Diabetic macular oedema (DMO) is an increasingly serious healthcare issue and a leading cause of blindness worldwide.1 Of the estimated 415 million people with diabetes globally, 7–12% have signs of DMO and 1–3% have visual impairment as a result.2,3 This burden affects populations in all territories and is likely to increase substantially when the prevalence of diabetes burgeons to a predicted 642 million by 2040.2–4 The availability of effective and tolerable treatments for the management of DMO is therefore critical in tackling a potential epidemic of blindness and visual impairment. Several treatments are available for DMO, including laser photocoagulation, intravitreal corticosteroids and anti-vascular endothelial growth factor (VEGF) agents. Prominent among the anti-VEGF agents is ranibizumab (Lucentis®, Genentech, California, US), which has an extensive body of clinical trial data supporting its efficacy and tolerability in DMO, and on which its approval by the US Food and Drug Administration and the European medicines Agency for use in this indication is based.5–10 This monoclonal antibody (MAb) treatment has been used successfully as monotherapy and in combination with laser photocoagulation and with other treatments.11–19 Manufacturing biological therapies, including MAbs, is an expensive process and their generally high cost has the potential to restrict usage in many territories, especially where healthcare resources are limited.20–22 Reducing the dose of MAbs could lower this cost, provided that efficacy is not compromised markedly. Currently the licensed dose for ranibizumab in DMO in Europe is 0.5 mg/month as an intravitreal injection. In the US, however, the licensed dose is 0.3 mg/month due to concerns of cardiovascular risks in diabetic patients. This article reports the findings of literature searches of clinical studies that investigated the comparative efficacy and safety and cost-effectiveness of the 0.5 mg dose of ranibizumab in DMO compared with the lower 0.3 mg dose. These searches aimed to identify if there was superior efficacy in terms of visual acuity (VA) and central retinal thickness (CRT) or safety for either dose level in DMO treatment and whether the lower dose could be used as an alternative in many cases and thereby reduce costs.
Literature search methods and results
Combined searches were made in BIOSIS Previews®, British Library Inside Conferences, Embase®, Embase® Alerts and MEDLINE®, with no date restriction. Title and abstract terms searched for included MeSH terms for clinical studies in DMO, or diabetic retinopathy restricted to ranibizumab and 0.5 mg or 0.3 mg. In total, 157 unique references were identified. After removal of irrelevant articles and duplicate reports of the same studies, 47 relevant studies were identified. Of these, four studies included groups given only 0.3 mg ranibizumab as a comparator or single therapy,
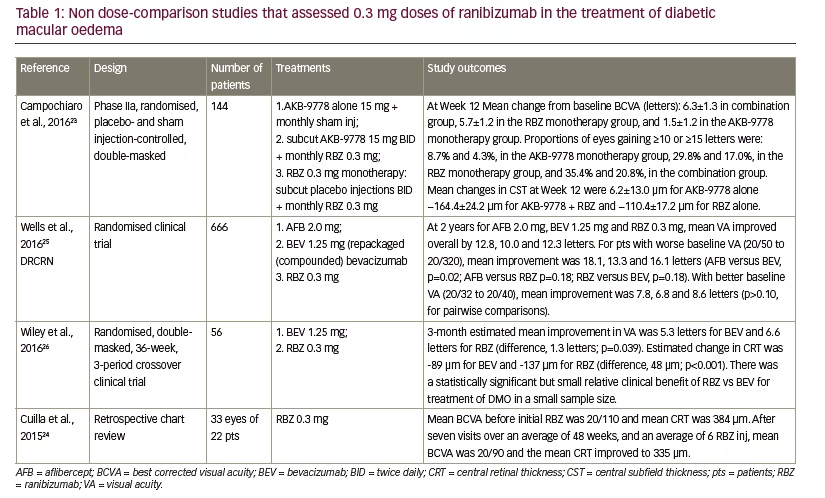
28 studies included groups given only 0.5 mg ranibizumab and only two studies directly compared the 0.3 mg and 0.5 mg ranibizumab doses. The designs, patient dispositions and findings of these categories are summarised in Tables 1, 2 and 3 (only completed studies with >40 patients for the ranibizumab 0.5 mg dose are tabulated).
The effect of different ranibizumab dosing regimens, the efficacy of ranibizumab compared with other treatments for DMO and in combination with other agents is outside the scope of this article.
Studies including only a 0.3 mg ranibizumab dose group – in diabetic macular oedema
There have been few studies that evaluated the 0.3 mg ranibizumab dose as a discrete group in the treatment of DMO (see Table 1). The four relevant studies ranged from 22 to 666 patients in size (mean 222 patients) and treatment durations varying from three months to two years.23–26 In three of the studies, ranibizumab monotherapy was a comparator against alflibercept, or bevacizumab or compared or in combination with AKB-9778 (a small-molecule competitive inhibitor of vascular endothelial-protein tyrosine phosphatase).23,25,26 One study was a retrospective chart review.24 Treatment durations were from one month to two years, during which best-correct visual acuity (BCVA) improved from +5.7 to +12.8 letters and improvements in VA. In one study there were no significant differences at the end of two years in VA for ranibizumab 0.3 mg compared with aflibercept 2.0 mg or bevacizumab 1.25 mg (p=0.18 for both) for patients with worse baseline VA (20/50 to 20/320).25 Results were similar in patients with better baseline VA (20/32 to 20/40). In another study ranibizumab 0.3 mg was significantly better than bevacizumab 1.25 mg in terms of VA -6.6 letters versus 5.3 letters (p=0.039).26 Reductions in CRT were -89 and -137 μm during treatment for bevacizumab and ranibizumab, respectively (p=<0.001). The ranibizumab treatment in these studies was well tolerated with no new adverse events profiles, i.e., these profiles being similar to those reported for the phase III studies.27,28
These studies had varying designs, study endpoints, patient populations and treatment durations, making direct comparisons difficult. However, a strong overall message that emerges from these studies is that 0.3 mg dose of ranibizumab is effective and has potential as a clinical treatment in DMO which was similar to or superior to that of some other treatments.
Studies including a 0.5 mg ranibizumab dose group (not 0.3 mg) in diabetic macular oedema
A greater body of clinical study evidence supports the use of the 0.5 mg dose of ranibizumab than the 0.3 mg dose in DMO treatment. Twelve key studies (mainly >100 patients) that investigated patients treated with ranibizumab 0.5 mg dose in DMO as single or comparator groups are summarised in Table 2.11,15,16,18,29–35 Patient populations among this group ranged from 41 to >600, with a mean of 249 patients. Treatment durations varied from five months to five years. The mean improvement in VA ranged from +6.1 to +10.3 letters (BCVA) with some studies reporting 46–60.8% of patients showing a ≥10-letter improvement during treatment. Reductions in CRT (where reported) ranged from -118.7 μm to -214.0 μm. In four of these studies (READ-2, RESPOND, RETAIN and REVEAL – see below for definitions of study names), ranibizumab 0.5 mg monotherapy was compared with either laser photocoagulation alone or a combination of ranibizumab 0.5 mg and laser treatment.11,16,33,35 In each case, laser treatment alone produced poorer improvements in VA and CRT than ranibizumab 0.5 mg. Combinations of ranibizumab 0.5 mg and laser treatment produced no better or poorer efficacy than ranibizumab 0.5 mg alone. In the DRCRNet study, patients with DMO received monthly ranibizumab and either laser treatment given promptly or deferred for ≥24 weeks.15 After 2 years, there was vision loss of ≥10 letters in 9% and 8% of patients, in the prompt and deferred laser treatment groups, respectively, and improvements of ≥10 letters in 46% and 58%, respectively. A total of 56% in the deferred
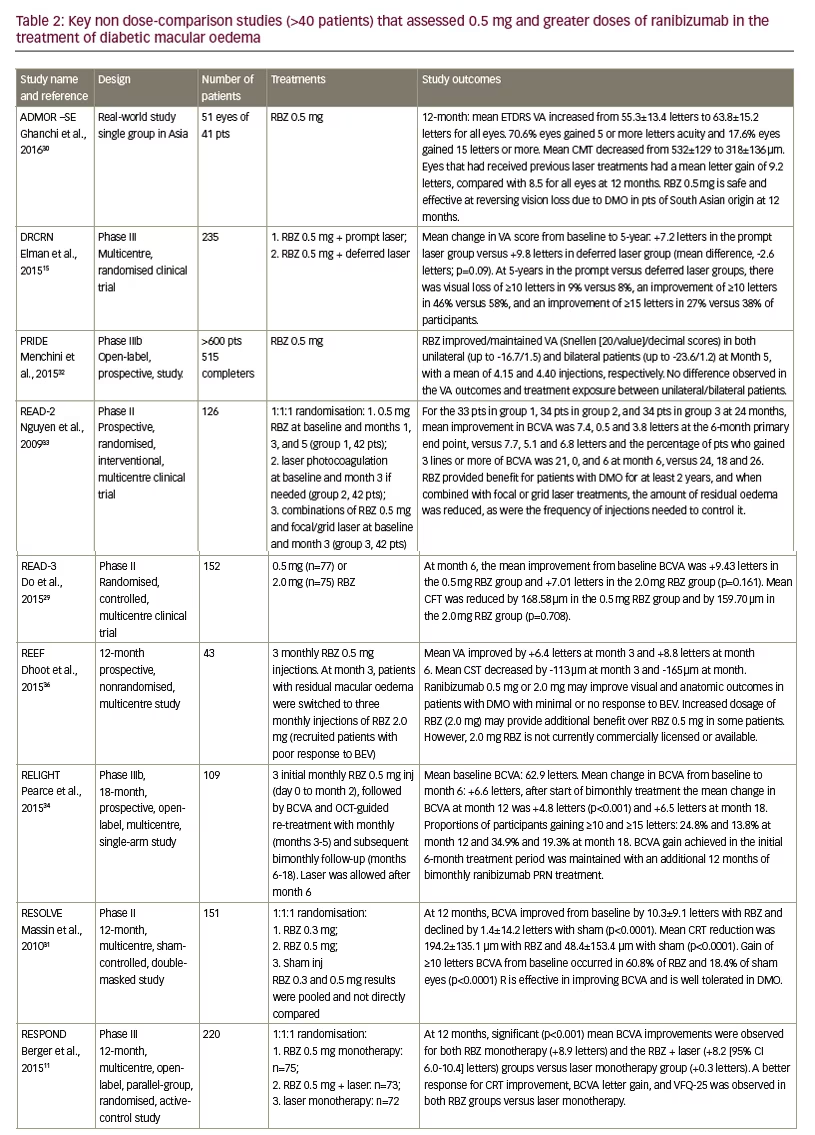
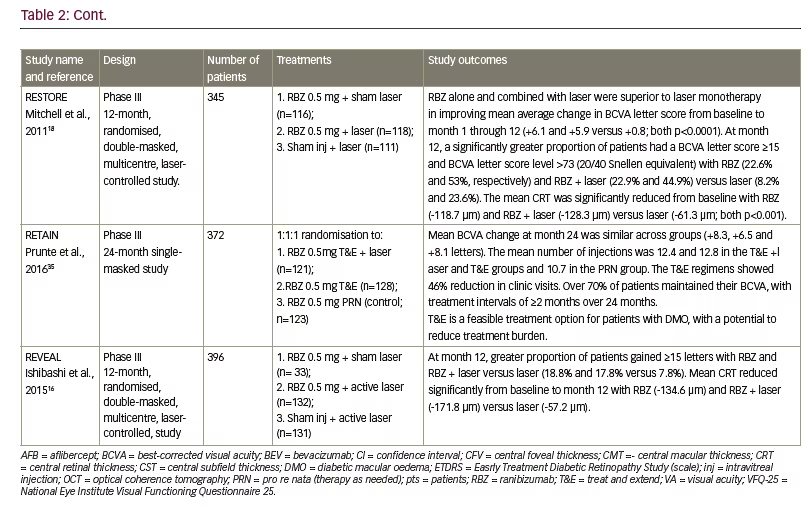
treatment group received no laser treatment at all over 5 years. This suggested that prompt laser treatment was not advantageous over deferred treatment and that for many patients, ranibizumab 0.5 mg is effective as monotherapy.
In the READ-3 study, patients with DMO were treated with either ranibizumab 0.5 mg or 2.0 mg. Improvements in BCVA were numerically lower with the higher dose (+9.43 versus +7.01 letters) but there was no statistically significant difference.29 The reduction in CRT was also lower with the higher dose. In the REEF study, patients with DMO were treated initially with ranibizumab 0.5 mg for three months and those with residual DMO were switched to ranibizumab 2.0 mg.36 VA had improved by +6.4 letters at month three and by +8.8 letters after month six. The study authors concluded that the 2.0 mg dose could provide additional benefit in some patients.
The RESOLVE study included groups of patients with DMO who were initially randomized to ranibizumab 0.3 mg and 0.5 mg, and sham injections but after month one, dose-doubling was permitted.31 This meant that there was overlap in the doses received by the two active treatment groups. In the analysis, the results of these groups were therefore pooled and compared with sham treatment; a direct comparison of the 0.3 mg and 0.5 mg results was thus not possible.
In all of these trials using the 0.5 mg dose, ranibizumab was considered safe and well tolerated. The occurrence and type of adverse events and serious adverse events were similar to patients receiving sham injections. Adverse events that were reported at low frequency (<3%) with ranibizumab over 12 months included conjunctival haemorrhage, eye pain, nasopharyngitis, hypertension, arterial thromboembolic events such as myocardial infarction. Serious ocular adverse events were reported in 1.8% of patients and included vitreous haemorrhage, retinal ischaemia, retinal artery occlusion and endophthalmitis.27 As with the 0.3 mg ranibizumab dose studies, the designs, treatment durations and endpoints were variable in the 0.5 mg dose studies but they showed consistent efficacy and tolerability in differing patient groups and locations. Overall, the study results for 0.3 mg or 0.5 mg intravitreal ranibizumab show similar efficacy for both doses.
Studies directly comparing 0.3 mg and 0.5 mg ranibizumab doses in diabetic macular oedema
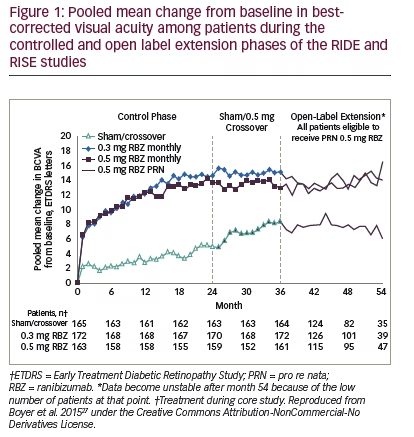
Only two large-scale studies have directly compared the ranibizumab 0.3 mg and 0.5 mg doses in DMO.27,28,37 These were the Phase III RIDE and RISE studies that were run from 2007–2012 and involved 759 adults with DMO having BCVA 20/40 – 20/320 and CRT ≥275 μm. (see Table 3). The study designs were identical; participants were randomised 1:1:1 to monthly ranibizumab 0.3 mg or 0.5 mg or sham injections for 24 months. After that, the remaining patients could opt to enter the open-label extension in which sham-treated patients could switch to 0.5 mg ranibizumab. After 36 months, all patients were switched to pro re nata (PRN)-based treatment for a maximum of 2 years.
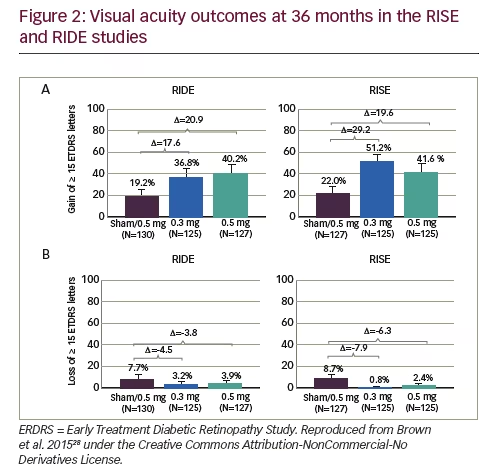
For the primary endpoint, the proportions of patients in both studies gaining ≥15 letters BCVA, at 24 and 36 months were similar in the ranibizumab 0.3 mg- and 0.5 mg-treated groups but markedly higher than in sham-treated patients. Indeed, the pooled changes in BCVA in the 0.3 mg treated groups was numerically higher than 0.5 mg at both time points (see Figure 1).27 Plots of changes in the proportions gaining ≥15 letters and changes in proportions with <15 letter loss over 36 months also show similar benefits from patients receiving either ranibizumab doses compared with those who received sham treatment (see Figure 2). During months 24–36, when the previously sham-treated patients were switched to ranibizumab 0.5 mg, there was an improvement in VA, but this did not match the improvements seen in the groups treated with either 0.3 mg or 0.5 mg ranibizumab throughout.27 This indicated that delaying ranibizumab treatment has a
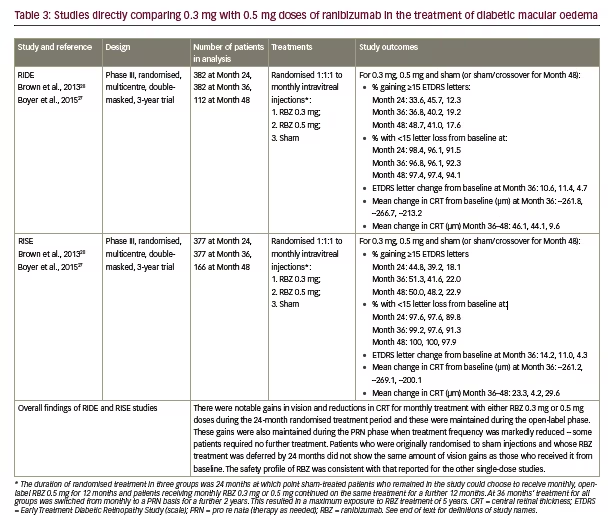
negative impact that cannot be fully reversed. During months 36 to 54, PRN-based treatment generally maintained the improvements VA seen at Month 36 in all three groups.
There were similarly marked reductions in CRT in both the ranibizumab dose groups during controlled treatment up to 36 months and these were substantially greater than reductions in the sham-treated groups.27,28 During months 36–48, however, when ranibizumab PRN treatment was instituted and patients received less intensive therapy, there was a trend towards slight retinal thickening (see Table 3). The proportions of patients showing improvements in diabetic retinopathy severity scores during controlled treatment and the open-label extension phase were similar in the ranibizumab 0.3 mg and 0.5 mg groups and greater than the sham/ crossover groups. The efficacy of the ranibizumab 0.3 mg and 0.5 mg doses in terms of their effects on VA, disease severity, CRT and long-term outcomes was therefore generally similar in the RIDE and RISE studies but tended to show a more marked effect than in studies where these doses were used singly. For safety and tolerability, during the sham-controlled treatment periods in the RIDE and RISE studies up to 24 months, the overall occurrence of ocular adverse events was similar in all groups (84.8–88.4%).28 Events such as glaucoma (2.8%, 2.8% and 2.4% for 0.3 mg, 0.5 mg and sham
crossover groups, respectively) and increased intraocular pressure (17.6%, 16.4% and 6.8%, respectively) occurred with similar frequency at both doses. Serious ocular adverse events were generally more frequent with ranibizumab 0.5 mg than 0.3 mg including angle-closure glaucoma (0%, 0.4% and 0.4%), endophthalmitis (0.8%, 0.8% and 0%), cataract (0.45 and 0.8% and 0%) and vitreous haemorrhage (0%, 0.8% and 2.8%). This pattern was continued during months 24–36 and during the subsequent PRN-based treatment phase.
Arterial thrombotic events occurred at similar rates in the two ranibizumab groups, which were higher than the sham/crossovertreated group (10.8%, 10.4% and 7.2% for ranibizumab 0.3 mg, 0.5 mg and sham/crossover groups, respectively). Among these, myocardial infarction and stroke were slightly more frequent during ranibizumab treatment. Stroke was also more common with the 0.5 mg than the 0.3 mg dose (4.8% versus 2.0%). Overall, the safety and tolerability profiles of both ranibizumab doses were similar in the RIDE and RISE studies and did not differ markedly from that of single-dose studies summarised in the previous sections.
Cost-effectiveness of ranibizumab doses in diabetic macular oedema
A few studies have reported positive cost-effectiveness findings for the ranibizumab 0.3 mg or 0.5 mg doses. One example is a 14-year cost-utility analysis using data from the RIDE and RISE studies.38 This study found that the 14-year incremental patient value gain for the 0.3 mg ranibizumab dose in DMO was 0.9981 quality-adjusted life year (QUALY). The direct cost for ranibizumab in one eye was determined to be US$30,116 and for two eyes was US$56,336. Savings from decreased depression, injury, skilled nursing facility admissions, nursing home admissions, and other vision-associated costs were estimated to be US$51,758, leaving an overall cost of US$4578. It was concluded that ranibizumab 0.3 mg confers considerable patient (human) value gain and accrues value to patients, public, insurers and society. A study in the UK, using findings from the RESTORE study, estimated that lifetime costs per patient in DMO treatment were £20,019 for ranibizumab 0.5 mg PRN, £22,930 for ranibizumab 0.5 mg treat and extend, based on 3-year treatment timeframe. 39 From these findings, ranibizumab 0.5 mg was considered costeffective and compared favourably with other DMO therapies.
Discussion
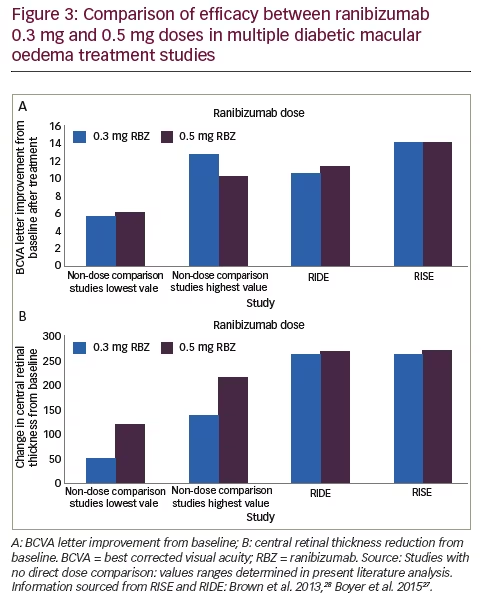
Looking at the data from trials that used only one dose of ranibizumab, it is apparent that the ranges of VA improvement in terms of increased BCVA letters and proportions of patients with increased letter scores produced by the 0.3 mg and 0.5 mg doses overlapped so that their efficacy appears substantially similar. The range of decreases in CRT also overlapped but clearly greater reductions were seen with the 0.5 mg dose (see Figure 3). The studies that are discussed above varied in design, endpoints, treatment durations and study populations so it is difficult to draw consistent and reliable conclusions that can be compared. Nevertheless, across a wide range of these studies the efficacy of the two doses appears similar. Safety findings indicate good tolerability with ocular adverse events generally occurring at similar frequencies with ranibizumab to those of sham injections.
The RIDE and RISE trials provide a more reliable basis for comparing the 0.3 mg and 0.5 mg doses of ranibizumab over long treatment durations than those that evaluated only a single dose. The overall improvements in VA and in CRT were quite similar between the two doses in both trials but of greater magnitude than seen in other studies (see Figure 3). The reasons for this greater efficacy may have been a result of more intense therapy over longer durations in the RIDE and RISE studies. The gains in VA and CRT for both doses were maintained over three years of intensive monthly treatment and continued during the further 18 months of PRN treatment. There was some degree of retinal thickening during the PRN phase suggesting that this less frequent treatment regimen may be insufficient for some patients. Patients who received sham treatment initially and were switched at 24 months failed to achieve the gains indicating that some disease progression is irreversible and early initiation of treatment following DMO diagnosis is vital. The overall findings led the authors of long-term outcomes to conclude that ‘in the pooled data from the RIDE and RISE trials efficacy was equivalent between the 0.3 mg and 0.5 mg doses’.28
The safety findings of all these trials indicate the generally favourable safety profiles and tolerability of both ranibizumab doses. Some ocular adverse events such as endophthalmitis, glaucoma and cataract may be expected with intravitreal injections but these were generally non-severe and few led to treatment discontinuation, particularly in the ranibizumab 0.3 mg group (0.4% and 1.2% for 0.3 mg and 0.5 mg, respectively at 36 months). In the RIDE and RISE studies ocular adverse events generally occurred at the same frequencies in ranibizumab and sham-treated patients. Among systemic events there was a slight increase in cardiovascular thrombotic events with ranibizumab including increased stroke risk. Since 40–50% of patients with DMO have bilateral DMO requiring simultaneous treatment40 and that diabetic patients have an increased risk of mortality and cardiovascular disease,41 the lower 0.3 mg dose of ranibizumab was considered more suitable than the 0.5 mg dose for use in DMO and was consequently licensed for this indication by the US Food and Drug Administration.28
Cost-effectiveness studies indicate that both ranibizumab doses improve quality of life and were considered by investigators to provide value when the cost of other treatments, welfare and the high financial impact of vision impairment to the patient are taken into account. However, the high cost of this treatment is still likely to limit or even prevent its use in many territories and in patients with inadequate healthcare insurance. Since the ranibizumab 0.3 mg dose appears to be equivalent to the 0.5 mg dose in terms of efficacy and has potential safety benefits, using the lower dose could be appropriate for many patients in Europe and elsewhere to reduce costs. This would make the treatment more accessible to a wider patient population with DMO. The 0.5 mg and higher ranibizumab doses could then be reserved for some patients including more severe or refractory cases as was suggested by the authors of the REEF study (see Table 2).36 The ever-increasing prevalence of DMO is likely to drive demand for effective but lower-cost treatments that can stem a rising burden of vision loss worldwide. Using the lower 0.3 mg dose of ranibizumab across all territories could help satisfy this need.








