not significant (+4 ±13; p=0.31). VA outcomes at 2 years were similar to the 1-year results. All treatment groups exhibited a similar reduction in mean central subfield thickness, which was greater than that in the sham and prompt laser group. From baseline the reductions were -131 ±129, -137 ±136, -127 ±140, and -102 ±151 for the ranibizumab and prompt laser, ranibizumab and deferred laser, triamcinolone and prompt laser, and sham and prompt laser, respectively. Improvement in VA was comparable in the sham and prompt laser and both ranibizumab treatment groups at 1 and 2 years in the subset of 273 eyes that were pseudophakic at baseline. In the 62 pseudophakic eyes at baseline treated with triamcinoline and prompt laser, VA improvements were better than for phakic eyes and comparable to that of the pseudophakic eyes in the ranibizumab groups and superior to that of pseudophakic eyes treated with sham and prompt laser at years 1 and 2. In terms of safety, no systemic events attributable to study treatments were observed. However, injection-related endophthalmitis occurred in three eyes (0.8 %) with ranibizumab treatment while increased intraocular pressure (IOP) and cataract surgery were more frequent in the triamcinolone group. RISE (NCT00473330) and RIDE (NCT00473382) were two parallel, multicentre, double-masked, sham injection-controlled, randomized trials with identical methodologies.2 Adult patients with DME vision loss (BCVA) 20/40–20/320 Snellen equivalent and central subfield thickness ≥275 μm on time-domain optical coherence tomography (OCT) were recruited. Patients received either monthly intravitreal injections of ranibizumab (0.5 or 0.3 mg) or sham injections. Macular laser was available perprotocol- specific criteria. The main outcome measure was the proportion of patients who gained ≥15 ETDRS letters in BCVA at 24 months from baseline. In the RISE study, a total of 377 patients were randomized (0.3 mg ranibizumab n=125, 0.5 mg ranibizumab n=125, sham n=127). At month 24, significantly more patients treated with ranibizumab gained ≥15 letters compared with the sham group (see Table 1). A total of 382 patients were randomized in RIDE, (0.3 mg ranibizumab n=125, 0.5 mg ranibizumab n=127, sham n=130). The results indicated that significantly more patients treated with ranibizumab gained ≥15 letters versus shamtreated patients (see Table 1).
In addition, in the ranibizumab groups, retinopathy generally improved as did macular edema. Furthermore, fewer macular laser procedures were performed in ranibizumab-treated patients than sham patients (over 24 months, mean 0.3 –0.8 versus 1.8 and 1.6, respectively).
Endophthalmitis was observed in four ranibizumab patients, but overall ocular safety was consistent with previous studies. The possible effects from systemic VEGF inhibition, namely total incidence of deaths from vascular or unknown causes, nonfatal myocardial infarctions, and nonfatal cerebrovascular accidents occurred in 4.9 and 5.5 % of sham patients and 2.4 to 8.8 % of ranibizumab patients. Dose-dependent increases in stroke was observed in the ranibizumab-treatment groups.
Aflibercept
Aflibercept was compared with laser treatment in two parallel, doublemasked, randomized, phase III, 52-week trials with similar design.3 VISTADME (NCT01363440) was conducted across 54 sites in the US and VIVIDDME (NCT01331681) at 73 sites in Europe, Japan, and Australia. In total, 872 patients (eyes) with type 1 or 2 diabetes mellitus with DME and central involvement were enrolled. Eyes were treated with either macular laser photocoagulation or aflibercept as five initial 2 mg intravitreal monthly injections followed by either 2 mg every 4 weeks or 2 mg every 8 weeks. The primary efficacy endpoint was change from baseline in BCVA ETDRS letters at week 52. In both VISTA and VIVID from baseline to week 52 the mean BCVA gains were significantly greater with aflibercept treatment
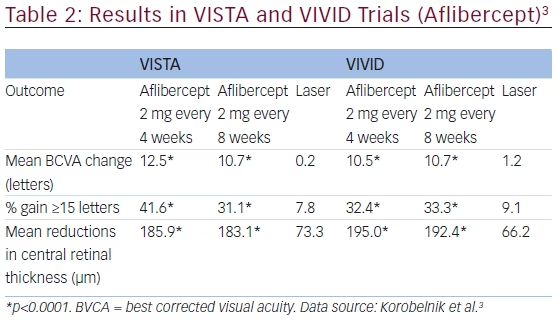
(p<0.0001) (see Table 2). Similar significant efficacy was observed in BCVA gains in both studies (p<0.0001). In addition, mean reductions in central retinal thickness (CRT) were significantly higher in patients treated with aflibercept (p<0.0001).
The incidence of serious adverse events including the Anti-Platelet Trialists’ Collaboration-defined arterial thromboembolic events and vascular deaths and ocular and nonocular adverse events were similar in all treatment groups. In particular, no cases of endophthalmitis or events suggestive of it such as hypopyon were observed.
Dexamethasone Intravitreal Implant
Two randomized, multicenter, masked, sham-controlled phase III clinical trials of dexamethasone intravitreal implant with identical protocols were completed and data pooled for analysis (Ozurdex MEAD study group).4 A total of 1,048 DME patients with BCVA 20/50 to 20/200 Snellen equivalent and CRT ≥300 μm by OCT were randomized in a 1:1:1 ratio to dexamethasone intravitreal implant 0.7 mg, 0.35 mg or sham procedure with a 3-year follow-up and ≤40 scheduled visits. Patients could be retreated no more often than every 6 months and laser treatment was not used in these studies. Patients treated at month 36 were followed for 39 months. The achievement of ≥15 ETDRS letters improvement in BCVA from baseline to the end of study was the predefined US Food and Drug Administration (FDA) primary efficacy endpoint. Over the 3 years of the study the mean number of treatments given was similar between groups (see Table 3). Patients treated with either dose of dexamethasone achieved a greater 15-letter improvement in BCVA and in the average

reduction in CRT than the sham group from baseline to study end. The rates of cataract-related adverse events were 67.9 %, 64.1 %, and 20.4 % in phakic eyes for dexamethasone 0.7 mg, 0.35 mg, and sham treatments, respectively. Generally, IOP increases were controlled with or without medication. Trabeculectomy was required by 0.6 % (two patients) in the dexamethasone 0.7 mg group and 0.3 % (one patient) with 0.35 mg.
Fluocinolone Acetonide
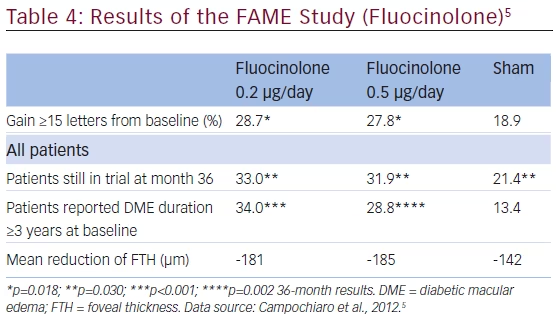
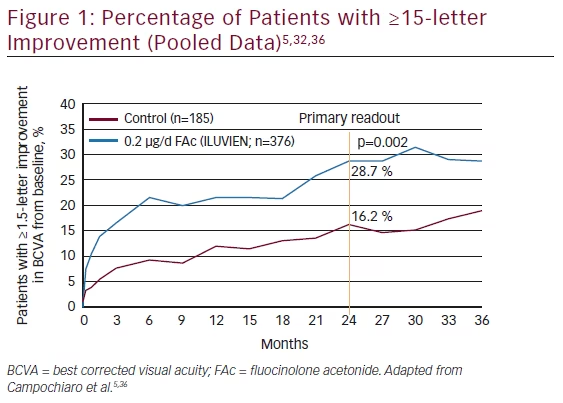
The Fluocinolone Acetonide for Diabetic Macular Edema (FAME) study assessed the long-term efficacy and safety of intravitreal implants that released either 0.2 μg/day (low dose) or 0.5 μg/day (high dose) in patients with DME.5 The study included two randomized, sham injection-controlled, double-masked, multicenter, 36-month clinical trials that enrolled patients with persistent DME despite ≥1 macular laser treatments. Patients had a mean duration of diabetes ranging from 16.1 to 17.1 years, mean duration of DME 3.5–3.9 years, mean BCVA 52.9–54.7 ETDRS VA score, and foveal thickness (FTH) 451.3–458.1 μm. Randomization was 1:2:2 to sham injection, low-dose implant, or high-dose implant (n=185, 375, and 393, respectively). Six weeks after receiving the study drug, patients were eligible for rescue laser treatment and additional study drug or sham injections were permitted after 1 year based on retreatment criteria. The main outcome measure was percentage of patients with improvement of ≥15 letters from baseline. More patients gained ≥15 letters score (using the last observation carried forward) in the fluocinolone groups than sham groups and the same trend occurred in patients still in the trial at month 36. (see Table 4, Figure 1). A pre-planned subgroup analysis of patients who reported DME for ≥3 years at baseline indicated a doubling of benefit compared with sham injections. In addition, fluocinolone-treated patients showed an improvement in ≥2 steps in the ETDRS retinopathy scale. In the low-dose group, 13.7 % of patients showed a ≥2 step improvement compared with 10.1 % in the high-dose group and 8.9 % in the sham group. This finding in the high-dose group may be because it released the drug for about 24 months, whereas the low-dose implant did so for 36 months and no retreatment was permitted. The majority of phakic patients treated with fluocinolone developed cataracts but following cataract surgery their visual benefit was similar to the pseudophakic patients. At 36 months incidence of incisional IOP-lowering surgery was 4.8 % and 8.1 % for the low- and high-dose groups, respectively, in a population that had to meet the current FDA label, which aims to mitigate the risk for IOP-lowering surgery.
There were several differences in the baseline characteristics from the phase III clinical trials that were the basis for the approval of the therapies. The most important ones are in the patients enrolled in the FAME study evaluating fluocinolone, where the percentage of patients who received prior macular laser is higher, but the mean duration of DME (3 years) was considerably longer than in the other trials.
Optimum Use of the Available Therapies in Clinical Practice
The availability of several therapies for the treatment of DME is beneficial but presents retinal specialists with the problem of determining how to use them in clinical practice. The current standard of care for clinically significant DME is anti-VEGF therapy. A doctor will generally start anti- VEGF therapy unless there is a clearly defined circinate exudate emanating from a defined microaneurysm outside the fovea. However, several confirmatory lines of evidence suggest that although many patients gain significant benefit from anti-VEGF therapy, a considerable number do not. The 36-month results of the RISE and RIDE trials showed an interesting difference in patients treated with ranibizumab from the start of the trials compared with sham-treated patients who were eligible to cross over to monthly 0.5 mg ranibizumab, although they were still masked.6 In the ranibizumab groups, VA outcomes at month 24 were maintained at 36 months and, similarly, improvement in CFT after 24 months was generally sustained. By contrast, patients switched to ranibizumab for 1 year showed lower average VA gains compared with those in the ranibizumab group after 1 year of treatment (2.8 versus 10.6 letters [ranibizumab 0.3 mg] and 11.1 letters [ranibizumab 0.5 mg]). This finding suggests that delayed treatment does not give the same VA improvement and supports the hypothesis that anti-VEGF therapy is effective when administered in the early disease stages.
Research has shown that as DME progresses, upregulation of inflammatory cytokines occurs and these cytokines have multiple interactions that impact the pathogenesis of the disease.7,8 A number of major inflammatory events are involved in the alteration of the blood retinal barrier and VEGF inhibition may not result in neutralization of other inflammatory molecules involved in the inflammatory cascade that leads to the breakdown of this barrier. This inflammatory cascade produces diverse anatomical and biochemical changes in the eye, such as worsening retinopathy.7,9–11 These findings reinforce the observations in the RIDE and RISE studies that anti-VEGF agents are useful in the early stages of disease when simple mechanisms induce edema, whereas at advanced stages, corticosteroids affect a greater number of inflammatory pathways. Patients will present with a range of inflammatory states and the most direct way to assess if their DME is primarily mediated by VEGF is to commence treatment with an anti-VEGF therapy.
Not all patients respond to anti-VEGF therapy. Available data, such as from the DRCR.net protocol I, indicate that approximately half of the patients are not optimally responding.1 In addition in this study about 26 % were nonresponders. In the DRCR protocol T study of 660 adults with DME, comparing the relative efficacy and safety of intravitreous aflibercept, bevacizumab, and ranibizumab in the treatment of DME, there were nonresponders in each treatment group. Improvement of ≥15 ETDRS letters occurred in 18 %, 16 %, and 15 % of patients in the aflibercept, bevacizumab, and ranibizumab treatment groups (with baseline letter score of 78-69 [equivalent to 20/32 -20/40]), respectively.12
The ability to identify factors associated with relatively good or poor outcomes would be useful to treating ophthalmologists to make more informed decisions on which patients should be treated with anti- VEGF therapy and for patient expectations. To address this question, an additional analysis of 361 eyes assigned to 0.5 mg ranibizumab (either with prompt or deferred laser) was undertaken to identify risk factors that predict treatment success or failure.13 A total of 37 baseline demographic, systemic, ocular, OCT, and fundus variables were assessed in terms of change in VA or OCT from baseline to year 1. Factors associated with either poor VA outcomes or OCT CSF thickness could not be evaluated as the number of patients with vision loss or increased CSF thickness were too small to analyze. However, after adjusting for baseline VA, younger age (p<0.001), absence of surface wrinkling retinopathy (p<0.001), and less-severe diabetic retinopathy were associated with a larger VA benefit. In addition, central subfield thickness during the first year of treatment also predicted better vision outcomes (p>0.001).
Although laser photocoagulation has been the main treatment for DME and reduces the risk for further vision loss, vision improvement is rare, occurring in approximately 12 % of patients after 3 years. Side effects include scotoma, altered colour perception, and night blindness.14,15 VEGF inhibitors demonstrate the ability to improve vision following multiple injections, but available data indicate that approximately 50 % of DME patients are not optimally managed on anti-VEGF therapy.13 The upregulation of cytokines, rather than VEGF as the predominant mediator as diabetic retinopathy progresses, suggests that anti-VEGF therapies may not be the best management over the disease course.16–19 Shortacting intravitreal steroids also improve vision although are associated with an increased incidence of IOP elevations.
In the FAME study, treatment with the fluocinolone implant showed markedly greater benefit compared with the sham-treated group in patients with duration of DME ≥3 years than those with DME <3 years.5 This finding suggests that eyes with persistent DME, which generally respond poorly to many treatments such as focal/grid laser photocoagulation, respond well to fluocinolone implants. It is unknown why sustained delivery of fluocinolone continues to provide benefit despite persistence of edema. Possibly, persistent edema exacerbates inflammation in the diabetic retina and this is accompanied by bystander damage to retinal neurones and the exacerbation of inflammation in persistent DME may cause it to exceed a critical threshold causing cell death and vision loss. This inflammation could be reduced below the critical threshold by sustained levels of fluocinolone-preserving vision. Thus maximum benefit is not achieved in patients with persistent DME just by treating the anatomic distortion of the retina but inflammation must also be addressed. A further benefit is the regression in retinopathy grade via the reduction in inflammation observed in patients treated with fluocinolone implants.
Use of Anti-VEGF Therapy in Real Life
Data from the RIDE and RISE clinical trials was based on 36 monthly ranibizumab injections, but this injection frequency is uncommon in reallife situations.16 A number of limitations exist in terms of clinical dosing, including coverage by insurance (or funding factors), and patient willingness to undergo repeated ocular injections. Treatment with aflibercept every 8 weeks gives good results. Furthermore, the long-term impact of repeat injections is not fully known, although results from several studies show good efficacy out to 5 years.20
In order to assess the clinical utilization of the anti-VEGF agents, bevacizumab and ranibizumab, together with disease monitoring in patients with branch or central vein occlusion or DME, a retrospective claims analysis has been completed.21 A total of 2,733 DME patients were included. Over the 2008, 2009, and 2010 cohorts, the mean annual number of bevacizumab injections increased (2.2, 2.5, 3.6, respectively) with mean ophthalmologist visits ranging from 4.4 to 6.5 and mean OCT examinations from 3.1 to 3.9. In addition, 57.7 % of patients received additional laser or intravitreal triamcinolone. Too few ranibizumab patients were identified for a meaningful analysis mainly because it was not reimbursed by health plans until after its approval for DME in August 2012. Even in the most recent 2010 cohort, including patients followed through 2011, <6 % of patients met the criteria for monthly injections and <16 % for monthly ophthalmologist visits. These monitoring intervals are in sharp contrast to those reported in RISE and RIDE and the +6- week administration schedule in the DRCR.net phase II bevacizumab study.22 The substantial differences in both injection frequency and the frequency of monitoring visits between the clinical studies and routine clinical practice suggests that in the clinical setting vision outcomes may be considerably lower than those reported in the clinical trials.
Furthermore, the RESTORE 12-month, double-masked, multicenter, lasercontrolled, phase III study investigated 0.5 mg intravitreal ranibizumab with either sham or active laser compared with laser and sham injections.

Although ranibizumab demonstrated significant superior change in BCVA letter score, only 22.6 % and 22.9 % of patients treated with ranibizumab or ranibizumab plus laser, respectively, gained a ≥15 BCVA letter score following seven injections. This is approximately half the percentage of patients responding in the RISE and RIDE trials probably reflecting the impact of fewer injections and more closely reflects real use.
Real-life US data are limited with ILUVIEN, but a small study is in progress in France and the initial findings are in line with the clinical trial data.23 In addition, clinical experience with ILUVIEN has supported the results of the FAME study.24
Translating the Clinical Trial Results to Clinical Practice
In clinical practice, retinal specialists use OCT as the primary driver of clinical decision-making. If the initial drug is anti-VEGF therapy and the doctor recognizes that optimal results are achieved with monthly injections, then after three to four treatments it should be possible to determine if a DME patient is an anti-VEGF responder by whatever criteria is chosen to claim effectiveness. For example, this could be complete resolution of edema or reduction in edema by a certain percent versus the baseline level. If the patient does not meet the criteria for response to an anti-VEGF therapy, possibly switching to another anti-VEGF treatment is an option since they have different levels of efficacy12 or a corticosteroid should be considered. If the decision is made to initiate treatment with a corticosteroid, it is highly likely that long-term therapy will be needed. In this situation, ILUVIEN provides 36 months of drug release with a single implant; however, the US indication requires a prior course of corticosteroid with no accompanying clinically significant rise in IOP (see Figure 2).25 At this point when use of a corticosteroid is indicated, an option for the retinal specialist is to try an Ozurdex implant or an intravitreal triamcinolone acetate injection (typically 1 or 2 mg), which will indicate whether the patient’s edema is responsive to a corticosteroid as well as the IOP response to an ocular steroid.26 The Ozurdex implant contains 700 μg of dexamethasone (in the NOVADUR® solid polymer delivery system). Animal studies evaluating the Ozurdex implant have shown that the peak concentration is reached in the retina and vitreous at day 60 and is detectable for 6 months. After the first 2 months, the steroid concentration declines until month 4, where it maintains a lower concentration until month 6.27 The ILUVIEN implant contains 190 μg of fluocinolone acetonide. In animal studies, following administration of the 0.2 μg/day implant, fluocinolone acetonide levels peaked in most tissues at day 2 or 8, reached steady state levels by month 3, and gradually decreased over the duration of the study. Fluocinolone acetonide was still present in most ocular tissues at 2 years.28,29 This finding was confirmed in the Fluocinolone Acetonide in Human Aqueous (FAMOUS) study at 36 months.30 Glucocorticosteroids can cause important side effects, including ocular hypertension, probably due to morphologic and biochemical changes in trabecular meshwork cells. Nehme et al. found that the glucocorticoid receptor binding affinity for dexamethasone, fluocinolone acetonide, and triamcinolone acetate was 5.4, 2.0, and 1.5 nM, respectively.31 Furthermore, dexamethasone significantly regulated transcripts associated with RNA posttranscriptional modifications, whereas fluocinolone acetonide, and triamcinolone acetate, respectively, modulated genes involved in lipid metabolism and cell morphology.12,31
The FAME clinical trials—analyses that supported the US indication— examined the IOP-lowering surgeries as a function of previous ocular steroid prior to study entry. The following factors were identified:
• All of the IOP-lowering surgeries occurred in those subjects with no prior history of ocular steroid injection.
• The exclusion criteria stated that a patient could not have a history of an “uncontrolled IOP response to a corticosteroid” that did not respond to topical therapy.32
• Thus none of the patients who entered into FAME who had a prior history of ocular steroid injection(s) without an uncontrolled pressure response went on to require incisional glaucoma surgery.32
Safety of ILUVIEN Treatment
ILUVIEN treatment is associated with accelerated development of cataract and elevated IOP, both known side effects of corticosteroid therapy.33 Identifying patients likely to have a clinically significant IOP response to ILUVIEN would be useful. Prior IOP response to corticosteroids might allow identification of steroid responders that might be better candidates for therapeutic options other than the ILUVIEN. As an alternative to an intravitreal corticosteroid exposure, a topical challenge could be considered.34 While failure to experience an IOP rise with a topical challenge does not guarantee a patient will not have an adverse IOP event, if they do experience an uncontrolled IOP rise, it is likely that they will have a higher risk for elevated IOP with ILUVIEN, and therefore may not be suitable candidates for the implant.
Excellent visual outcomes were reported following cataract surgery, of pseudophakic patients, 31.6 % gained 15 letters, while those who underwent cataract surgery gained 42.3 %.
Concerns that inflammation caused by phacoemulsification can exacerbate DME have been raised, but patients with ILUVIEN have been shown to not have worse outcomes after surgery, most likely because steroids were already in the eye.35
In the FAME study, the majority of patients (>95 %) did not require incisional surgery to treat elevated IOP and 62 % of patients did not require any IOPlowering medication during the 36-month study period.5
Conclusion
Approximately 50 % of DME patients show varying degrees of suboptimal response to anti-VEGF therapy, as shown in several studies. In patients who do not adequately respond to this therapy, two corticosteroid dosage forms are also available.13
In patients who do not adequately respond to this therapy, two corticosteroid dosage forms are also available. Before ILUVIEN can be used, the patient should have been previously treated with a course of corticosteroids and did not have a clinically significant rise in IOP. This is consistent with the clinical approach when anti-VEGF therapy is not providing an adequate response or there is a recent history of an arterial thrombotic event and retinal specialists try a shorter-acting intraocular corticosteroid, such as intravitreal triamcinolone acetonide or Ozurdex, or they proceed with a topical corticosteroid challenge to determine if a long-acting implant is an option. This will demonstrate the possible patient’s steroid-induced IOP response profile as: no IOP response, IOP response that can be controlled with topical drops, or an uncontrolled IOP response. Once the IOP response is known, a clinical decision can be made regarding the administration of ILUVIEN. Physicians are fortunate to have four complementary pharmacotherapies to treat this serious and growing disease, although the optimal therapy for each patient needs to be carefully considered.







